IJCRR - 13(20), October, 2021
Pages: 40-45
Date of Publication: 24-Oct-2021
Print Article
Download XML Download PDF
Characterization and Green Biosynthesis of Silver Nanoparticles using Raphanus sativus and its Antibacterial Activity against Urinary Tract Infection Causing E. coli
Author: K. Jagathy, S. Malathi
Category: Healthcare
Abstract:Introduction: Biofilm production is an important phenomenon by bacteria such as E.coli that contribute to multiple drug resistance. The green synthesis of nanoparticles (AgNPs) with the use of plants and vegetables has more advantages because they are safe to handle and easy available. Aim: In the present study silver nanoparticles were synthesized by an eco-friendly and cost-effective method using Raphanus sativus [leaf]. This acts as reducing and stabilizing agents. Methods: The formation of Ag NPs has been characterized and confirmed through Scanning Electron Microscope [SEM], X�Ray Diffraction [XRD], Fourier Transform Infra-Red [FTIR] and Ultraviolet [UV] visible absorption spectrum. The key step in this process is the identification of virulence determinants that may serve as targets for vaccine and drug development. Results: The phytochemical investigation uncovered the presence of rich measures of Poly phenols and other biochemicals in these concentrates, which fill in as decreasing and capping agents for converting silver nitrate into AgNPs. FTIR confirmed the role of biomolecules in the bioreduction and efficient stabilization of AgNPs. UV\?Visspectra showed a band of around 279 nm, which implies the characteristics of AgNPs. XRD patterns have shown the crystalline nature of the synthesized AgNPs with fcc structure. SEM analysis revealed the spherical shape of the synthesized AgNPs with an average particle size of 8 nm. Effectiveness of the green synthesized nanoparticles by Aqueous extract against UTI e.coli strain was measured in terms of zone of inhibition in millimetres. Conclusion: These green synthesized silver nanoparticles had significant antibacterial activity. The present study is supportive evidence that Leaf of R. sativus, generally wasted, have important medicinal constituents.
Keywords: Raphanus sativus, Silver nanoparticles, leaf extract, Scanning electron microscope, polyphenols, antioxidant, Phyto�chemicals, Characterization
Full Text:
Introduction:
Nanosized silver nanoparticles have discovered colossal applications in the field of high affectability biomolecular identification and diagnostics, antimicrobials, cancer prevention agents and therapeutics, catalysis1 and miniature hardware. Nanoparticles can be blended by different methodologies like compound and photochemical responses in invert micelles, microwave helped, warm deterioration, electrochemical, sonochemical measures.
Plant interceded natural combination of nanoparticles is acquiring significance because of its straightforwardness, financially savvy and eco-friendliness. AgNPs could be efficient nanoparticles as they possess good catalytic and high performance permanent magnetic properties and also possess biomedical and cytotoxic activity.
Raphanus sativus (Radish) is a common vegetable crop in Asia. It has a lot of nutrients B and C just as gelatin, phytin, manganese, iron and copper. Leaves are utilized to treat looseness of the bowels, asthma, hack, the runs, urinary plot disease and hunger. 2 It contains ferulic corrosive, gentisic corrosive, Raphanus, erucic corrosive, sinapate, raphanin and sulforaphane. The seeds are carminative, diuretic and purgative. Roots have been utilized for treating syphilis, haemorrhoids, gonorrhoea,3malignant growth and urinary complaints.
In this investigation, plant-mediated synthesis of AgNPs was carried out using aqueous leaf extract of Raphanus sativus and characterized using UV-visible spectroscopy, Fourier transforms infrared spectroscopy and scanning electron microscopy. The antibacterial activities of the green synthesized AgNPs have been investigated against Gram-negative bacteria. In the present study, results showed that the reported AgNPs are having bactericidal and cytotoxic activities.
Materials and Methods:
To obtain silver nanoparticles the deionized water was used as a reaction medium, the reducing agent used was vegetable extract, i.e radish extract and the reagent used for the synthesis was silver nitrate which does not evolve any toxic hazard on the atmosphere.
Preparation of Fresh Radish Extract
Radishes were procured from the local market (Nov 2019) [figure: 1]. Aqueous extract of Raphanus sativus was prepared using 25 gm. It was washed completely in deionised water, dried, cut into little pieces and were bubbled in 150 ml of deionized water for 5-10 minutes. The concentrate was separated through Whatman No.1 channel paper and utilized for additional exploration.

