IJCRR - 14(3), February, 2022
Pages: 06-11
Date of Publication: 01-Feb-2022
Print Article
Download XML Download PDF
Studies on Characterization and Optimization Parameters of Zinc Oxide Nanoparticles Synthesis
Author: Sirohi Sandeep, Chinta Koteswara Rao, Andanamala Vijay Rajesh, Talapudi Sruthi, DV Surya Prakash
Category: Healthcare
Abstract:Introduction: Zinc oxide nanoparticles (ZnO NPs) are metal oxide nanomaterials and are widely used in a variety of sectors due to their peculiar physicochemical properties. In the present work, the zinc oxide nanopowder was first chemically synthesized and then detailed characterization is done by various analytical techniques, such as UV-Vis spectroscopy and Fourier Transform Infra-Red (FTIR). Aim: Studies on Characterization and Optimization Parameters of zinc oxide nanoparticles synthesis Methods: In the current study, nanoparticles of zinc oxide were synthesized by a wet chemical process with the help of precursors zinc nitrate and sodium hydroxide the properties of starch were used as a stabilizing material. The powder form of zinc oxide nanoparticles that appeared white in color was purified through the centrifugation process and was stored in an Eppendorf tube at 40C for further work. Results: Synthesis conditions of zinc oxide nanoparticles were optimized using different parameters are metal ion concentrations (0.1M to 0.8M), contact time variations (0.5hr to 4hr), the effect of pH (4 to 9), and effect of temperatures (55 to 850C). The resultant ZnO NPs powder was characterized by UV-Vis spectroscopy and FTIR analysis. Conclusion: This research work was showed the best results of ZnO NPs synthesis and the further part will continue on antimicro�bial activity.
Keywords: Zinc nitrate, Zinc oxide, Nanoparticle, Optimization, FTIR, UV-VIS Spectra
Full Text:
INTRODUCTION
Nanobiotechnology essentially combines biotechnology and nanotechnology to produce bio-synthetic, natural, and nanoscale-based goods.1 Nanomaterial have evolved as effective antimicrobial properties for killing microbes. Nanomaterials have a unique potential to kill germs, and researchers are currently focusing their efforts on microbial resistance to metal ions, antibiotics, and the formation of resistance stains.2 The recent development in porous and nanometric materials prepared by non-conventional methods has opened new vistas in the area of zinc oxide nanoparticles.3 Nano-sized innovations have risen over the previous decade due to advancements in sciences and technology. However, metal nanoparticles have occupied utmost consideration in recent years because of their exceptional properties and potential application in catalysis, plasmonics, optoelectronics, biological sensor, and pharmaceutical applications.4 Synthesis and characterization of nano-scale substances have been an important subject for studying in applied sciences. Metal oxide nanoparticles have been extensively utilized in the previous year’s.5 The synthesis of metal oxides has made it possible to utilize the properties of nanoparticles at ground level. Now a day’s zinc oxide nanoparticles have been given utmost importance because of their unique properties like UV filtration, antibacterial, high catalytic, antifungal, and photochemical action, etc. Inorganic materials, for instance, metal and metal oxides have been laid utmost importance over the previous decade because of their unique ability to understand tedious processes. Metal oxides, for instance, ZnO, TiO2, CaO, and MgO are of peculiar interest since they are very stable under unusual tedious process conditions, these metal oxides are also known as comfortable materials for the use of human beings and animals.6 The application of silver and zinc oxide nanoparticles has been seen as an important tool to stop infectious diseases because of their uncommon antimicrobial properties. The intrinsic property of different metal oxide nanoparticles is characterized its shape, size, composition, morphology, crystallinity, and various other properties.7 Zinc oxide nanoparticles are used as an effective antibacterial and antifungal agent when they are incorporated into various materials, such as cosmetics, paints, and textiles. Zinc oxide has been used in personal care products due to its bacteriostatic, fungistatic, and bactericidal properties. Zinc oxide, on the other hand, has unusual properties when reduced to nano-size compared to its bulk equivalent. Zinc oxide nanoparticles have unique features that allow them to interact with bacteria more effectively.8 As a result, less zinc oxide nanoparticles are needed to achieve the same or better fungal static and biostatic behavior. It is an excellent chemical additive for cotton fabrics in the textile sector due to its non-toxicity and skin compatibility.9
MATERIALS AND METHODS
Chemicals
Zinc nitrate, soluble starch, sodium hydroxide, MES buffer, Tris-HCl, Zinc nitrate hexahydrate.
