IJCRR - 13(15), August, 2021
Pages: 181-183
Date of Publication: 10-Aug-2021
Print Article
Download XML Download PDF
Study of Biological Efficiency Biological Active Additive \"Avena-Uz\"
Author: Farmanova Nodira Takhirovna, Nurullayeva Dilobar Khamid Qizi, Sultanova Rano Khakimovna, Abdurahmonova Sabohat Abdugabarovna
Category: Healthcare
Abstract:Introduction: To expand the range of domestic biologically active additives, the composition of dietary supplements \"Avena-Uz\" was developed, which includes dry extracts of sowing oats and rose hips. As a result of the research, the quality standards of the studied dietary supplement were established. Aims: Study of the specific effectiveness of \"Avena-Uz\". Methodology: The study of choleretic activity was carried out according to the generally accepted method of N.P. Skakun and A.N. Oleinik Results: The research results showed that the rate of bile secretion in rats receiving choleretic dietary supplements compared with healthy animals for 4 hours of the experiment significantly increased on average by 25.6%. At the same time, there was an increase in the content of bile acids in bile by 39.8% and cholesterol by 3.1% in comparison with the control animals. Conclusions: The study of the specific efficacy of \"Avena-uz\" showed that the dietary supplement can stimulate the exocrine function of the liver, and also normalizes cholesterol, triglycerides in the blood, the content of digestive enzymes - alkaline phosphatase and amylase.
Keywords: BAA (Biologically active additives), Control group, Efficiency, Oat extract, Bile cholesterol
Full Text:
INTRODUCTION
All over the world, in recent years, there has been an increase in interest in drug-free methods of prevention and treatment of the body. At present, biologically active additives can be attributed to a fairly large part of the pharmaceutical industry - compositions of biologically active substances intended for direct intake with food or introduction into food products.2,3,4 Not being drugs, they are used to improve and normalize the function of organs, reduce the risk of diseases, are used as enterosorbents, etc.5,6,7 To expand the range of domestic biologically active additives at the department of the Tashkent Pharmaceutical Institute, a composition of dietary supplements "Avena-uz" was developed, which includes dry extracts of oats and rose hips.8
As a result of the research, the quality standards of the studied dietary supplement were established, which served as the basis for the preparation of the corresponding regulatory documentation.3,5
To recommend the developed dietary supplement "Avena-uz" in medical practice, an assessment of its biological effectiveness was carried out.
EXPERIMENTAL PART
The effectiveness of the Avena-uz dietary supplement was evaluated on non-fertile white rats with a weight of 200-210 g. Animals were kept under vivarium conditions on a standard diet. 24 hours before the start of the experiment, the animals were deprived of food. The experimental animals were divided into 2 groups of 6 animals. Animals of the first group (control) were injected intraventricularly with purified water in a volume of 1 ml per 200 g. Animals of the second group (experimental) were treated with BAS bile "Avena-uz" at a dose of 5 ml/kg for 30 days. After 30 days, the bile function of the liver was examined: the abdominal cavity was opened, the common bile duct of the addicted animals was isolated and cannulated. The choleretic effect of the preparation was judged by the total amount of bile isolated, the concentration and the number of its components (cholesterol and total bile acids).2,4
RESULTS
The animals of the experimental group remained active, tidy, willingly ate food, the hair was smooth, shiny, and the animals responded adequately to external stimuli. The results of the experiment are shown in Table 1.

