IJCRR - 13(9), May, 2021
Pages: 63-70
Date of Publication: 07-May-2021
Print Article
Download XML Download PDF
Role of Body Mass Index, Mean Arterial Pressure, and Uterine Artery Doppler at 11-14 Weeks in Prediction of Pregnancy-Induced Hypertension in Low-Risk Population
Author: Kumari M, Das S, Chanania K, Panda AK
Category: Healthcare
Abstract:Introduction: Hypertensive disorders of pregnancy represent the most common medical complications with increased maternal and perinatal morbidity. Prediction in the first trimester and prevention with aspirin can help to reduce complications.
Objective: To find out the ability of uterine artery pulsatility index along with mean arterial pressure (MAP) and body mass index (BMI) for prediction of pregnancy-induced hypertension.
Methods: This is a prospective observational study conducted in the Institute of Medical Sciences & SUM Hospital. 200 low-risk pregnant women included at 11-14 weeks of gestation to calculate MAP, BMI. Uterine artery doppler of both arteries are performed during the Nuchal Translucency scan. The average uterine artery pulsatility index was calculated.
Result: 7/200 (3.5%) population developed Pregnancy Induced Hypertension( PIH). Mean Arterial Pressure was significantly higher in early-onset Pre-eclampsia (86.65 \? 3.86) in comparison to normal subjects (80.19 \? 7.37) with (p= 0.040).BMI was not significantly higher (22.50 \? 2.32) compared to normal subjects (22.32 \? 2.02) (p= 0.863). Uterine artery pulsatility index was significantly higher in early-onset pre-eclampsia(p< 0.001), late-onset Pre-eclampsia (p= 0.001) and pre-eclampsia(p = < 0.001 inclusive of early and late).Body mass index and mean arterial pressure were not significantly different for the prediction of late-onset pre-eclampsia with a p-value of 0.846 and 0.960 respectively. Keeping 95th centile as cutoff (2.5), the detection rate of pre-eclampsia with uterine artery pulsatility index is 71.4% with p < 0.001.
Conclusion: Combination of uterine artery doppler along with MAP and BMI calculation can effectively screen women at risk of PIH, more so for early-onset PIH before 34 weeks without any extra cost or burden on the health care system.
Keywords: Pre-eclampsia, Prediction, Mean Arterial Pressure, Body Mass Index, Uterine Artery Pulsatility Index
Full Text:
Introduction
Hypertensive disorders represent the most common medical complication of pregnancy affecting between 2-10% of all gestations and account for approximately a quarter of all antenatal admissions.1 Prediction of pregnancy-induced hypertension is difficult by a single test due to its heterogeneous nature.
It can be assessed to some extent by clinical risk factors (primigravida, PIH in a previous pregnancy, chronic hypertension and renal disease, family history of preeclampsia, type 1 and 2 Diabetes Mellitus, Systemic Lupus Erythematosus, advanced maternal age (>40years), BMI> 30, multi-fetal gestation, In Vitro Fertilization pregnancies, biophysical factors ( mean arterial pressure, body mass index, rollover test and uterine artery doppler in 1st and 2nd trimester )and biochemical markers like serum level of Pregnancy Associated Plasma Protein-A, placental growth factor in 1st trimester.2
A prevalent theory for the development of preeclampsia relates to abnormal vasculature maturation of the arteries within the placenta. Incomplete trophoblastic invasion is the main cause leading to pre-eclampsia.3,4 Doppler imaging permits non-invasive evaluation of uteroplacental circulation. During pregnancy, the normal uterine artery Doppler waveform shows low-resistance flow during diastole with the peak end-diastolic flow increasing as gestational age increases. Assessment of the uterine artery waveform can give indirect evidence of abnormal placentation by displaying a persistently high-resistance flow pattern. Among all parameters, the pulsatility index (PI) performs best.3 Singleton pregnancies identified at 11–13 weeks gestation by combined screening with maternal factors and biomarkers as being at high risk for pre-eclampsia (PE), administration of aspirin (150 mg/day from 11–14 to 36 weeks of gestation) can reduce preterm PE, reduces the rate of delivery at < 32 weeks of gestation by about 90% and that of at< 37 weeks by 60%.4 The current study was carried out to find out the ability of uterine artery pulsatility index along with mean arterial pressure and body mass index (BMI) at 11 – 14 weeks of pregnancy for prediction of pregnancy-induced hypertension in a low-risk population of eastern India.5,6
Materials and Methods
This is a prospective observational study conducted in IMS & SUM Hospital under SOA university from August 2018 to August 2020. Pregnant women with singleton pregnancy attending antenatal OPD of our hospital at the gestational age of 11-14 weeks were included in the study. Institutional ethical committee approval was obtained. Informed consent was ensured from all participants.