Synthesis of zinc oxide nanoparticles
1% soluble starch was prepared by dissolving 0.5gm in 500 ml of lukewarm distilled water. Zinc nitrate 14.874gm (0.1mol) was added in the above solution followed by constant stirring in magnetic stirrer for 60 minutes for the complete dissolution of zinc nitrate.10 0.2 M (300ml) NaoH was added dropwise under constant stirring. After 2hr of stirring the completed reaction solution was incubated overnight. The supernatant solution was then discarded very carefully and the rest of the solution was centrifuged 10 min for 10,000 rpm.11 The supernatant was discarded again. The nanoparticle obtained were washed twice using distilled water to remove by-products and excessive starch bound with nanoparticles. After that, it was dried overnight at 80°C. Zinc hydroxide was transformed to ZnO NPs during drying.
Optimization of process parameter for zinc oxide nanoparticles synthesis
a. Effect of metal ion concentration on zinc nanoparticles synthesis
The concentration of Zinc nitrate hexahydrate is an important parameter. Different concentrations ranging from (0.1–0.8M) of zinc nitrate hexahydrate used as substrate and effect on the NP synthesis was ascertained.12
b. Effect of contact time on zinc nanoparticles synthesis
Zinc nanoparticles synthesis was evaluated at different (30 min to 3 hour) contact time taking absorbance with UV–VIS spectroscopy.13
c. Effect of temperature on zinc nanoparticles synthesis
Impact of temperature on zinc nanoparticles synthesis was evaluated at different temperature (25°C-95°C) of incubator, keeping other conditions are same.
d. Effect of pH on zinc nanoparticles synthesis
The production of zinc nanoparticles was optimized by varying the pH (5-9). The pH was adjusted to 5.5 –10.5 (MES buffer, 4mM for pH 5.5-6.5 and Tris-HCl, 4mM for pH 7.5-10.5) at an interval of pH 1.0.
Characterization techniques for synthesized ZnO NPs
a.UV-VIS Spectra Analysis of ZnO Nanoparticles
Ultraviolet-Visible Spectroscopy technique is used for optical characterization of nanoparticles. Its absorption spectra is ranges from 300 to 700nm scale.14,15 V-530 ( JASCO) model of this spectrometer is used for this research work. (I am modified above the matter)
b. Fourier Transform Infrared spectrometer (FTIR)
It is identity of various varieties of chemical substances is viable by this technique.16 The spectroscopy merely dependent on the records that particles soak up particular frequencies which might be feature of their shape named as resonant frequencies.
RESULTS AND DISCUSSION
Chemical Synthesis of Zinc oxide nanoparticles
Wet chemical method as described by Taguchi was used for the fabrication of the zinc oxide nanoparticles using sodium hydroxides, zinc nitrate as precursors while starch acted as stabilizing agent in reaction Figure-1A. After synthesis powder form of zinc oxide nanoparticles seems to be white in colour as seen in Figure-1B. The same method was applied by Vigneshwaran et al. in 2006 and they found small crystals of zinc oxide nanoparticles white in colour. Reddy et al., 2006 also used top-down approach for the quick biosynthesis of nanoparticles. Liqianga et al. (2006) found the photoluminescence spectrum of zinc oxide nanocrystals with an excitation wavelength at 325nm with the same method.17 Chakarborti et al. (2010) used sol-gel injection for the synthesis of zinc oxide nanoparticles inside a porous silica matrix they successfully synthesized Zn2SiO4 crystals using similar approach.

-
ZnO NPs synthesis. (B) Synthesized ZnO nanoparticles in Eppendorf after purification
Optimization of ZnO NPs synthesis
Optimization of ZnO NPs was done during the present study with respect to particle size distribution. Wet chemical method was used for optimal conditions. Following steps are involved in the optimization of ZnO NPs.
