IJCRR - 14(11), June, 2022
Pages: 37-46
Date of Publication: 03-Jun-2022
Print Article
Download XML Download PDF
Factors Affecting the Production of Astaxanthin in the Microalgae Haematococcus pluvialis: A Review
Author: Muhsinin Soni, Aligita Widhya, Rostinawati Tina, Levita Jutti
Category: Healthcare
Abstract:Astaxanthin, a natural red pigment that belongs to the carotenoid group, has been known as a super antioxidant due to its very strong antioxidant activity (65 times higher than vitamin C, 54 times more potent than -carotene, and 14 times higher than vitamin E). Haematococcus pluvialis is known as microalgae with a high astaxanthin content. The benefit of astaxanthin in health issues is mainly its potential as the treatment for degenerative diseases caused by reactive oxygen or nitrogen species. Thus, it is important to develop Haematococcus pluvialis microalgae as a rich source of natural astaxanthin in the health and pharmaceutical industries.
Keywords: Astaxanthin, Antioxidants, Haematococcus pluvialis, Carotenoids, Microalgae, Anticancer
Full Text:
INTRODUCTION
Astaxanthin (3,3'-dihydroxy-β-carotene-4,4'dione) is a secondary metabolite belonging to the carotenoid group.1–3 Astaxanthin has a high value in the pharmaceutical, nutraceutical, and cosmetic fields because of its potent antioxidant potential with an IC50 value of 39.1 ± 1.14 ppm.4 The antioxidant activity produced by astaxanthin is 65 times higher than that of vitamin C, 54 times more powerful than β-carotene, 14 times higher than vitamin E, and 20 times stronger than its synthetic form.5 Due to its potent antioxidant activity, astaxanthin can be used to treat several degenerative diseases caused by free radicals.6
Various sources of astaxanthin in nature can be obtained from several microorganisms such as the fungus Phaffia rhodozyma, microalgae Chlorella zofingiensis, and Haematococcus pluvialis.7–9 However, of these microorganisms, H. pluvialis is known to show the highest astaxanthin accumulation capacity of up to 4% dry weight under stress conditions.10,11
The market price of astaxanthin also varies, ranging from $2,500 to 7,000/kg. In 2014, the global market potential of astaxanthin was approximately 280 tonnes for $400 million. However, more than 95% of the market is synthetic astaxanthin types that are sourced from petrochemicals. This happens because the production cost of synthetic astaxanthin is relatively cheaper than natural astaxanthin obtained from microalgae.12 This synthetic type of astaxanthin has 20 times lower antioxidant power than the natural type.5 In addition, related to safety issues, synthetic astaxanthin types are still not allowed to be consumed by humans due to differences in stereochemical form with natural type. Therefore, its use is only permitted as feed and dye for aquaculture organisms.10
Astaxanthin production can be done by various methods, including culture, chemical synthesis, and genetic engineering. The culture method can be done by adding stress induction to microalgae because it is known that H. pluvialis is a microalgae that can accumulate astaxanthin under stress. These stress conditions can be caused by several factors, including light stress,13,14 nutritional deficiency,15 salinity stress,16,17 the addition of Fe2+,18,19 and so on. In addition, another method is chemical synthesis using asta-C15-triarylphosphonium salt and C10-dialdehyde with the Wittig reaction,20 which produces synthetic astaxanthin with antioxidant activity 20 times lower than natural astaxanthin. Then another method, genetic engineering, in several research journals has been widely reported overproduction of astaxanthin in several microorganisms such as fungi and bacteria.21 This review article contains biological and physiological conditions, biochemical content, and methods of producing astaxanthin from H. pluvialis by culture and genetic engineering.
Biology of H. pluvialis
-
Taxonomy
H. pluvialisis a biflagellate unicellular microalgae that lives in freshwater. According to Lorenz (1999),22 the classification of H. pluvialis microalgae is as follows:
.png)
-
Habitat
The habitat of H. pluvialisis spread evenly in the world, especially in temperate areas. This microalgae has been isolated in Europe, Africa, North America, and Himachal Pradeslv India.23,24 H. pluvialis is also found in various environmental conditions with extreme climates, which may be lethal to other types of microalgae. This is because H. pluvialis can defend itself by forming encysts (cells become closed with a thick membrane) quickly when under stress and extreme conditions.25
-
Morphology
The cell structure of H. pluvialis is similar to that of some groups of volvocalean green microalgae. The life cycle of H. pluvialis consists of four phases with different cellular morphology, namely macrozooid (zoospore), microzooid, palmella, and hematocyst (aplanospore).10,26 The following is the morphology of H. pluvialis microalgae with descriptions (A) Motile macrozoid cells (zoospores) with a size <10 m or 20 m, (B) Microzoid cells, (C) Palmella cells with accumulation of astaxanthin, (D) Hematocyst cells with accumulation of astaxanthin with size > 50 m.