Nonviable pregnancy detected at that gestation like missed abortion, molar pregnancy, fetal abnormality, hypertensive disorders on treatment with antihypertensives, aspirin and heparin, multi-fetal gestation and women with other endocrine metabolic disorders like pre-gestational diabetes, thyroid disorders were excluded.
200 women satisfying inclusion criteria were included in the study. After implementing the informed consent process, detailed patient history and examination was performed. For all women, clinical history was enquired for risk factors like age, parity, BMI, previous history of preeclampsia, thrombophilia. Routine antenatal investigations like blood group, Complete Blood Count, thyroid profile, FBS, urine routine and microscopy, serology, NT scan performed at 11-14 weeks along with uterine artery doppler for PI value. (>95th percentile i.e 2.5).5 Follow up of antenatal mothers was done till delivery for the development of PIH.
Blood pressure (BP) measurement was done after 5-minute rest in women in sitting position with arm positioned at heart level in both arms simultaneously and 2 sets of BP measurements done at a 1-minute interval. The appearance of the first Korotkoff sound was taken as systolic pressure and the last sound heard as the diastolic pressure and the nearest even digit was taken.6 Calculation of mean arterial pressure (MAP) was done with this formula.
MAP=Systolic pressure + (2*diastolic pressure) /3
An average of four readings was taken.
Body Mass Index (BMI) Measurement
The weight of the woman was recorded during the first antenatal visit with a digital weighing machine after calibration and height were measured in cm. BMI was calculated as weight (kg) divided by the square of height (in meter). BMI = weight (kg) /height (m2) and then subcategorized as BMI (< 18.5 = underweight, 18.5- 24.9 = Normal , 25.0- 29.9 = Overweight , 30.0 and above= obese.) 7,8
Uterine Artery Doppler Measurement
Trans-abdominal uterine artery Doppler was done using a curvilinear probe (3.5 MHz). 9,10 Routine first-trimester scan preset with colour Doppler velocity 30–40 cm/s, pulsed wave Doppler velocity 80–100 cm/s, low filter (50–100 Hz) and pulsed Doppler sample volume 2 mm. The angle of insonation was kept less than 30º. The peak systolic velocity was ensured more than 60 cm/s in the uterine artery. When three similar consecutive waveforms obtained, mean PI was obtained as the average of the left PI and right PI. 11
All women included in the study were followed up with regular antenatal care at 4 weeks interval or a minimum of 4 visits. Any onset of PIH for each woman noted and gestational age of onset of PIH also noted. Early-onset PE is defined as onset before 34 weeks of gestation and late-onset when PE sets at or after 34 weeks of gestation.12 Preterm PIH patients were given proper medical management for control of PIH and fetal maturity. Delivery in such cases was optimised as per institutional protocol.
Statistical analysis
Statistical analysis was done with Microsoft Excel 2007 software and further analyzed in SPSS version 24. All the categorical variables were expressed in terms of number/frequency and percentages. Association between two categorical variables were obtained by using the Chi-square test or Fischer-exact test. All the continuous variables were expressed in terms of mean/median and standard deviation/interquartile range. Significance level in comparison of means was obtained by independent sample t-test or Mann-Whitney U test. A Box-whisker plot was used to describe the continuous variables. Bar and pie charts were used to describe the categorical variables. The receiver operating characteristic (ROC) curve was used to determine the predictability of the variables for the outcomes and the area under the curve (AUC) with a 95% confidence interval was also used to describe the curve. A p-value less than 0.05 was considered statistically significant.
Results
The current prospective observational study was carried out to find out the ability of MAP, BMI, uterine artery pulsatility index values at 11 – 14 weeks of pregnancy for prediction of PIH. 200 low-risk pregnant mothers were included in our study for the analysis.