Effect of metal ion concentrations on zinc nanoparticles synthesis
The impact of different concentrations (0.1M to 0.8M) of zinc nitrate for the formation of ZnO nanoparticles was optically monitored by UV-Vis spectroscopy and FTIR. It is clearly seen from figure-3 that absorption spectrum of ZnO NPs was in the range of 380 - 420 nm at different concentration of zinc nitrate.18 The optimum concentration for the synthesis of ZnO NPs was exhibited at 0.3M concentration resulting in sharpest peak among all concentrations at 390 nm (figure-7), which stored in anhydrous form in eppendorf (figure-2). Increasing concentrations of zinc nitrate were used in optimization of metal ion concentration. It clearly seen from the present study that concentration of zinc nitrate from 0.1M to 0.3M exhibited upward formation of peaks however at concentration 0.3M the peak was highest subsequently concentration of zinc nitrate from 0.4M to 0.8M, there was hump formation in the peaks indicating poor absorption of zinc nitrate.19 Hence, it was concluded on the basis of present results that increasing concentration of metal ions beyond the limit leads to decreasing the formation of ZnO NPs. Similar observations for the optimization of nanoparticles with different metal ion concentrations were also reported by Kumar et al. (2013). For the better control of synthesis, optimization and morphological characteristics Bhattacharjee et al. (2011) also revealed similar results. Jamdagni et al. (2016) revealed that when they increased the concentration of flower extract from 0.25ml to 1ml, the maximum absorption was reported at 1ml of flower extract in 50ml of zinc acetate. If we alter this volume either increase or decrease then simultaneously decrease in the absorption values, their results also corroborated the present findings.

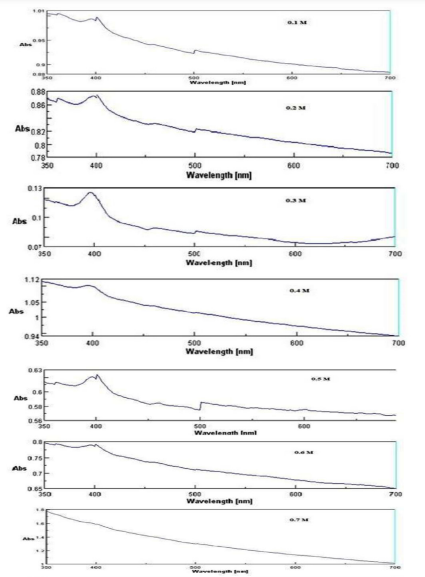 .
.

Effect of contact time on zinc nanoparticles synthesis
The contact or incubation time is one of the influencing factor for the synthesis of nanoparticles. The obtained results of UV- Vis spectra are depicted in figure-4a and 4b. For contact hours, a spectrum up to 3 hours was recorded. The sharpest absorption peak was observed in 393 nm (after 2 hr) which remain stable after 3 hr.20 While in 0.5 and 1 hr no peak were observed. Chan (2008) found the effect of contact time on the synthesis of nanoparticles, their findings were also similar to our findings. They further also revealed that sharpness and enhancement of UV-Vis absorption peaks with an increase in incubation period. However, in the present work it is optimized at 2 hour duration and stable up to several hours as illustrated in figure-4b.
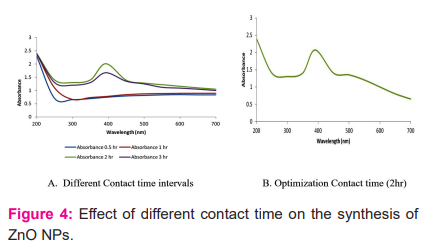
Effect of temperature on zinc nanoparticles synthesis
Temperature is another most important physical factor for the synthesis of ZnO NPs, UV-Vis spectroscopy signify the role of temperature during nanoparticles synthesis.21 The optimization of the suitable temperature for the reduction of nanoparticles in the reaction process was optimized between 250C to 950C however no spectra were observed between 250C to 450C. Spectra between 550C to 650C are visualized similar pattern with low and extremely wide absorption peak, while absorption peak increased considerable on 750C as depicted in figure- 5a. A sharp and stable peak was observed at 850C with the absorption 390 nm figure- 5b. No prominent peak was observed at 950C. No major peak shifted during optimization process in the synthesis of ZnO NPs.22 However it was noticed that high temperature (850C) was most favourable for ZnO NPs synthesis. Rajendran et al. (2010) found different particle size of ZnO, depending on the range of pH and Temperature. Dissipation of zinc ion in nuclei was increased with higher temperature due to enhanced rate of reduction. Shaymurat et al. (2011) also worked on the ZnO NPs with different ranges of temperature and reported similar findings.