The macrozoid, microzoid, and palmella phases are also known as the green vegetative phase. The microzoid phase (zoospore) is when the cell has a spherical, elliptical or pear-shaped shape with two flagella of the same length and appears anteriorly and has cup-shaped chloroplasts (Figure 1A). In this phase, with the optimum environment, flagellated cells undergo rapid division and growth, producing 2-8 daughter cells.
.png)
Figure adapted from Shah et al. (2016),27 which is licensed under the Creative Commons Attribution License.
However, suppose the environmental conditions are unfavorable (stress). In that case, the cell will remove the flagella and begin to expand in size by forming an amorphous structure layered on the inside of the extracellular matrix and develops into non-motile cells called palmella (Figure 1B).28 In this phase, the H. pluvialis cell wall thickens and consists of three layers. The first layer is a trilaminar layer containing materials such as sporopollenin, an algaenan that is resistant to acetolysis.29 According to Kim et al. (2016),30 the content of algaenans in the cell walls of H. pluvialis microalgae will inhibit the extraction process using several solvents such as acetone, methanol, dichloromethane. The second and third layers contain mannose and cellulose.28,31,32
The hematocyst phase was also referred to as the non-motile phase with astaxanthin accumulation (Fig. 1C and 1D). This phase occurs when the state of stress continues. This stress state can be in the form of nutritional deficiency, light stress with a certain intensity, salinity stress, and the addition of certain chemicals that can induce stress. Under these conditions, the palmella will turn into an asexual form or hematocyst (aplanospore). Mature hematocysts accumulate large amounts of carotenoids, especially astaxanthin, stored in lipid droplets in the cytoplasm.28
.png)
Figure 2: Illustration of the life cycle of H. pluvialis.
Figure adapted from Wayama et al. (2013),33 which is licensed under the Creative Commons Attribution License.
After the environmental conditions return to normal and optimal, the hematocyst (aplanospore) will germinate again to form a microzoid (zoospore) which will re-initiate the start of a new vegetative growth cycle (Figure 3).33
Biochemical Content
The cellular content of the H. pluvialis microalgae varies between the green phase and the red phase due to its unique life cycle. The biochemical range of H. pluvialis in the green phase and red phase according to 34 is listed in Table 1.
.png)
Description (-): no data reported
According to Table 1, H. pluvialis produced 81.2% Astaxanthin (including ester) in the red phase. This amount is the highest compared to primary metabolites (Proteins, Lipids, Carbohydrates) and other carotenoid compounds. The green phase does not produce astaxanthin. H. pluvialis enters a logarithmic phase (growth phase) and produces more primary metabolites during this phase.
Astaxanthin
-
Sources of Astaxanthin
Natural sources of astaxanthin are found in several organisms, including algae, bacteria, fungi, salmon, shrimp, lobster.35 But for the mass production of astaxanthin, microorganisms such as fungi and microalgae are more widely used because of their rapid growth. Some of the natural astaxanthin-producing microorganisms are listed in Table 2.
.png)
According to Table 2, H. pluvialis is the microalgae that produce the most significant amount of astaxanthin (up to 3.8%) (excluding esters). HPLC and LC-MS methods for analyzing astaxanthin compounds. The biomass of H. pluvialis was homogenized and extracted with acetone several times. The extracts were combined, evaporated with a rotavopar, and then redissolved in acetone.
-
Astaxanthin Biosynthesis
Astaxanthin biosynthesis in H. pluvialis is a complex series of processes that occur under stress conditions along with triacylglycerol (TAG) accumulation. Both compounds are deposited in lipid droplets in the cytosol during the red phase. The formation of astaxanthin begins with the glycolysis process, which produces pyruvate and glyceraldehyde-3-phosphate (G3P). Furthermore, pyruvate, together with glyceraldehyde-3-phosphate (G3P), will form the compound Isopentenyl Pyrophosphate (IPP) as the primary precursor in the synthesis of carotenoids.