The mean age of the study participants 25.11 ± 2.78 years with a minimum of 20 years to a maximum of 35 years. The majority were primigravida 159/200 (79.5%) whereas 41/200 (20.5%) were multigravida. Only 20/200 (10%) of the study participants had an abortion history. The mean BMI of the study participants was 25.11 ± 2.78 kg/m2 with a minimum of 20 to a maximum of 29.20 kg/m2. BMI category wise distribution revealed that more than 90% of the subjects had normal weight while only 7.5% were overweight.
Mean MAP among the study participants was 80.32 ±7.36 mm of Hg. The mean uterine artery pulsatility index(PI) value of the study participants was 1.48 ± 0.45 (Table 1). Among the study participants, 7 subjects (3.5%) developed preeclampsia during the follow-up period. The incidence of early-onset pre-eclampsia was 2% while that of late-onset pre-eclampsia was 1.5%.
Early-onset Preeclampsia
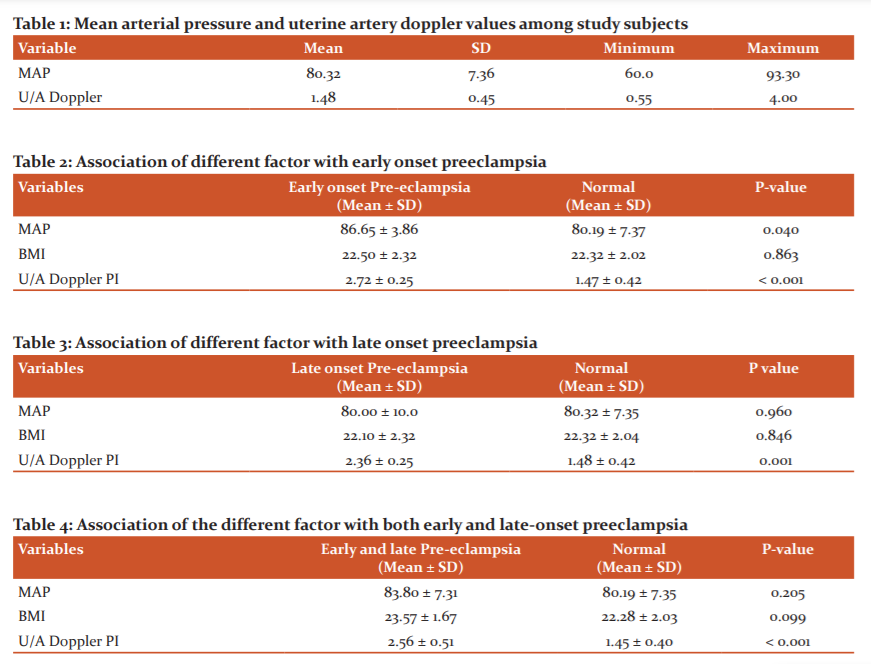
Figure 1 ROC curve showing the ability of uterine artery PI value in predicting early-onset pre-eclampsia. The area under the curve (AUC) was 0.984 with a p-value of 0.001 and a 95% CI of 0.967 – 1.00. Further analysis using Youden’s index was found that at a uterine artery PI value of 2.51, the predictive ability of early-onset pre-eclampsia was 100% sensitive and 97.9% specific.
Figure 2 shows the ROC curve showing the ability of MAP in predicting early-onset pre-eclampsia. The area under the curve (AUC) was 0.793 with a p-value of 0.045 and a 95% CI 0.694 – 0.892. Using, Youden’s index it was found that at a MAP of 83.15, the predictive ability of early-onset pre-eclampsia was 100% sensitive and 70.5% specific.
MAP, Uterine artery Doppler PI were statistically significant in early-onset PE compared to normal subjects with a p-value of 0.040 and 0.001 respectively. BMI was not significantly higher in early-onset PE compared to normal with (p= 0.863) (Table2).