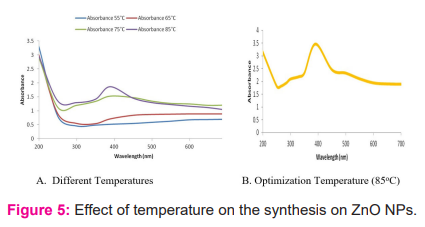
Effect of pH on zinc nanoparticles synthesis
pH has an unavoidable factor for the nanoparticles synthesis. The effect of pH in the synthesis of ZnO NPs was also estimated by using UV-Vis spectroscopy.23 The production of ZnO nanoparticles were mostly determined by the pH of the reaction media. The present results are depicted by figure – 9a and 10b. It was also observed during the present research work, that there was no absorption peak at 4 pH, However at pH 7 and 8 absorption band were also formed broadening band patterns indicates synthesis of large size of nanoparticles while highest absorption and peak formation occurred at pH 9 which suggest the optimum pH for the synthesis of ZnO NPs.24 Jamdagni et al. (2016) discussed about the two major governing factors i.e. pH and temperature in biological synthesis of ZnO nanoparticles. The absorbance values increased when the pH level was raised from 9 to 12. At pH 12 and 13 characteristic absorption peak was recorded while at pH 9 no absorption peak i.e. straight line was reported, However, at pH 12 the sharpness and absorbance both were recorded better that is called as optimized pH.
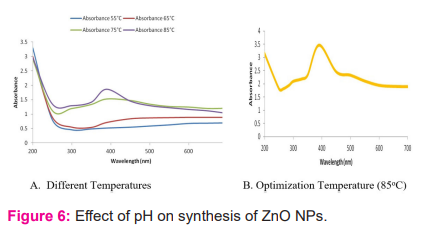
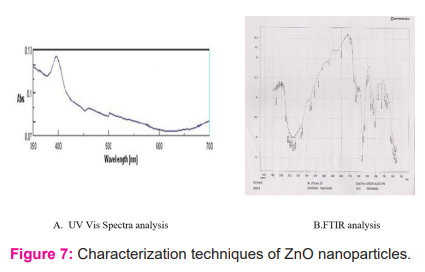
Characterization techniques of ZnO nanoparticles
UV-VIS Spectra Analysis of ZnO Nanoparticles
The absorption spectra of chemically synthesized ZnO NPs prepared with the help of soluble starch are depicted in Figure-7a. The spectrum of these nanoparticles were in the range of 390-420 nm with absorption maxima at 392 nm in the figure. Similar results were also presented in the study carried out by Vigneswaran et al. (2006).
FTIR analysis of ZnO Nanoparticles
FTIR spectroscopy analyzed Zn oxide (control) and synthesized ZnO nanoparticles which were obtained after optimization through FTIR.25 Various peaks were found at 3169,3119, 2939, 2429, 2339, 2074, 1919, 1764, 1634, 1509, 1384, 1154, 1104, 1019, 934 and 834 cm-1 in Zn oxide samples as control. The IR spectrum of Zn oxide showed sharp absorption bands at 3169 and 3119 cm-1 that may be due to O-H bond stretching vibrations N-H stretch. Peaks at 2429 to 1919 correspondence to stretching vibrations of C ≡ C stretch of alkynes. The 1764 to 1509 peaks result from the stretching bands of function group C = O. The peak of 1384 to 1104 results from aromatic amines and the peak 1019 and 934 indicates the C-N stretching (Rastogi and Arunachalam, 2011). The peaks of below 834 and above 614 indicate alkanes and C-H band. Further, the FTIR spectra of ZnO NPs represents peaks at 3444, 3384, 3342, 3288, 3225, 3201, 2973, 2946, 2898, 2745, 2343, 1767, 1638,1590, 1591, 1497, 1383, 1236, 1152, 1077, 1020, 924, 831, 759 and 738 cm-1 due to symmetric stretching vibration of surface bounded zinc oxide molecules. Therefore, on the basis of these results it is concluded that Zn oxide worked as both reducing as well as capping agents in ZnO NPs formation. Morley et al. (2007) also found similar observations with respect to the bands at 1390 and 1155 can be attributed to the C-N stretching vibrations of the aliphatic and aromatic amines. Here FTIR spectrogram of ZnO NPs at 0.8 M concentration of Zinc nitrate was shown in figure 7b.