Astaxanthin belongs to the carotenoid group, is one of the C40 tetraterpenes synthesized from the isoprene unit Isopentenyl Pyrophosphate (IPP). In principle, IPP synthesis can originate from two different pathways: the mevalonate pathway (MVA) occurring in the cytosol and the non-mevalonate pathway (MEP) or the 1-deoxy-D-xylulose-5-phosphate (DOXP) pathway occurring in chloroplasts.43–45
In H. pluvialis, IPP is synthesized from the non-mevalonate pathway. Furthermore, IPP undergoes isomerization to dimethylallyl diphosphate (DMAPP). Some research results indicate that the conversion is catalyzed by the enzyme isopentenyl pyrophosphate isomerase (IPI) encoded by the ipi1 and ipi2 genes during astaxanthin accumulation.2 However, the results of another study also stated that neither of the ipi1 and ipi2 genes was increased as long as H. pluvialis cells accumulated astaxanthin.46 Another study reported that another enzyme with similar activity, namely 4-hydroxy-3-methylbut-2-enyl diphosphate reductase (HDR), was more likely to be responsible for catalyzing the intermediate conversion of IPP to DMAPP.46–48
Elongation of the isoprene chain begins with a DMAPP molecule, and the addition of three IPP molecules is catalyzed by the enzyme geranyl-geranyl pyrophosphate synthase (GGPS).49,50 The next step of this process is the formation of the compound C20 geranyl-geranyl pyrophosphate (GGPP). GGPP is converted to C40-phytoene as a precursor of astaxanthin and other carotenoids with the help of the phytoene synthase (PSY) enzyme encoded by the psy gene coupled with the head-to-tail condensation of two GGPP molecules.50
Figure 4. Biosynthesis of astaxanthin in H. pluvialis27
The formation of lycopene takes place through four desaturation steps catalyzed by two enzymes, namely the enzyme phytoene desaturase (PDS), which is encoded by the pds gene and z-carotene desaturase (ZDS), which is encoded by the zds gene.51,52 The desaturation reaction will increase the number of conjugated double bonds in the carbon chain to form chromophore groups in carotenoids, change the colorless molecule of phytoene to the lycopene, and produce a red color.50
.png)
Lycopene undergoes cyclization catalyzed by the enzyme lycopene cyclase (LCY-e and LCY b), which is encoded by the lcy gene. Cyclization of carotenoid biosynthesis in most organisms produces a-carotene (a precursor to lutein) and β-carotene (a precursor to carotenoids including astaxanthin). The last two oxygenation processes are catalyzed by the β -carotene ketolase (BKT) enzyme encoded by the bkt gene, and the β-carotene hydroxylase (CrtR-b or BKH) enzymes encoded by the bkh or crtR-b genes are the final stages of astaxanthin synthesis.53–55
-
Pharmacological Activity of Astaxanthin
Astaxanthin as a nutraceutical has a variety of pharmacological activities, including those in Table 3.
.png)
.png)
.png)
Description: ROS: Reactive Oxygen Species; KATO-III: Human gastric carcinoma cell line; SNU-1: Human gastric carcinoma cell line; GLUT4: Glucose transporter type 4; IRS-1: Insulin receptor substrate-1; INF-γ: Interferon -γ; IL-2: Interleukin-2; ALT: alanine aminotransferase; AST: aspartate aminotransferase.
Astaxanthin Production Method
-
Culture Method
Culture System
In general, microalgae culture systems are divided into three: photoautotrophic, heterotrophic, and mixotrophic systems. H. pluvialis allows cultivation using all three methods, either with open or closed systems.27 In the phototropic system, microalgae are very dependent on light as an energy source and CO2 as a carbon source, both light from lamps or the sun. In heterotrophic systems, microalgae growth requires organic carbon as an energy source. Commonly used organic carbon substrate sources include glucose, acetate, and glycerol.62 In this condition, the microalgae cell density achieved was higher than the phototropic condition, so that the cost required for harvesting was lower. The mixotrophic system is a combination of phototropic and heterotropic methods. The microalgae that grow in this system can assimilate sunlight and organic carbon as energy sources simultaneously or alternately.
Culture systems, especially those that require light (photoautotrophic), are divided into 2: closed and open culture systems. Advantages and disadvantages of culture with closed and open systems can be seen in Table 4.
.png)
.png)
Cultural Conditions
The success of microalgae culture is strongly influenced by several important factors, including nutritional and environmental factors. H. pluvialis culture conditions can be seen in Table 5.