Late-onset preeclampsia
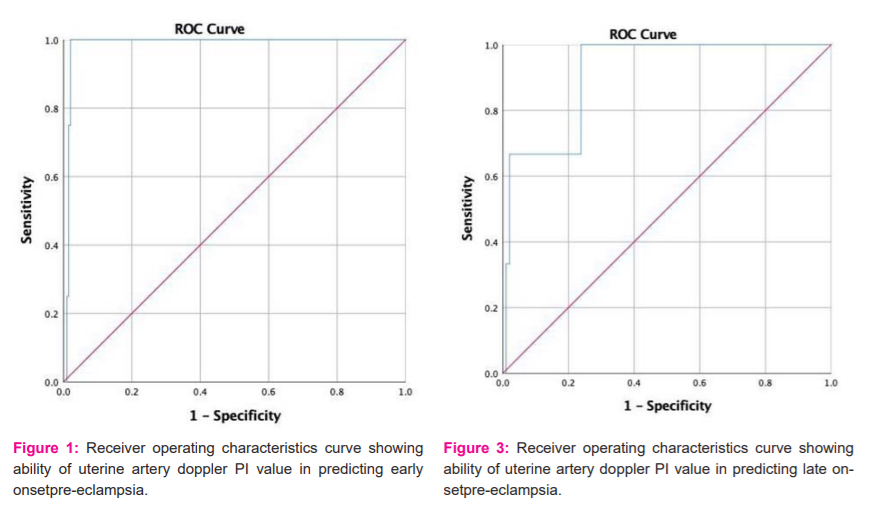
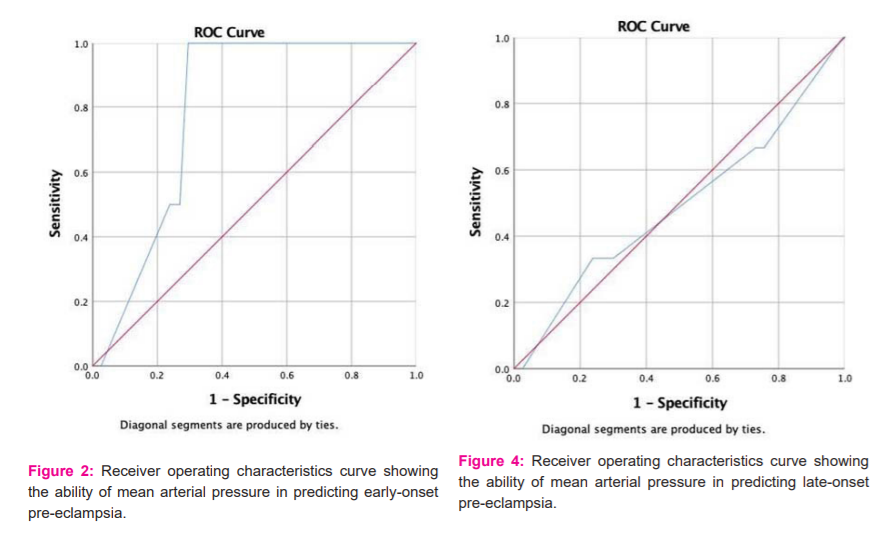
Figure 3 shows the ROC curve showing the ability of uterine artery PI value in predicting late-onset pre-eclampsia. The AUC was 0.910 with a p-value of 0.015 and a 95% CI 0.788 – 1.00. By Youden’s index it was found that at uterine artery PI value of 2.51, the predictive ability of late-onset pre-eclampsia was 100% sensitive and 76.2% specific. Figure 4 shows the ROC curve showing the ability of MAP in predicting late-onset PE The AUC was 0.492 with a p-value of 0.963 and a 95% CI 0.131 – 0.853. MAP, BMI were not significantly higher in late-onset PE compared to the normal population with a p value of 0.960 and 0.846 respectively. But uterine artery PI was significantly higher (p= 0.001)in late-onset PE compared to the normal population (Table 3).