Quantitative Phytochemical analysis:
For qualitative analysis of active phytochemicals in R. Sativus leaf extract. Preliminary Phytochemical analysis was carried out on Aqueous extract using standard protocol for determination of phytoconstituents including tannins, saponins, phlorotannins, anthraquinones, carbohydrates, reducing sugars, steroids, phytosterol, flavonoids, alkaloids, amino acids, terpenoids, chalcones and cardiac glycosides as described by Karunakar Rao et al. 4
Synthesis of silver nanoparticle
The leaf extract was prepared by boiling method. (fig:2 leaf extract)1mM silver nitrate was prepared (fig 4 A). The silver nanoparticle was synthesized. Green synthesised silver nanoparticles were reddish-brown. The colour of the extract changed from light yellow to reddish-brown after the addition of silver nitrate and on incubation for 90 minutes. The colouration was due to the excitation of the surface Plasmon vibration of the AgNo3. Change in colour after the reduction of silver ions to silver nanoparticles is shown in figure 4 B. The reduction rate and formation of nanoparticles can be increased further by an increase in incubation time. [Figure: 2]

Characterization of silver nanoparticles
The concentrate was centrifuged twice to confine the AgNPs and to kill the undesirable excess. It was dried in a hot air stove for 30 minutes at a temperature of 1000C. Integrated silver nanoparticles were affirmed by UV–Vis spectroscopy and it was done utilizing UV-Vis spectrophotometer in the 200–1100 nm range. Point by point examination of the morphology, size and appropriation of the nanoparticles was recorded by Scanning Electron Microscopy (SEM) machine and the presence of reflection by XRD designs. The conceivable useful gatherings in the combination and adjustment of nanoparticles were distinguished by performing FTIR investigation.
Urine Sample Processing:
MICROSCOPY:
Smears were prepared by placing a loopful of the sample on a clear glass slide and gram staining was done for microscopic examination.
CULTURE:
All the samples were inoculated onto Blood agar and MacConkey agar and the plates were incubated at 37ºC. Identification was done with IMVIC tests.
VIRULENCE FACTOR TESTING – BIOFILM [TISSUE CULTURE PLATE ASSAY]:
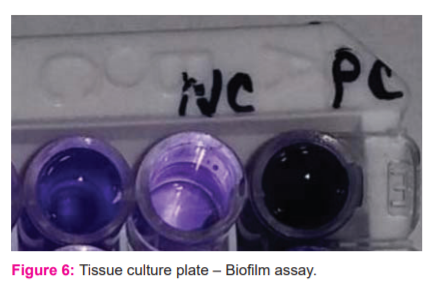
Isolates from fresh agar plates were inoculated in Trypticase Soy Broth and incubated for 24 hours at 37°C, then diluted with fresh Trypticase Soy Broth in 1 in 100 dilutions. Individual wells of sterile, polystyrene, 96 well?flat bottom tissue culture plate (TCP) wells filled with 0.2 ml aliquots of the diluted cultures and only broth served as a control to check sterility and nonspecific binding of media. The TCP was incubated for 18–24 h at 37°C. After incubation content of each well was gently removed by tapping the plates. Then wells were washed four times with 0.2 ml of PBS (pH 7.2) to remove free-floating “planktonic” bacteria. Wells were stained with crystal violet (0.1%). Excess stain was rinsed off by washing with deionized water, and the plate was kept for drying. 5 If the biofilm is formed by organisms, then wells are uniformly stained with crystal violet. The optical density (OD) of stained adherent bacteria was determined with a micro ELISA auto reader at a wavelength of 570 nm (OD 570 nm). An experiment was repeated thrice, and the data then were averaged, and standard deviation was calculated. The mean OD value obtained from media control was deducted from all the test OD values.
Results and Discussion:
Qualitative phytochemical analysis:
Phytochemical analysis revealed the presence of tannins, saponins, flavonoids, phlorotannins, anthraquinones, carbohydrates, reducing sugars, steroids, phytosterol, alkaloids, amino acids, terpenoids, cardiac glycosides and chalcones in R. Sativus niger extracts (Table 1).

In the study Rama koyyati et al., 6 states that polyphenols and alkaloids present as important phytochemicals in radish peel extract
Characterization of Green synthesized silver nanoparticles
UV-visible spectroscopy
The synthesis of silver nanoparticles had been confirmed by UV-visible spectroscopy. The UV-visible spectrum showed a distinct absorption beak at (279nm). This was due to the excitation of surface Plasmon resonance (SPR) by AgNPs. AgNPs have free electrons, which give rise to the SPR absorption band, due to the combined vibration of electrons of metal nanoparticles in resonance with the light wave. Thus the reduction of AgNPs in the aqueous solution of the silver complex during the reaction with the leaf extract of Raphanus sativus was confirmed by the UV- visible spectra (figure 3). Satyavati R.7in her study reveals that green synthesized silver nanoparticles by Coriandrum leaves had been absorbed in a peak value of 250nm by UV-visible spectroscopy
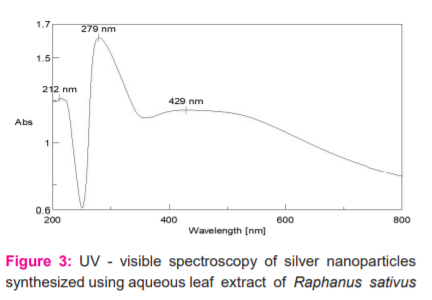
Detailed analysis of the morphology, size and distribution of the nanoparticles was seen by Scanning Electron Microscopy (SEM). The possible functional groups in the synthesis and stabilization of nanoparticles were identified by performing FTIR analysis.