.png)
Stress Induction
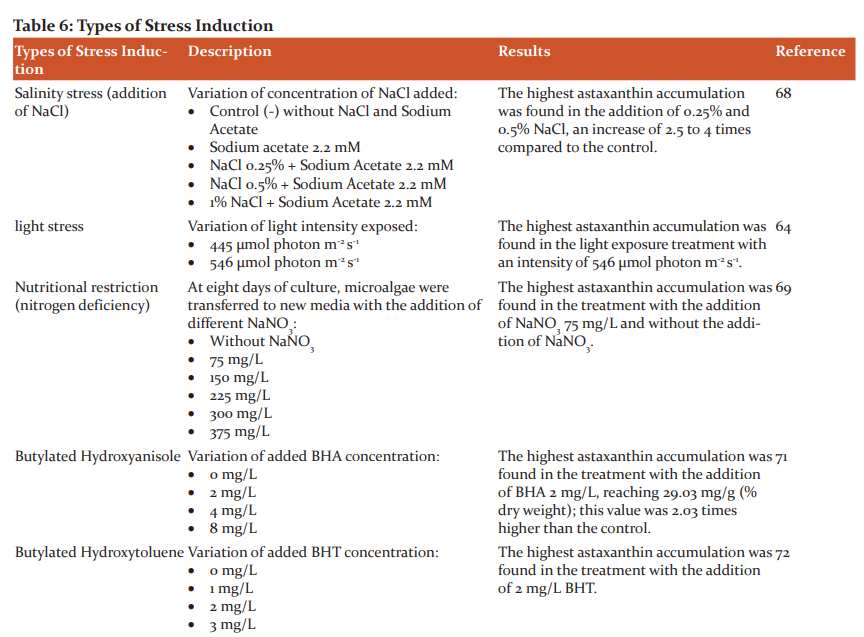
H. pluvialis can accumulate astaxanthin under stress. The accumulation of astaxanthin is a response of microalgae to protect themselves from oxidative stress conditions.70 Several studies of stress induction, either physically or chemically, are listed in Table 6.
 Harvesting
Harvesting
An efficient harvesting technique is an important step that must be done to get a high concentration of harvested biomass. Several harvesting methods commonly used for H. pluvialis are flotation and centrifugation methods.27,73
Centrifugation is a method of harvesting microalgae based on the application of rotary power to precipitate microalgae cells so that they are separated from the liquid growth medium. The separation was supported by the difference in density between the microalgae cells and the liquid medium in which the cells grew. The centrifugation method can produce microalgae in a paste with a solid content of up to 15%. Several studies also show that the faster the centrifugation cycle, the microalgae biomass obtained can reach up to 95%.
This method is a separation process based on gravity where the microalgae cells attach to air or gas bubbles so that the cells float on the surface. Under these conditions, microalgae cells can be harvested easily. In certain types of microalgae, cells can flow naturally if the lipid content in the cells increases. In the flotation method, the need for operational costs will be even greater if it involves flocculants.
Extraction
Extraction methods commonly used include maceration and percolation.74 Astaxanthin is a lipophilic compound and is soluble in organic solvents and oils. Organic solvents such as acetone, DMSO, methanol, n-hexane, and vegetable oils such as olive oil, soybean oil, and corn oil have been used for astaxanthin extraction.35,74
-
Genetic Engineering
The development of biotechnology today also supports the use of microalgae as a producer of bioactive compounds. Most of the bioactive compounds produced by microalgae are secondary metabolites, which have low cellular production. So that the mass production of bioactive compounds from microalgae culture (without modification and engineering) is still not efficient; on the other hand, the synthesis of bioactive compounds with chemicals, especially astaxanthin compounds, will produce products that are stereochemically different from the natural products so that they are not allowed to be consumed by humans.5 However, with the advancement of biotechnology, the "factory" of microalgae biomass can be made more optimal. The use of science and methods of mutagenesis and genetic engineering is a solution that must continue to be developed. Several studies on the production of carotenoid compounds such as astaxanthin by genetic engineering are listed in table 7.

.png)
CONCLUSION
This study provides valuable pieces of information on astaxanthin, particularly regarding its pharmacology activities, biosynthesis pathway, various methods of its production in microalgae, harvesting, and extracting techniques, that will add insight to uncover the critical area of astaxanthin from microalgae.