Prediction of PE (including early and late-onset)
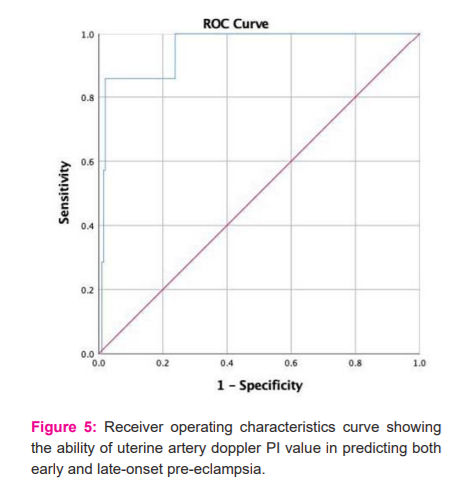
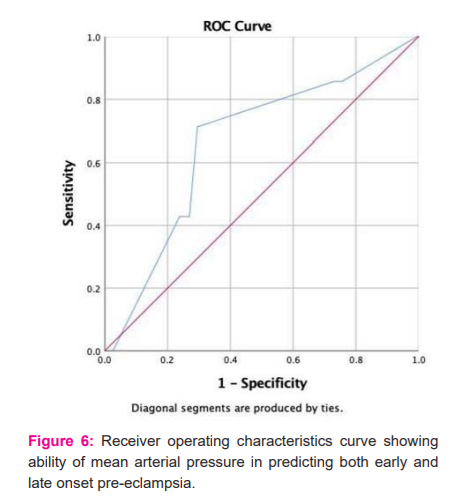
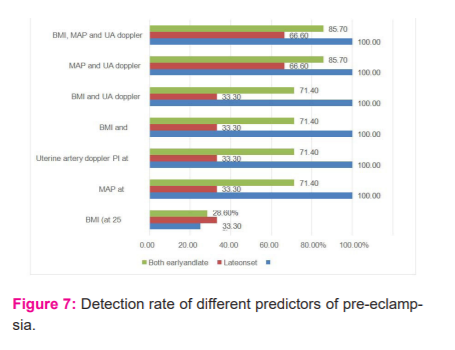
The ROC curve showing the ability of uterine artery doppler PI value in predicting both early and late-onset pre-eclampsia (Figure 5). The AUC was 0.953 with a p-value of < 0.001 and a 95% CI 0.892 – 1.00. Further analysis using Youden’s index was found that at a uterine artery doppler PI value of 2.51, the predictive ability of early-onset pre-eclampsia was 100% sensitive and 76.2% specific. ROC curve showing the ability of mean arterial pressure in predicting late-onset pre-eclampsia. The AUC was 0.492 with a p-value of 0.963 and a 95% CI 0.131 – 0.853. (Figure 6). It shows the ROC curve showing the ability of mean arterial pressure in predicting both early and late-onset pre-eclampsia. The AUC was 0.664 with a p-value of 0.141 and a 95% CI ( 0.464 – 0.864). MAP and BMI are not significantly higher for the prediction of pre-eclampsia with a p-value of 0.205 and 0.099 respectively when considered individually. Uterine artery PI is significantly higher in PE (both early and late) with a p value <0.001 which is statistically significant (Table 4). At the cutoff level of 25 kg/m2, the detection rate of BMI of pre-eclampsia was 28.6%. Similarly, for MAP and uterine artery doppler PI it was 71.4% and 100% respectively. BMI and MAP combinedly detected 71.4% of pre-eclampsia cases while in any combination with uterine artery doppler PI the detection rate rises to 100%. The details were given in Table 4 and Figure 5.
Discussion
Pregnancy-induced hypertension is a burden on the health care system. It is associated with increased risk of placental abruption, acute renal failure, cerebrovascular and cardiovascular complications, disseminated intravascular coagulation, maternal death, fetal growth restriction, stillbirth.12 Therefore, the ability to predict PIH is vital.
Many screening tests have been investigated to predict preeclampsia, but only a few of them reached satisfactory sensitivities and specificities due to the heterogeneous nature of the disease. Preeclampsia prediction could not be achieved by a single test. The current prospective observational study was carried out to find out the ability of uterine artery doppler values along with mean arterial pressure and body mass index (BMI) at 11–14 weeks of pregnancy for prediction of pregnancy-induced hypertension. 200 pregnant women were included in our study and followed up till delivery.
In our study, which includes low-risk women, the incidence of pre-eclampsia is 3.5%. It includes both early-onset and late-onset pre-eclampsia. This is similar to studies like Osungbadeetal. and WHO in 2005 that projected incidence of preeclampsia ranges between 2% and 10% of pregnancies worldwide and 3%to 10% according to Duley et al. in 2009. around 2% globally. 1,13,14
The incidence of early-onset pre-eclampsia in our study is 2% and the incidence of late-onset pre-eclampsia is 1.5%. The incidence of late-onset preeclampsia in our study is similar to Poon et al. where it was 1.5%.15 BMI can be considered as a predictor of pre-eclampsia. According to Syngelaki et al. study which included a large sample size of 41,577 women the median BMI was 24.4 and which demonstrated increasing maternal BMI increase chances of stillbirth, pre-eclampsia, gestational hypertension, Gestational Diabetes Mellitus and subsequent miscarriages. 16
According to Anderson et al. study, which included 22,500 women in retrospective cohort analysis concluded overweight and obesity increases the risk of pre eclampsia.17
According to Mrema et al. study, which included 17,738 women concluded overweight and obesity have a 1.4 and 1.8 times increased risk of pre-eclampsia than normal BMI.18 In our study, the median BMI is 22.5 and the mean BMI of PIH patients is higher than the normal population i.e (23.57±1.67) and (22.28 ±2.03) respectively. The majority of women who developed preeclampsia had higher BMI than the normal population. However, it was not statistically significant.
