IJCRR - 13(18), September, 2021
Pages: 35-39
Date of Publication: 26-Sep-2021
Print Article
Download XML Download PDF
Anti-diabetic, Anti-oxidant, Fluorescence and Filter Characterizations of Bis Glycine Lithium Bromide Monohydrate (BGLBMH) Macro and Nano-scaled Crystals
Author: Alphonse Arockia Jenecius, Ramachandra Prasad Divya, Kannuswamy Senthil Kannan, Irudaya Antonat Sophia, Balaraman Ravindran, Ramamurthi Krishnaveni
Category: Healthcare
Abstract:Introduction: Crystals are meant for versatile applications and utility in all fields and mainly in electronic and biofields. Aim: Our aim, the present investigation is focused on Bis Glycine Lithium Bromide Monohydrate (BGLBMH crystals), its synthe�sis and the various characterizations. Methodology: BGLBMH Macro crystals are put in order by slow evaporation solution growth method and nanocrystal by mill�ing method. The Single-crystal XRD, Powder XRD analysis, filter, anti-diabetic and anti-oxidant (AD and AO) studies were performed here. Result: The single-crystal XRD study reveals the macro-crystalline lattice parameters with a, b, c in Å as 7.5397, 17.4174, 8.2727 and \? as 118.14o as the system is monoclinic with a space group of P21/c. The macro and nano scales are analyzed for fluorescence spectral activity. The crystals speciality is THG and shows the SHG NLO value of 1.25 times that of KDP because of the strongest H bonds and the bandgap is 3.08 eV which is 403 nm as emission FL value for macro scaling and 397 nm for nano scaling with a bandgap of 3.12 eV. The nano outline of BGLBMH crystals is 250 nm and 34 nm correspondingly for the initial and final one. Conclusion: The BGLBMH have good scope for anti-diabetic by the Glycine, bromide presence and have increased in inhibi�tion as concentration increases and the IC value as 37.5 for macro and in a nano form, it is 30.4. Also, the AD - nm variations will have good efficiency when the size of the sample decreases from 250 to 34 nm. The BGLBMH macro and nanocrystals are used in filter applications also as the data are represented and concluded with the inferences and reported with the utilities for electronic and pharma utilities.
Keywords: AD, AO, Crystals, Fluorescence, Influx, Nano
Full Text:
1. Introduction
Complexes of glycine have recently attracted attraction due to their potential applications in ferroelectricity,1-3 dielectric properties4 and nonlinear optical properties.5-9 Nonlinear optical materials10-14 are very important for the current researchers due to their importance for producing the second and third harmonic generations.15-17 Single-crystal X-ray beam structure arrangement examination uncovers that the hydrated type of glycine lithium bromide takes shape in the monoclinic framework, with spacegroup P21/c. Fluorescence, filter utility and AD and AO work for BGLBMH macro and nano scalings.18-22.
2. Experimental
2.1. Synthesis
Bis-glycine lithium bromide (BGLBMH) salt was combined by dissolving Analar grade glycine and lithium bromide in a 1:1 proportion in double-distilled refined water as indicated by the accompanying response:
 LiBr + CH2NH2COOH
LiBr + CH2NH2COOH
Li+[CH2NH2COOH].Br−
The temperature of the arrangement was kept up at a consistent temperature of 52°C, and the BGLB salt was acquired by the dissipation of the dissolvable. The immaculateness of the salt was expanded by progressive recrystallizations by suitable solutions.
2.2. Crystal growth
BGLBMH crystals were developed, at first by slow evaporation was utilized to develop single crystals of BGLBMH at room temperature. A saturated solution of 250 ml was set up as per dissolvability information. It was kept in an undisturbed territory for evaporation to get fine crystals of BGLBMH.
3. Results and Discussions
3.1 SXRD and PXRD studies and milling scheme
The Single-crystal XRD (SXRD) and Powder XRD (PXRD) for macro and nanocrystals of BGLBMH crystals are represented by data of SXRD by Table.1 and PXRD pattern by Fig.1 and macro crystals are of size as 13 x 7 x 5 mm3 and powder XRD pattern shows the indexed data of BGLBMH with the 2θ territory value extended from 10-80 degrees at a pace of 1°/min and the macro crystals are off with the crystalline system of monoclinic in nature and PXRD intensity from 0-100000 values and the different planes of reflections were regulated utilizing the product AUTOX93 and matched up with powder XRD pattern of the existing one with the earlier cases.
The nano scaling is obtained from milling the macro crystals using Milling type as Retsch mill for the time extent of above 40 hours with a milling swiftness of 250 rpm as given in Table.2 and the variations of time Vs nm scaling is given in Fig.2.