ACKNOWLEDGEMENT
The authors thank the Ministry of Education, Culture, Research, and Technology (grant No. 10/E1/KPT/2021). The funding source did not involve in the study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
CONFLICT OF INTEREST
None declared.
FUNDING
Source of funding from Ministry of Education, Culture, Research, and Technology (grant No. 10/E1/KPT/2021).
AUTHOR’S CONTRIBUTIONS
Soni Muhsinin (SM) is responsible for the conception of the study. SM and Widhya Aligita (WA) collected the articles, drafted them, and wrote the manuscript. Tina Rostinawati (TR) and Jutti Levita (JL) supervised, reviewed, and finalized the manuscript. All authors have read and approved the final manuscript to be published.
References:
1. Chen G, Wang B, Han D, Sommerfeld M, Lu Y, Chen F, et al. Molecular mechanisms of the coordination between astaxanthin and fatty acid biosynthesis in Haematococcus pluvialis (Chlorophyceae). Plant J. 2015 Jan;81(1):95–107.
2. Gao Z, Meng C, Zhang X, Xu D, Zhao Y, Wang Y, et al. Differential Expression of Carotenogenic Genes, Associated Changes on Astaxanthin Production and Photosynthesis Features Induced by JA in H. pluvialis. 2012;7.
3. Su Y, Wang J, Shi M, Niu X, Yu X, Gao L. Metabolomic and network analysis of astaxanthin-producing Haematococcus pluvialis under various stres sconditions. 2014;170:522–9.
4. Infant SB, Elumalai S, Rajes KG. Antioxidant and Anti-skin cancer potential of a Ketocarotenoid pigment Astaxanthin isolated from a green microalga Haematococcus pluvialis Flotow. 2016;7.
5. Capelli B, Baghci D, Cysewsky GR. Synthetic astaxanthin is significantly inferior to algal-based astaxanthin as an antioxidant and may not be suitable as a human nutraceutical supplement. 2013;
6. Mularczyk M, Michalak I, Marycz K. Astaxanthin and other Nutrients from Haematococcus Pluvialis—Multifunctional Applications. Mar Drugs. 2020 Sep 7;18(9):459.
7. Liu J, Sun Z, Gerken H, Liu Z, Jiang Y, Chen F. Chlorella zofingiensis as an Alternative Microalgal Producer of Astaxanthin: Biology and Industrial Potential. 2014;12:3487–515.
8. Lorenz RT, Cysewsky GR. Commercial potential for Haematococcus microalgae as a natural source of astaxanthin. 2000;18:160–7.
9. Mulders KJM. Phototripic Pigment Production with Microalgae: Biological Constraint and Opprtunities. 2014;50:229–42.
10. Butler T, McDougall G, Campbell R, Stanley M, Day J. Media Screening for Obtaining Haematococcus pluvialis Red Motile Macrozooids Rich in Astaxanthin and Fatty Acids. Biology. 2017 Dec 26;7(1):2.
11. Zhang Y, Shi M, Mao X, Kou Y, Liu J. Time-resolved carotenoid profiling and transcriptomic analysis reveal mechanism of carotenogenesis for astaxanthin synthesis in the oleaginous green alga Chromochloris zofingiensis. Biotechnol Biofuels. 2019 Dec;12(1):287.
12. Panis G, Rosales Carreon J. Commercial astaxanthin production derived by green alga Haematococcus pluvialis: A microalgae process model and a techno-economic assessment all through production line. 2016;18:175–90.
13. Hu Q, Huang D, Li A, Hu Z, Gao Z, Yang Y, et al. Transcriptome-based analysis of the effects of salicylic acid and high light on lipid and astaxanthin accumulation in Haematococcus pluvialis. Biotechnol Biofuels. 2021 Dec;14(1):82.
14. Li Q, Zhao Y, Ding W, Han B, Geng S, Ning D, et al. Gamma-aminobutyric acid facilitates the simultaneous production of biomass, astaxanthin and lipids in Haematococcus pluvialis under salinity and high-light stress conditions. Bioresour Technol. 2021 Jan;320:124418.
15. Chekanov K, Lobakova E, Selyakh I, Semenova L, Sidorov R, Solovchenko A. Accumulation of astaxanthin by a new Haema tococcus pluvialis strain BM1 from the White Sea coastal rocks (Russia). 2014;12:4504–20.
16. Cui J, Yu C, Zhong D, Zhao Y, Yu X. Melatonin and calcium act synergistically to enhance the coproduction of astaxanthin and lipids in Haematococcus pluvialis under nitrogen deficiency and high light conditions. Bioresour Technol. 2020 Jun;305:123069.