Blood pressure measurement, part of our routine antenatal checkup and MAP calculation being an inexpensive tool can be used for the prediction of pre-eclampsia. The detection rate (DR) of pre-eclampsia in our study by mean arterial pressure at the cut-off of 83.15 mmHg is 71.4% which is in line with studies of Caritas et al.19 study with a detection rate of 66% and Friedmann et al. with a DR of 64% and Quass et al with a DR of 65% and Kucs et al and Cnossen JS et al.20-22 The DR of MAP is better for early-onset preeclampsia compared to late-onset with DR of 100% which is similar to studies of Oney et al. and Rogers et al. with DR of 93%.24,25The DR of late-onset preeclampsia in our study is 33.3% which is similar to SylwiaKuc study with DR of 31% and O’Gorman N, Wright with DR of 43% respectively.26,27
In our study, uterine artery doppler was significantly higher in preeclampsia with a p value of < 0.001 which is statistically significant as the similar result was obtained in the study by Gomez et al., which included 999 women, where the mean PI value of the pre-eclampsia patients was significantly higher (2.46 ± 0.28) than normal women (1.435 ± 0.45).28 In our study, it was found that at a uterine artery pulsatility index value of 2.51, the predictive ability of early-onset pre-eclampsia is 100% and sensitivity 97.9%. According to our study,
The uterine artery is a better predictor for early-onset PE concerning late-onset PE with DR of 100% and 33.3% respectively. This is quite similar to one of the largest study to the date of Plasencia et al. which included 6015 women. Screening by uterine artery doppler helps identify women who develop severe early-onset PE than the late-onset disease.29 It could predict pre-eclampsia requiring delivery before 34 weeks was 82% compared to 31% for late pre-eclampsia. In another study by Pilalis et al. including 878 women concluded that uterine artery doppler PI value of 95th centile is useful for early prediction giving a mean PI value higher in preeclamptic women compared to normal.30 Khong et al. in 2015 concluded that as an isolated marker Uterine Artery PI is sensitive in predicting pre eclampsia.31Another meta-analysis by Velauthar et al. including 55,974 women concluded first-trimester uterine artery a useful predictor of pre eclampsia.32
The AUROC of our study for uterine artery PI value was 0.953 with a p-value of < 0.001 which is statistically significant. According to Erdogdu et al. study including 412 women with AUROC was 0.79 for prediction of pre-eclampsia and 0.83 for early PE.33 If we take the 95th percentile of our study (2.48) as cut off, then sensitivity for early-onset was 100% and specificity was 96%. For late-onset, sensitivity was 33.3% and specificity was 95%. The uterine artery pulsatility index 95th centile of our study is 2.48 which is closer to the 95th centile of study by Gomez et al. (PI value 2.5). Hence, for our population, we can take uterine artery PI value of 2.5 as cut off for prediction of PIH.34
In our study, on combining BMI with Mean arterial pressure (MAP) the detection rate of pre-eclampsia is 71.4%. On combining BMI, MAP and uterine artery doppler PI, the detection rate improves to 85.7%. This goes in line with the study of LC Poon et al. which included 8016 women, The detection rate of early PE at a 10% false?positive rate increased from 47% in screening by maternal factors alone to 81% in screening by maternal factors and the Uterine Artery PI value.35 Another study by Wright et al. including 58,884 women which states maternal characteristics, uterine artery PI and MAP can detect 90 % of pre-eclampsia cases requiring delivery before 34 weeks.36 We have taken the period of gestation as 11-13+6 weeks as this is the antenatal visit where history taking and all routine investigations along with sonography (NT scan) is done. Uterine artery doppler can be performed in another few minutes without much effort. Blood pressure and measurement of BMI is part of routine checkup.