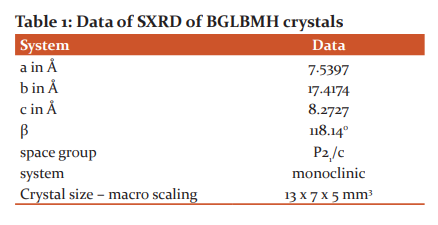
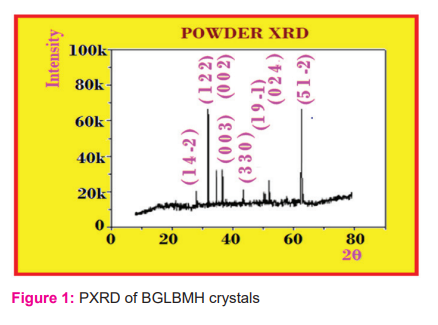

3.2. Fluorescence (FL) and NLO - SHG
Fluorescence (FL) is the practice of emanation of radiance by a particle after absorbing preliminary radiation (excitation level). Much of the time, the discharged light has a more drawn out frequency, and in this way lower vitality, than the assimilated radiation.
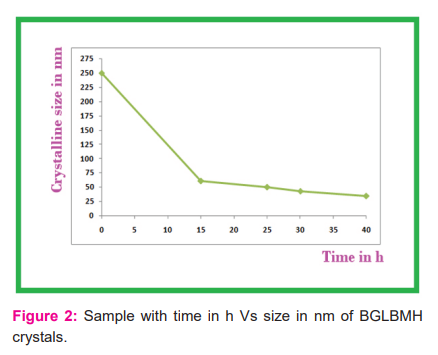
The bandgap is 3.08 eV which is the emission of 403 nm as emission FL value for macro scaling and 397 nm for nano scaling with the band gap of 3.12 eV. As the size of the sample from macro to nano of 34 nm, the FL value varies by 6 nm in decremented value and energy value varied by 0.04 eV as incremental one, the Fig.3 and Fig.4 represents the FL value for macro scaling and nano scaling for BGLBMH crystals.
The SHG of BGLBMH is analysed and from that, the crystal shows efficient output related to lasers for frequency doubling and is of 1.25 times than the referenced specimen of KDP for the non-linear effect and is mainly due to the strongest hydrogen bond present in the specimen.
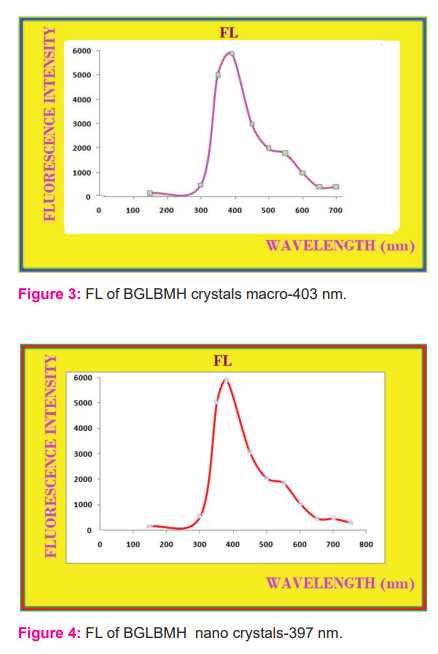
3.3. Anti-diabetic (AD) study and influx study
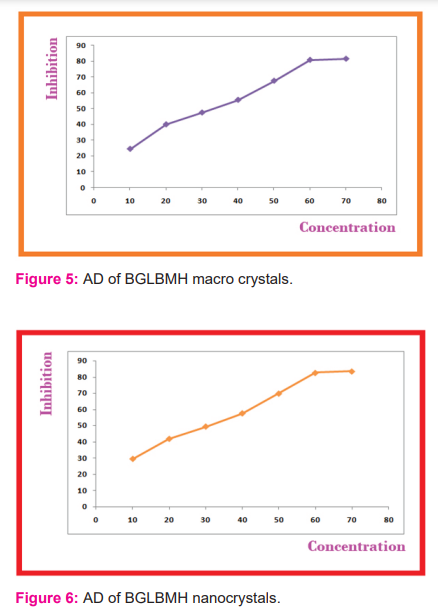
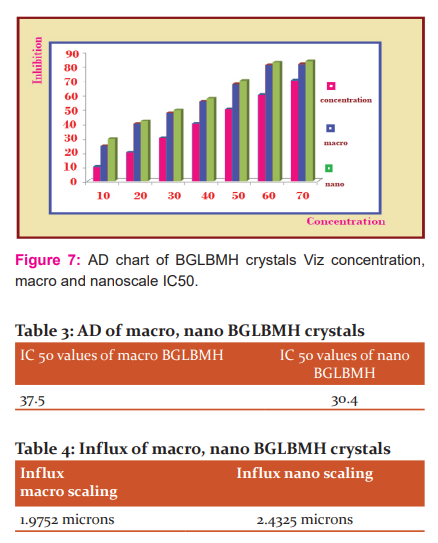
The Anti-diabetic (AD) studies for the grown BGLBMH crystals were carried out. The IC 50 values for macro and nano scales are 37.5 and 30.4 correspondingly for the AD of BGLBMH crystals and the inhibition values are increased as the macro is converted to nanoscale and in IC 50 case its decreased from macro to nano scaling, which is suitable for AD work as mentioned in Table.3 and the graphical representation for macro and nano scaling is shown in Fig.5 as well as Fig.6-7.