17. Luo QL, Wang KP, Xiao K, Wang CG, Hu ZL. A rapid and high-quality method for total RNA isolation from Haematococcus pluvialis. Genet Mol Res [Internet]. 2017 [cited 2021 Sep 14];16(2). Available from: http://www.funpecrp.com.br/gmr/ year2017/vol16-2/pdf/gmr-16-02-gmr.16029614.pdf
18. Hong ME, Hwang SK, Chang WS, Kim BW, Lee J, Sim SJ. Enhanced autotrophic astaxanthin production from Haematococcus pluvialis under high temperature via heat stress-driven Haber–Weiss reaction. Appl Microbiol Biotechnol. 2015 Jun;99(12):5203–15.
19. Zhao Y, Xing H, Li X, Geng S, Ning D, Ma T, et al. Physiological and Metabolomics Analyses Reveal the Roles of Fulvic Acid in Enhancing the Production of Astaxanthin and Lipids in Haematococcus pluvialis under Abiotic Stress Conditions. J Agric Food Chem. 2019 Nov 13;67(45):12599–609.
20. Krause W, Henrinch K, Paust J, Ernst H. Preparation of Astaxanthin. 1997.
21. Xie W, Liu M, Lv X, Lu W, Gu J, Yu H. Construction of a Controllable β-carotene Biosynthetic Pathway by Decentralized Assembly Strategy in Saccharomyces cerevisiae†. 2013;
22. Lorenz RT. A Technical Review of Haematococcus Algae. 1999;
23. Pringsheim EG. Nutritional requirements of Haematococcus pluvialis and related species. 1966;2:1–7.
24. Suseela MR, Toppo K. Haematococcus pluvialis—a green alga, richest natural source of astaxanthin. 2006;90:1602–3.
25. Proctor VW. Some controlling factors in the distribution of Haematococcus pluvialis. 1957;38:457–62.
26. Elliot AM. Morphology and life history of Haematococcus pluvialis. 1934;82:250–72.
27. Shah MdMR, Liang Y, Cheng JJ, Daroch M. Astaxanthin-Produring Green Microalga Haematococcus pluvialis: From Single Cell to High-Value Commercial Products. 2016;7.
28. Hagen C, Siegmund S, Braune W. Ultrastructural and chemical changes in the cell wall of Haematococcus pluvialis (Volvocales, Chlorophyta) during aplanospore formation. 2002;37:217–26.
29. Kim JH, Park JJ, Lee BJ, Joo MK, Chun HJ, Lee SW, et al. Astaxanthin inhibits proliferation of human gastric cancer cell lines by interrupting cell cycle progression. 2016;10:369–74.
30. Kim D-Y, Durairaj V, Ramasamy P, Jong-In H, Kyubock L, Ji-Yeon P, et al. Cell-wall disruption and lipid/astaxanthin extraction from microalgae: Chlorella and Haematococcus. 2016;199:300–10.
31. Damiani MC, Leonardi PI, Pieroni OI, Cáceres EJ. Ultrastructure of the cyst wall of Haematococcus pluvialis (Chlorophy�ceae): wall development and behaviour during cyst germination. 2006;45.
32. Praveenkumar R, Lee K, Lee J, Oh YK. Breaking dormancy: an energy-efficient means of recovering astaxanthin from microalgae. 2015;17:1226–34. 33. Wayama M, Ota S, Matsuura H, Nango N, Hirata A, Kawano S. Three-dimensional lultrastructural study of oil and astaxanthin accumulation during encystment in the green alga Haematococ�cus pluvialis. 2013;
34. Grewe CB, Griehl C. The carotenoid astaxanthin from Haematococcus pluvialis. 2012;122–44.
35. Ambati RR, Phang SM, Ravi S, Aswathanarayana RG. Astaxanthin: Sources, Extraction, Stability, Biological Activities and Its Commercial Applications—A Review. 2014;12:128–52.
36. Ranga RA, Raghunath RRL, Baskaran V, Sarada R, Ravishankar GA. Characterization of Microalgal Carotenoids by Mass Spectrometry and Their Bioavailability and Antioxidant Properties Elucidated in Rat Model. 2010;58:8553–9.
37. Orosa M, Torres E, Fidalgo P, Abalde J. Production and analysis of secondary carotenoids in green algae. 2000;12:553–6.