FTIR ANALYSIS
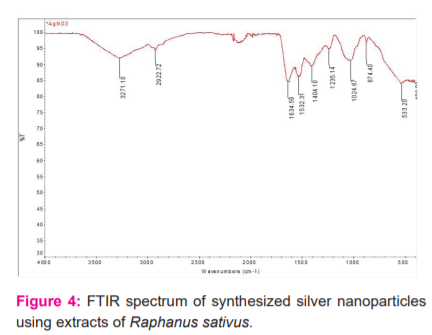
FTIR measurement was carried out to identify the possible biomolecules responsible for capping and efficient stabilization of Ag nanoparticles by plant leaf extract.
To determine the presence of the functional group in biomolecule by interaction with metal ion by FTIR spectrum analysis. The peaks observed from extraction of Raphanus sativus are 3271, 2922, 1634, 1532, 1404, 1235, 1024, 874, 533, 400 cm-1. From the results the 3271 cm-1 corresponds to OH stretching vibration of alcohol, 2922 cm-1 indicated the presence of CH stretching vibration of an aromatic compound, 1634 cm-1 is associated with C=O stretching of carboxylic acid, biomolecule such as carbohydrate and protein,1404 cm-1 indicates the presence of CC bond in the aromatic ring, 1532 NH of the amide linkage, 1235 is attributing to CN stretching of amines, 1024 corresponds to COC, 874 assigned to CH, 533 corresponds to C-C-CN and C-C=O from the results which shows the presence of function group by reduction of silver nanoparticle. [figure: 4] A study by Garima Singh8 et al., mentioned that aromatic compounds present in the herbal plants shown specific peaks from 1630 – 1854 cm-1.
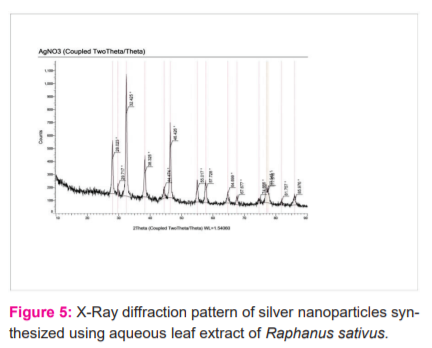
XRD analysis:
The XRDpatterns of dried AgNo3 synthesized silver nanoparticles using extracts of Raphanus sativus. A number of bragg reflection [fig: 5] shows a characteristic peak (at 2θ = 37.27°, 46.31°, 55.44° and 76.23°).
Virulence factors like biofilm activity by TCP method detected as biofilm producers. [figure: 6] A pathogenic study by Jayesh P10 proves that the antibacterial efficacy of metal nanoparticles quite effective due to strain specificity.
EVALUATION OF ANTIBACTERIAL ACTIVITY
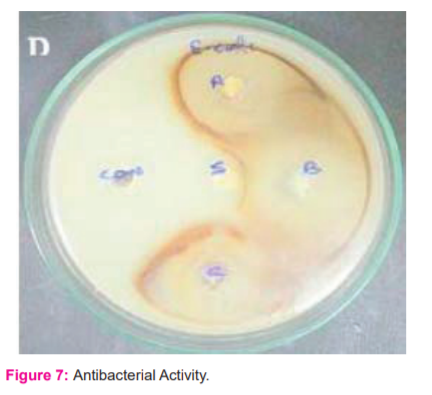
The silver nanoparticles synthesized from Raphanus sativus exhibited the antibacterial to bacterial strain E.coli [UTI causing bacteria]. The results are presented in (Table: 2) as the average values of the zone of inhibition radii and MIC. [figure: 7]
The antibacterial activity and acting mechanism of silver nanoparticles (SNPs) on Escherichia coli ATCC 8739 were investigated in this study by analyzing the growth, permeability, and morphology of the bacterial cells following treatment with SNPs. The experimental results of Wen Ru Li et al.,9 indicated 10 milli g/ml SNPs could completely inhibit the growth of 10 (7) cfu/ml E. coli cells in liquid Mueller-Hinton medium.