The AD activity of BGLBMH is mainly due to the presence of Glycine and bromide and not due to the lithium by the chemical structure and properties. The cumulative column chart is shown in Fig.7 with a concentration in macro and nanoscale.
Influx value of 1.9752 microns of BGLBMH represents that the macro influx refers that BGLBMH is a good NLO crystal as it is less than 10 microns as the case represents the non-centrosymmetricity and 2.4325 microns is a nano-level influx. So, the BGLBMH macro and nanocrystals are used in filter applications also as the data are represented in Table.4.
3.4. AO-DPPH, FRAP and the total value of Antioxidant activity
DPPH radical explore ability assay represented is carried out [21]. DPPH outlines violet or the purple colour in methanolic solution and become lighter out to shadows of yellowish colour in the existence of antioxidants. 250µl of the test solution and 1ml of DPPH solution was added all along with 0.4 ml of 50 mM tris HCl buffer and the amount was made up to 2 ml with purified water and the tube was protected in dark arrangement for 0.5-1 h and the reading was measured at 517 nm using spectro-photometer of make LT 291 labtronics microprocessor. Ferric reducing capability of the extorts was explore using the potassium ferricyanide-ferric chloride technique. 1 ml of the extorts was assorted with 0.1M phosphate-buffered solution and also with 2 ml of the 0.1% potassium ferricyanide. The assortment was protected at 500oC for 0.5-1 h and 2 ml of the 10% trichloroacetic acid solution was supplemented to stop the effect, by centrifugation at 5000 rpm for around 8 minutes the clear solution was estranged and added 0.1% FeCl3 solution (2 ml) this was measured at 700 nm using a spectrophotometer. The total antioxidant activity of the trial was resolved by using the phosphor molybdenum process. The assay was mainly on the reduction of Mo (VI) to Mo (V) by the trial and succeeding creation of green phosphate composite at the value of acid pH. 0.2 ml trial was collected with 2 ml of reagent solution (0.6 M sulphuric acid, 28 mM sodium phosphate and 4 mM ammonium molybdate). The solution was protected at 90°C for 1.5 h. following the action of cooling in room warmth; the absorbance of the solution was measured at 695 nm using a spectrophotometer. The totality antioxidant action is articulated as the amount of gram equivalent of ascorbic acid as shown in Table.5.

Conclusion
BGLBMH crystals are put in order by slow evaporation solution growth method and nanocrystal by milling method. BGLBMH are of the system as monoclinic with space group of P21/c by SXRD and the nano outline of BGLBMH crystals are reported for 34 nm likewise indexed by PXRD and crystals speciality is THG and band gap is 3.08 eV which is of 403 nm as emission FL value for macro scaling and 397 nm for nano scaling with the bandgap of 3.12 eV. Influx value of 1.9752 microns of BGLBMH represents that the macro influx refers that BGLBMH is a good NLO crystal and is 1.25 times than KDP, the non-centrosymmetricity and 2.4325 microns in nanoscale represents that it is good and better prospects in filter utility. The macro crystals have good scope for anti-diabetic by the Glycine, bromide presence and have increased in inhibition rate as concentration increases and the IC value as 37.5 and in a nano form, it is 30.4 and has good AD activity of BGLBMH crystals. Both macro and nano BGLBMH crystals are increased in inhibition values while concentration increases as shown in column chart and AO for BGLBMH is also analyzed and reported.
Acknowledgements
Author(s) is grateful to EGSP management for R&D collaborative work - permission to complete this paper and for the permission to publish.
Declaration of interest statement & conflict statement
All authors are equally contributed and there are no conflicts to declare.
ORCID ID (Dr K.SenthilKannan) 0000-0002-8935-6470
Funding
No funding is received and authors contribute their own funding for studies
Author’s contribution
Alphonse Arockia Jenecius 1 - Antioxidant study
Ramachandra Prasad Divya2* - crystal growth
Kannuswamy SenthilKannan3* - crystal growth and studies and write up and nano scaling work
Irudaya Antonat Sophia4 - FL study and write up
Balaraman Ravindran5 - Overall corrections & suggestions
Ramamurthi Krishnaveni6 - AD work and write up
References:
-
Pepinsky R, Vedam K, Okaya Y. New room-temperature ferroelectric. Phy Rev. 1958;110:1309–11.