Conclusion
Pathophysiological changes in pre-eclampsia are believed to occur in early pregnancy and antedate clinical manifestation of the disease. A combination of biophysical, biochemical markers along with uterine artery PI is the best option for screening pre-eclampsia. In many areas of India laboratories with biochemical marker, the facility is not available or not feasible. Also, laboratories should be standardised throughout for acceptable results. According to the present study results, BMI, MAP, uterine artery pulsatility index when used individually as predictors are less sensitive compared to when they are used in combination. Obstetrician/sonologist should be trained for taking proper uterine artery Doppler measurement. It should be done according to the Fetal Medicine Foundation criteria.
Conflict of Interest: The authors declare there is no conflict of interest among them.
Source of Funding: Nil
Author’s Contribution: Das S - Concept, study design. data analysis, statistical analysis and manuscript preparation.
Kumari M- Data collection and manuscript drafting,
Chanania K1 and Panda AK -Collection of data, data analysis, statistical analysis.
References:
1. World Health Organization. Report of a WHO technical consultation on birth spacing: Geneva, Switzerland 13-15 June 2005.
2. ACOG Committee on Obstetric Practice. ACOG practise bulletin. Diagnosis and management of preeclampsia and eclampsia. Am Colle Obstet Gynecol Int J Gynecol Obstet 2002;77(1):67-75.
3. Salem MA, Ammar IM. First-trimester uterine artery pulsatility index and maternal serum PAPP-A and PlGF in the prediction of preeclampsia in primigravida. J Obstet Gynaecol Ind 2018;68(3):192-6.
4. Poon LC, Wright D, Rolnik DL, Syngelaki A, Delgado JL, Tsokaki T, et al. Aspirin for Evidence-Based Preeclampsia Prevention trial: effect of aspirin in the prevention of preterm preeclampsia in subgroups of women according to their characteristics and medical and obstetrical history. Am J Obstet Gynecol 2017; 217(5):585-591.
5. Gómez O, Figueras F, Fernández S, Bennasar M, Martínez JM, Puerto B, et al. Reference ranges for uterine artery mean pulsatility index at 11–41 weeks of gestation. Ultrasound Obstet Gynecol 2008;32(2):128-132.
6. Frese EM, Fick A, Sadowsky HS. Blood pressure measurement guidelines for physical therapists. Cardiopulm Phys Ther J 2011;22(2):5.
7. Abrams B, Parker J. Overweight and pregnancy complications. Int J Obes 1988;12(4):293-303.
8. Sood N, Simon P, Ebner P, Eichner D, Reynolds J, Bendavid E, et al. Seroprevalence of SARS-CoV-2–specific antibodies among adults in Los Angeles. J Ame Med Ass 2020; 323(23):2425-2427.
9. Lyall F. The human placental bed revisited. Placenta 2002; 23(8-9):555-562.
10. Kim YM, Bujold E, Chaiworapongsa T, Gomez R, Yoon BH, Thaler HT, et al. ailure of physiologic transformation of the spiral arteries in patients with preterm labour and intact membranes. Am J Obstet Gynecol 2003;189(4):1063-9.
11. Khalil A, Nicolaides KH. How to record uterine artery Doppler in the first trimester. Ultras Obstet Gynecol 2013;42(4):478-9.
12. MacKay AP, Berg CJ, Atrash HK. Pregnancy-related mortality from preeclampsia and eclampsia. Obst Gynecol 2001;97(4):533-8.
13. Osungbade KO, Ige OK. Public health perspectives of preeclampsia in developing countries: implication for health system strengthening. J Pregnancy 2011;5(3):234-238.
14. Duley L. The global impact of pre-eclampsia and eclampsia. Seminars in perinatology J Pregnancy 2009;33(3):130-137.
15. Poon LC, Staboulidou I, Maiz N, Plasencia W, Nicolaides KH. Hypertensive disorders in pregnancy: screening by uterine artery Doppler at 11–13 weeks. Ultrasound Obstet Gynecol: Off J Int Soc Ultras Obstet Gynec 2009;34(2):142-148.
16. Syngelaki A, Bredaki FE, Vaikousi E, Maiz N, Nicolaides KH. Body mass index at 11–13 weeks’ gestation and pregnancy complications. Fetal diagn Ther 2011;30(4):250-265.
17. Anderson NH, Sadler LC, Stewart AW, Fyfe EM, McCowan LM. Ethnicity, body mass index and risk of pre?eclampsia in a multiethnic New Zealand population. Obstet Gynaecol 2012;52(6):552-558.