CONCLUSION
Metal nanoparticles have been extensively used in the recent past. Inorganic materials like, metal and metal oxides have gained a lot of interest in the recent years due to their properties which help them withstand adverse process conditions. Now a day’s zinc oxide nanoparticles are considered as important material because of their number of unique properties like antibacterial, antifungal, UV filtration property, high catalytic and photochemical activity. Zinc oxide nanoparticles are useful as effective antibacterial and antifungal agents. In the current study, nanoparticles of zinc oxide were synthesized by wet chemical process with the help of precursors zinc nitrate and sodium hydroxide the properties of starch were used as a stabilizing material. The powder form of zinc oxide nanoparticles appeared white in color was purified through centrifugation process and was stored in eppendorf tube at 40C for further work. Synthesis conditions of zinc oxide nanoparticles were optimized using different parameters, are metal ion concentrations (0.1M to 0.8M), contact time variations (0.5hr to 4hr), effect of pH (4 to 9), and effect of temperatures (55 to 850C). The resultant Zno NPs powder was characterized by UV-Vis spectroscopy and FTIR analysis.
ACKNOWLEDGEMENT
The authors would like to thanks the Department of Biotechnology, Meerut Institute of Engineering and Technology, Meerut, Uttar Pradesh.
Financial Support: Meerut Institute of Engineering and Technology, Meerut provide the financial support for this research work.
Conflict of interest: None declared
Authors’ Contribution: Dr.Sandeep Sirohi and Dr.Vijay Rajesh are conceptualized in this research while Dr. Surya Prakash and Dr. Koteswar rao are supervised the study.
References:
-
Keerthika E, Ishwarya K, Jayashree L, Maripandian S, Nivetha, Irivichetty Sai Chandana. Potential Antibacterial Activity of Green Synthesized Copper Nanoparticles and its Characterization. Int J Cur Res & Rev. 2021; 13(19): 27-32.
-
Suganya S, Ahila SC, MuthuKumar B, Vasantha Kumar M. Evaluation of Impact Strength of Silver Nano-Particles Reinforced Heat-Activated Polymethylmethacrylate (Pmma) Resin at Various Proportions. Int J Cur Res & Rev. 2021, 13(13): 13-17.
-
Mishra PK, Mishra H, Ekielski A, Talegaonkar S, Vaidya B. Zinc oxide nanoparticles: a promising nanomaterial for biomedical applications. Drug Discov. 2017; 12: 1825-1834.
-
Lopez C, Paez JR. Synthesis and characterization of ZnO nanoparticles: Effect of solvent and antifungal capacity of NPs obtained in ethylene glycol. Appl Phys A, 2017; 123(12): 748-752.
-
Tyagi PK, Mishra M, Khan N, Tyagi S, Sirohi S. Toxicological study of silver nanoparticles on gut microbial community probiotic. Env Nano Monit Manag. 2016, 5:36-43.
-
Paul S, Ban KD. Synthesis, Characterization and the Application of ZnO Nanoparticles in Biotechnology. Int J Adv Che Engg Bio Sci, 2014; 1(1): 1-5.
-
Arora AK, Devi S, Jaswal VS, Singh J, Kinger M, Gupta VD. Synthesis and Characterization of ZnO nanoparticles. Oriental J Chem, 2014;30(4): 1671- 1679.