-
Hoshino S, Okaya Y, Pepinsky R. Crystal structure of the ferroelectric phase of (glycine) H2SO4. Phy Rev. 1959;115:323–31.
-
Deepthy A, Bhat HL. Growth and characterization of ferroelectric glycine phosphite single crystals. J Cryst Growth. 2001;226:287–93.
-
Hoshino S, Mitsui T, Jona F. Dielectric and thermal study of Tris-Glycine sulfate and Tris-glycine fluoberyllate. Phy Rev. 1957;107:1255–8.
-
Subramanian P, Hariharan N, Sobhanadri J. Electron paramagnetic resonance study of free radicals in γ – irradiated single crystal of tris(glycine) calcium (ll) dibromide. Pramana J Phys. 1986;26:367–72.
-
Hariharan M, Rajan SS, Srinivasan R. Crystal structure of a complex of glycine with zinc chloride. Z Kristallogr. 1989;188:217–22.
-
Narayan Bhat M, Dharmaprakash SM. New nonlinear optical material: glycine sodium nitrate. J Cryst Growth. 2002;235:511–6.
-
Natarajan S, Mohan Rao JK. Crystal structure of bis(glycine)calcium(II)dichloride tetrahydrate. Z Kristallogr. 1980;152:179–88.
-
Natarajan S, Ravikumar K, Rajan SS. Crystal structure of diaqua nitrato glycine calcium(II) nitrate. Z Kristallogr. 1984;168:75–82
-
Narayanan P, Venkataraman S. Crystal structure analysis of some addition compounds of glycine. Z Kristallogr. 1975;142:52–81.
-
Ravikumar K, Rajan SS, Natarajan S. Structure of diaquabis (glycine) cobalt (II) bromide. Z Kristallogr. 1985;171:201–7.
-
Baran J, Drozd M, Pietraszko A. Crystal structure and vibrational studies of glycine LiNO3 and glycine NaNO3 crystals. J Polish Chem. 2003;771:1561–78.
-
Rajesh NP, Kannan V, Ashok M. A new nonlinear optical semi-organic material: cadmium thiourea acetate. J Cryst Growth. 2004;262:561–6. doi: 10.1016/j.jcrysgro.2003.10.064
-
Xue D, Zhang S. Effect of hydrogen bonds on optical nonlinearities of inorganic crystals, Chem. Phys. Lett.1999;301(5–6):449–52.
-
Yu D, Xue D, Ratajczak H. Bond-valence parameters for characterizing O–H2O hydrogen bonds in hydrated borates. J Mol Struct. 2006;792:280–5. doi: 10.1016/j.molstruc.2005.11.047
-
Blois MS. Antioxidant determinations by the use of a stable free radical. Nature. 1958;181:1199–200.
-
SenthilKannan K, Venkatachalam K, Thamarikannan P, Kalaipoonguzhali V, Kannan S, and Jothibas M, Computational and experimental studies of acetoacetanilide crystals. AIP Conf Proc. 2020; 2270, 040014: 1-4 https://doi.org/10.1063/5.0019332
-
SenthilKannan K, Thamaraikannan P, Kannan S, Kalaipoonguzhali V, Ashok KR, and Venkatachalam K, Computational analysis of LPNPTH anisotropies for dementia for Alzheimer’s syndrome by DFT and molecular harbouring. AIP Conf Proc. 2020;2270, 040008:1-5 https://doi.org/10.1063/5.0019331
-
SenthilKannan K, Gunasekaran S, Synthesis, Growth and Interpretation of (Organic NLO) 4 Bromo-4l Chloro Benzylidene Aniline (BCBA) Crystals. Int J Front Sci Tech.2013;3(3):29-36.
-
SenthilKannan K, UV, SEM, powder XRD of AAC crystals, Int. J Chemtech Res. 2014; 6(5):3187-90.
-
Sathishkumar R, SenthilKannan K, Magesh CJ, Venkatapathy K, Vimalan M, Sankar D, et al. Synthesis, Growth and Characterization of 2,5 – Dimethyl-N-(3-phenyl prop-2-en-1-ylidene) Aniline (2,5 DPPA) Single Crystal: A New Second-Order Nonlinear Optical Material for Photonic and Optoelectronic Applications. Optik. 2021; 226(2),165947:1-11, https://doi.org/10.1016/j.ijleo.2020.165947
-
SenthilKannan K, Growth and Characterisation-XRD, NLO of Bis 2 Amino Pyridinium Maleate (B2APM) crystals, Int J Eng Sci Mat. 2018; 7:1-3.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License