38. Wang Y, Peng J. Growth-associated biosynthesis of astaxanthin in heterotrophic Chlorella zofingiensis (Chlorophyta). 2008;24.
39. Banerjee K, Ghosh R, Homechaudhuri S, Mitra A. Biochemical Composition of Marine Macroalgae from Gangetic Delta at the Apex of Bay of Bengal. 2009;10.
40. EFSA (European Food Safety Authority). Safety and efficacy of panaferd-AX(red carotenoid-rich bacterium Paracoccus carotinifaciens as feed additive for salmon and trout. 2007;546:1–30.
41. Kim JH, Kang SW, Kim SW, Chang HI. High-Level Production of Astaxanthin by Xanthophyllomyces dendrorhous Mutant JH1 Using Statistical Experimental Designs. Biosci Biotechnol Bio�chem. 2005 Jan;69(9):1743–8.
42. Iwamoto T, Hosoda K, Hirano R, Kurata H, Matsumoto A, Miki W, et al. Inhibition of Low-Density Lipoprotein Oxidation by Astaxanthin. J Atheroscler Thromb. 2000;7(4):216–22.
43. Eisenreich W, Rohdich F, Bacher A. Deoxyxylulose phosphate pathway to terpenoids. 2001;6:78–84.
44. Lichtenthaler HK. The 1-Deoxy-D-Xylulose-5-Phosphate pathway of isoprenoid biosynthesis in plants. 1999;50:47–65.
45. Lichtenthaler HK, Rohmer M, Schwender J. Two independent biochemical pathways for isopentenyl diphosphate and isoprenoid biosynthesis in higher plants. 1997;101:643–52.
46. Gwak Y, Hwang YS, Wang B, Kim M, Jeong J, Lee CG. Comparative analyses of lipidomes and transcriptomes reveal a concerted action of multiple defensive systems against photooxidative stress in Haematococcus pluvialis. 2014;65:4317–34.
47. Hoeffler JF, Hemmerlin A, Grosdemange-Billiard C, Bach TJ, Rohmer M. Isoprenoid biosynthesis in higher plants and in Escherichia coli: on the branching in the methylerythritol phosphate pathway and the independent biosynthesis of isopentenyl diphosphate and dimethylallyl diphosphate. 2002;366:573–83.
48. Rohdich F, Hecht S, Krieger C, Amslinger S. Studies on the non-mevalonate terpene biosynthetic pathway: metabolic role of IspH (LytB) protein. 2002;99:1158–63.
49. Britton G. Biosynthesis of carotenoids,in Carotenoids in Photosynthesis SE-4, eds A. 1993;96–126.
50. Cunningham FX, Gantt E. Genes and enzymes of carotenoid biosynthesis in plants. 1998;49:557–83.
51. Li Y, Sommerfeld M, Chen F, Hu Q. Effect of photon fluxdensities on regulation of carotenogenesis and cell viability of Haematococcus pluvialis (Chlorophyceae). 2010;22:253–63.
52. Nawrocki WJ, Tourasse NJ, Taly A, Rappaport F, Wollman FA. The plastid terminal oxidase: its elusive function points to multiple contributions to plastid physiology. 2015;66:49–74.
53. Linden H. Carotenoid hydroxylase from Haematococcus pluvialis: cDNA sequence, regulation and functional complementation. 1999;1446:203–12.
54. Steinbrenner J, Linden H. Regulation of two carotenoid biosynthesis genes coding for phytoene synthase and carotenoid hy- during stress-induced astaxanthin formation in the green alga Haematococcus pluvialis. 2001;125:810–7.
55. Vidhyavathi R, Venkatachalam L, Sarada R, Ravishankar GA. Regulation of carotenoid biosynthetic genes expression and carotenoid accumulation in the green alga Haematococcus pluvialis under nutrient stress conditions. 2008;59:1409–18.
56. Campoio T, Oliveira F, Otton R. Oxidative stress in human lymphocytes treated with a fatty acid mixture: role of carotenoid astaxanthin. 2011;25:1488–1456.
57. Ishiki M, Nishida Y. Impact of divergent effects of astaxanthin on insulin signaling in L6 cells. 2013;54:2600–12.
58. Chalyk NE, Klochkov VA, Bandaletova TY, Kyle NH, Petyaev IM. Continuous astaxanthin intake reduces oxidative stress and reverses age-related morphological changes of residual skin surface components in middle-aged volunteers. 2017;48:40–8.