-
Talam S, Rao KS, Gunnam N. Synthesis, Characterization, and Spectroscopic Properties of ZnO Nanoparticles. Int Sch Res Notice Nano. 2012; 1: 1-6.
-
Xie Y, He Y, Irwin PL, Jin T, Shi X. Antibacterial activity and mechanism of action of zinc oxide nanoparticles against Campylobacter jejuni. App and Env Mic. 2011; 77: 2325-2331.
-
Xu M, Fujita D, Kajiwara S, Minowa T, Li X, Takemura T, Iwai H, Hanagata N. Contribution of physicochemical characteristics of nano-oxides to cytotoxicity. Biomaterials. 2010; 31: 8022-8031.
-
Rai M, Yadav A, Gade A. Silver nanoparticles as a new generation of antimicrobials, Biot Adv. 2009; 27: 76-83.
-
Wang H, Xie C, Zeng D. Controlled growth of ZnO by adding H2O. J of Cry Gro. 2005; 277(1–4): 372-377.
-
Vigneshwaran N, Nachane RP, Balasubramanya RH, Varadarajan PV. A novel one-pot green synthesis of stable silver nanoparticles using soluble starch. J Carb Res. 2006; 341(12): 12-18.
-
Reddy VR. Gold nanoparticles: synthesis and applications. Synlett. 2006; 11:1791-1792.
-
Liqianga J, Yichuna Q, Baiqia W, Shudana L, Baojianga J, Libina Y, Weia F, Honggang F, Jiazhong S. Review of photoluminescence performance of nano-sized semiconductor materials and its relationships with photocatalytic activity. Sol Ene Mat Sol Cells. 2006; 90:1773-1787.
-
Chakarborti S, Chatterjee T, Joshi P, Poddar A, Bhattacharyya B, Singh SP, Gupta V, Chakarborti P. Structure and Activity of Lysozyme on Binding to ZnO Nanoparticles. J Langmuir. 2010; 26 (5): 3506–3513.
-
Kumar SS, Venkateswarlu P, Rao VR, Rao GN. Synthesis, characterization and optical properties of zinc oxide nanoparticles. Int Nano Letters. 2013; 3(30): 1- 5.
-
Bhattacharjee CR, Purkayastha DD, Nath A. Homogeneous Chemical Precipitation Route to ZnO Nanosphericals. Assam Uni J Sci & Tech. 2011; 7(2):122-127.
-
Jamdagni P, Khatri P, Rana JS. Green synthesis of zinc oxide nanoparticles using flower extract of Nyctanthes arbour-tristis and their antifungal activity. J Kin Sau Uni Sci. 2016; 30(2): 168-175.
-
Chan H, Tsai MH. Synthesis and Characterization of ZnO Nanoparticles Having Prism Shape by A Novel Gas Condensation Process. Rev of Adv Mat Sci. 2008; 18:734-743.
-
Rajendran R, Balakumar C, Ahammed HAM, Jayakumar S, Vaideki K, Rajesh EM. Use of zinc oxide nanoparticles for production of antimicrobial textiles. Int J of Engg Sci & Tech. 2010; 2(1): 202-208.
-
Shaymurat T, J Gu, C Xu, Z Yang, Q Zhao, Y Liu, Y Liu. Phytotoxic and genotoxic effects of ZnO nanoparticles on garlic (Allium sativum L.): A morphological sudy. J Nano tox, 2011; 6(3): 241-248.
-
Keerthana S, Kumar A. Potential risks and benefits of zinc oxide nanoparticles: a systematic review. J cri rev in tox. 2020; 50:47-71.
-
Rastogi I, Arunachalam J. Sunlight-based irradiation strategy for rapid green synthesis of highly stable silver nanoparticles using aqueous garlic (Allium sativum) extract and their antibacterial potential. J Mat Chem & Phy. 2011; 129: 558- 563.
-
Morley KS, Webb PB, Tokareva NV, Krasnov AP, Popov VK, Zhang J, Roberts CJ, Howdle SM. Synthesis and characterization of advanced UHMWPE/ sliver nanocomposites for biomedical applications. Eur Poly J. 2007; 43: 307- 314.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License