SEM analysis:
Morphological character and size details of the green synthesized silver nanoparticles using the leaf extract Raphanus sativus was presented by SEM images. The size of the nanoparticles was investigational from the SEM image between the ranges of 40-90 nm (Fig 8). This size difference in the nanoparticles is due to the presence of proteins or other biomolecules from extract Raphanus sativus, which was bound in the surface of the nanoparticles and also the result showed that the green synthesized silver nanoparticles were spherical. The figure indicates that the synthesized silver nanoparticles are well separated showing no agglomeration.
Study of antimicrobial activity of nanosilver (ns) in tissue culture media, presented the SEM images too explicit the structure and mechanism of nanoparticle efficacy on bacteria. 11

Conclusion:
In this investigation, silver nanoparticles which were synthesized from Raphanus sativus leaf extricate indicated antibacterial action. Subsequently, it is demonstrated from this investigation that the silver nanoparticles blended from Raphanus sativus leaf separate appear to be encouraging and successful antibacterial operators against bacterial strains and strong cell reinforcement. This organic science approach towards the blend of silver nanoparticles is exceptionally fundamental exertion being tended to in nanomedicine on account of its differed favorable circumstances. Plant remove being very eco well disposed and practical can be utilized for the enormous scope combination of silver nanoparticles in nanotechnology preparing ventures.
Acknowledgement:
The authors would like to thank the authorities of the Microbiology department, Indo American college, Cheyyar, Tamilnadu, India for providing the laboratory facilities and support for this work.
AUTHORS CONTRIBUTION: Jagathy made substantial contributions to conception, acquisition of data, took part in drafting the article, or revising it critically for important intellectual content, Malathy made physiochemical analysis and final approval of the version to be published, and agreed to be accountable for all aspects of the work.
FUNDING: No funding sources
CONFLICT OF INTEREST: None declared
ETHICAL APPROVAL: The study was approved by the Institutional Ethics Committee and its number IRB/SAC/231
References:
-
Yogeswari R, Sikha B, Akshay Kumar O, Nayak P. Green synthesis of silver nanoparticles using Ocimum sanctum (Tulasi) and study of their antibacterial and antifungal activities.J. Microbiol. Antimicrob. 2012; 4(6): 103-109, November 2012-
-
Farooqui MA, Chauhan PS, Krishnamoorthy P, Shai J. Extraction of silver nanoparticles from the leaf extracts of Clerodendrum inerme. DIG J NANOMATER BIOS. 2010; 5: 43-49.
-
Jayesh P, Arup Kumar C, Siddhartha D, Suparna M. Strain specificity in antimicrobial activity of silver and copper nanoparticles. Acta Biomaterialia, 2008;4 (3):707–716.
-
Karunakar Rao K, Manisha RD, Ramchander M, Prashanthi Y. Microwave-assisted green synthesis of silver nanoparticles using Stigmaphyllon little leaves, their characterization and anti-microbial activity. Dig. J. Nanomater. Biostructures Dig J Nanomater bios. 2013; 3(1): 13-16.
-
Kim JS, Kuk E, Yu KN, Kim J-H, Sung-Jin Par K. Antimicrobial effects of silver nanoparticles. Nanomedicine: Nanotechnology, Biology and Medicine. 2007; 3(1): 95– 101
-
Koyyati R, Nagati V, Merug R, Manthurpadigya P. Biological synthesis of silver nanoparticles using Raphanus sativus var. longipinnatus leaf extract and evaluation of their antioxidant and antibacterial activity. Int J Med Pharm. 2013; 3 (4): 89-100.
-
Satyavati R, Balamurali Krishna M, Venugopal Rao S, Saritha R, Narayana Rao. Biosynthesis of Silver Nanoparticles Using Coriandrum Sativum Leaf Extract and Their Application in Nonlinear Optics. Advanced Science Letters.Vol. 3, 138–143, 2010. 27.
-
Singh G, Bhavesh R , Kasariya K , Sharma AR, Rajendra Pal SingH. Biosynthesis of silver nanoparticles using Ocimum sanctum (Tulsi) leaf extract and screening its antimicrobial activity. J. Nanoparticle Res.. 2011; 28(13):2981–2988.
-
Li WR, Xie XB, Shi QS, Zeng HY Ou-Yang YS, Chen YB. Antibacterial activity and mechanism of silver nanoparticles on Escherichia coli. Appl Microbiol. Biotechnol 2010; 85: 1115-1122
-
Jayesh R, Arup Kumar C, Siddhartha D, Suparna M. Strain specificity in antimicrobial activity of silver and copper nanoparticles. Acta Biomaterialia, 2008; 4(3): 707–716.
-
Salisu IB, Abubakar AS, Sharma M, Ramesh N. Pudake. Study of antimicrobial activity of nanosilver (ns) in tissue culture media.Int. J Curr Res. 2014; 6 (13): 01-05
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License