59. Lin KH, Lin KC, Lu WJ, Thomas PA, Jayakumar T, Sheu JR. Astaxanthin, a carotenoid, stimulates immune responses by enhancing in-γ and il-2 secretion in primary cultured lymphocytes in vitro and ex vivo. 2016;17:44.
60. Hussein G, Goto H, Oda S, Sankawa U, Matsumoto K, Watanabe H. Antihypertensive potential and mechanism of action of astaxanthin: III. Antioxidant and histopathological effects in spontaneously hypertensive rats. 2006;29:684–8.
61. Shen M, Chen K, Lu J, Cheng P, Xu L, Dai W, et al. Protective effect of astaxanthin on liver fibrosis through modulation of TGF-1 expression and autophagy. 2014;
62. Masojidek J, Torzillo G. Mass Cultivation of Freshwater Microalgae. 2014;
63. Witono JR, Miryanti A, Santoso H, Kumalaputri AJ, Novianty V, Gunadi A. Studi Awal Pertumbuhan dan Induksi Mikroalga Haematococcus Pluvialis. 2018;2:275–81.
64. Imamoglu E, Dalay MC, Sukan FV. Influences of different stress media and high light intensities on accumulation of astaxanthin in the green alga Haematococcus pluvialis. 2009;26.
65. Zahra F. Pengaruh Penambahan Fe2+ terhadap Pertumbuhan dan Kadar Astaxanthin pada Kultur Haematococcus Pluvialis dalam Fotobioreaktor. [Bandung]: Institut Teknologi Bandung; 2017.
66. Mascia F, Girolomoni L, Alcocer MJP, Bargigia I, Perozeni F, Cazzaniga S, et al. Functional analysis of photosynthetic pigment binding complexes in the green alga Haematococcus pluvialis reveals distribution of astaxanthin in Photosystems. Sci Rep. 2017 Dec;7(1):16319.
67. Fabregas J, Dominiguez A, Regueiro M, Maseda A, Otero A. Optimization of culture medium for the continuous cultivation of the microalga Haematococcus pluvialis. 2000;53:530–5.
68. Sarada R, Usha T, Ravishankar GA. Influence of stress on astaxanthin production in Haematococcus pluvialis grown under different culture conditions. 2002;623–7.
69. Rohimawati R, Marwani E. Produktivitas Astaxanthin dari Haematococcus pluvialis pada Pemberian Konsentrasi Nitorgen Berbeda dalam Fotobioreaktor. 2017;2:1–5.
70. Brown DR, Gough LA, Deb SK, Sparks SA, McNaughton LR. Astaxanthin in Exercise Metabolism, Performance and Recovery: A Review. Front Nutr. 2018 Jan 18;4:76.
71. Shang M, Ding W, Zhao Y, Xu JW, Zhao P, Li T, et al. Enhanced astaxanthin production from Haematococcus pluvialis using butylated hydroxyanisole. 2016;236:199–207.
72. Zhao Y, Yue C, Ding W, Li T, Xu JW, Zhao P, et al. Butylated hydroxytoluene induces astaxanthin and lipid production in Haematococcus pluvialis under high-light and nitrogen-deficiency conditions. 2018;
73. Panis G. Commercial Astaxanthin Production Derived by Green Alga Haematococcus pluvialis: A Microalgae Process Model and techno-Economic Assessment All Through Production Line [Master Thesis (45EC)]. [Netherlands]: Utrecht Univer�sity; 2015.
74. Singh S, Rather AH. Extraction of Astaxanthin from The Encysted Cells of Haematococcus Pluvialis with Different Solvents. 2018;4:115.
75. Gassel S, Schewe H, Schmidt I, Schrader J, Sandmann G. Multiple improvement of astaxanthin biosynthesis in Xanthophyllomyces dendrorhous by a combination of conventional mutagenesis and metabolic pathway engineering. Biotechnol Lett. 2013 Apr;35(4):565–9.
76. Neveen AS., GhadaSI, Shaimaa ES.,Mostafa MAE. The Potential Role of Carotenoid Pigment Isolated from a New Rhodotorula Species in Ameliorating Cerebral Ischemic Stroke Experimentally. IJCRR. 15 (1), 71-80.
77. Pinki S., Priyanka S., Shreyasi, Dubey, Ayushi S. Effect Of Different Processing Methods On Polyphenolic Content And Antioxidant Activity Of Broad Beans (Vicia faba). IJCRR. 8 (13), 6-11.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License