18 . Mrema D, Lie RT, Østbye T, Mahande MJ, Daltveit AK. The association between pre-pregnancy body mass index and risk of preeclampsia: a registry-based study from Tanzania. BMC Pregn Childbirth 2018;18(1):1-8.
19. Caritis S, Sibai B, Hauth J, Lindheimer M, VanDorsten P, Klebanoff M, et al. Predictors of pre-eclampsia in women at high risk. Am J Obstet Gynecol 1998;179(4):946-951.
20. Gallo D, Poon LC, Fernandez M, Wright D, Nicolaides KH. Prediction of preeclampsia by mean arterial pressure at 11-13 and 20-24 weeks' gestation. Fetal Diagn Therap 2014;36(1):28-37.
21. Quaas L, Wilhelm C, Klosa W, Hillemanns HG, Thaiss F. Urinary protein patterns and EPH-gestosis. Clin Nephrol 1987;27(3):107-110.
22. Kuc S, Koster MP, Franx A, Schielen PC, Visser GH. Maternal characteristics mean arterial pressure and serum markers in early prediction of preeclampsia. PloS One 2013; 8(5):e63546.
23. Cnossen JS, Vollebregt KC, De Vrieze N, TerRiet G, Mol BW, Franx A, Khan KS, Van Der Post JA. Accuracy of mean arterial pressure and blood pressure measurements in predicting pre-eclampsia: systematic review and meta-analysis. Br Med J 2008 15;336(7653):1117-1120.
24. Öney T, Kaulhausen H. The value of the mean arterial blood pressure in the second trimester (MAP-2 value) as a predictor of pregnancy-induced hypertension and preeclampsia. A preliminary report. J Hypert Pregn 1983;2(2):211-216.
25. Rogers MS, Chung T, Baldwin S. A reappraisal of the second trimester mean arterial pressure as a predictor of pregnancy-induced hypertension. J Obstet Gynaecol 1994;14(4):233-236.
26. Kuc S, Koster MP, Franx A, Schielen PC, Visser GH. Maternal characteristics mean arterial pressure and serum markers in early prediction of preeclampsia. PloS One 2013; 8(5):e63546.
27. O’Gorman N, Wright D, Syngelaki A, Akolekar R, Wright A, Poon LC, et al. Competing risks model in screening for preeclampsia by maternal factors and biomarkers at 11-13 weeks gestation. Am J Obstet Gynecol 2016; 214(1):103-e1.
28. Gomez O, Martinez JM, Figueras F, Del Rio M, Borobio V, Puerto B, Coll O, Cararach V, Vanrell JA. Uterine artery Doppler at 11–14 weeks of gestation to screen for hypertensive disorders and associated complications in an unselected population. Ultrasound Obstet Gynecol 2005;26(5):490-494.
30. Pilalis A, Souka AP, Antsaklis P, Daskalakis G, Papantoniou N, Mesogitis S, et al. creening for pre?eclampsia and fetal growth restriction by uterine artery Doppler and PAPP?A at 11–14 weeks' gestation. Ultras Obstet Gynecol2007 Feb;29(2):135-140.
31. Khong SL, Kane SC, Brennecke SP, da Silva Costa F. First-trimester uterine artery Doppler analysis in the prediction of later pregnancy complications. Dis Mark 2015; 2015.
32. Velauthar L, Plana MN, Kalidindi M, Zamora J, Thilaganathan B, Illanes SE, et al. First?trimester uterine artery Doppler and adverse pregnancy outcome: a meta?analysis involving 55 974 women. Ultras Obst Gynecol 2014;43(5):500-507.
33. Erdo?du E, Ar?soy R, Kumru P, Ard?ç C, Pekin O, Tu?rul S. The role of first trimester uterine artery Doppler in the prediction of preeclampsia. Perinat J Perinatoloji Derg. 2014;22.
34. Gómez O, Figueras F, Fernández S, Bennasar M, Martínez JM, Puerto B, et al. Reference ranges for uterine artery mean pulsatility index at 11–41 weeks of gestation. Ultras Obstet Gynecol 2008;32(2):128-32.
35. Poon LC, Nicolaides KH. First?trimester maternal factors and biomarker screening for preeclampsia. Prenat Diagn 2014;34(7):618-27.
36. Akolekar R, Syngelaki A, Poon L, Wright D, Nicolaides KH. Competing risks model in early screening for preeclampsia by biophysical and biochemical markers. Fetal Diagn Ther 2013;33(1):8-15.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License