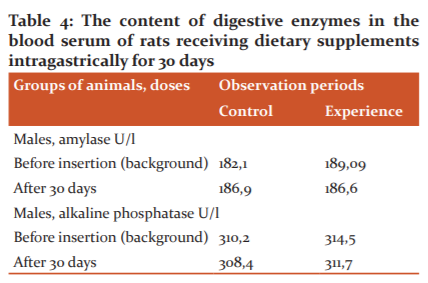
The research results showed that the rate of bile secretion in rats receiving choleretic dietary supplements compared with healthy animals for 4 hours of the experiment significantly increased on average by 25.6%. At the same time, there was an increase in the content of bile acids in bile by 39.8% and cholesterol by 3.1% in comparison with the control animals. Consequently, the introduction of dietary supplements stimulated bile secretion, which is expressed not only in an increase in bile secretion but also in an increase in the content of its main components. The morphological composition of peripheral blood (the number of erythrocytes, leukocytes, platelets, haemoglobin level), biochemical parameters (the level of cholesterol, triglycerides, alkaline phosphatase, amylase) were studied before the introduction, as well as 30 days after the start of the drug administration. The counting of blood cells was performed on an automatic blood counter. Biochemical parameters and enzyme activity were determined using a biochemical semi-automatic analyzer.

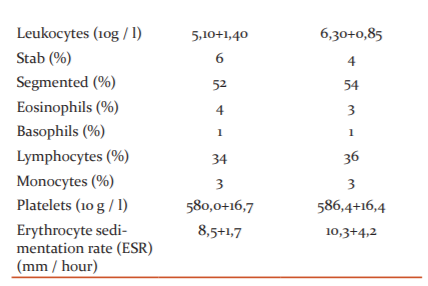
The complex laboratory data obtained in each observation from rats treated with dietary supplements were compared with the data of control animals. During the experiment, no statistically significant differences were found in the number of erythrocytes, leukocytes, platelets, haemoglobin levels in animals that received the studied dietary supplement compared to the control.
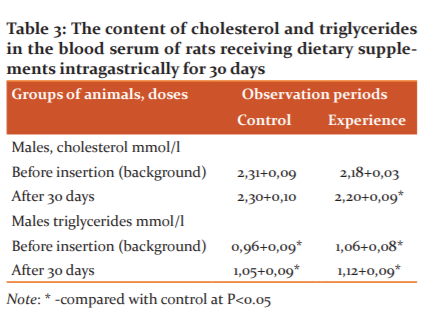
As can be seen from the table, intragastric administration of dietary supplements to rats daily for 30 days reduces the level of cholesterol and triglycerides in the blood of experimental animals.
CONCLUSION
Thus, the study of the specific effects of the biologically active supplement "Avena-uz" showed that biologically active additives have the property to stimulate the external secretive function of the liver, does not show statistically significant differences in the number of red blood cells, white blood cells, platelets, haemoglobin levels in animals.
The biologically active supplements received compared to the control, as well as normalizes the cholesterol, triglycerides in the blood, the content of digestive enzymes - alkaline phosphatase and amylase.
ACKNOWLEDGEMENT
The authors are grateful for the help in carrying out this study to the head of the pharmaceutical enterprise "Novopharmkomplekt" Nurullaev A.A. and Ibragimov K.A.
Conflict of Interest:
The authors declared that there is no conflict of interest.
Source of Funding: NIL
Contribution:-
Farmanova NT. - development of the composition of the biologically active additive, writing and correction of the article.
Nurullaeva DH.- development of composition and technology of biologically active additive, preparation of samples.
Sultanova RK - conducting studies on the effectiveness of a biologically active additive, interpreting the results.
Abdurakhmanova SA - conducting studies on the effectiveness of a biologically active additive.
References:
-
Bespalov VG, Nekrasova VB, Iordanishvili AK. Modern view of biologically active food additives and their use for therapeutic and prophylactic purposes in clinical medicine. Med. XXI Century. 2007; (9): 86-94.
-
Mavlanov Sh.R., Khakimov 3.3., Rakhmanov A.Kh. The influence of lesbohol on the glycogen content in the liver during its acute toxic lesion. Inf Jimmu Pharmac. 2017; (1):129-134.
-
Severina AS, Shestakova MV. The place of dietary supplements in the treatment of diabetes mellitus. Diab. 2007; (2):147-151.
-
Stefanov AV. Preclinical studies of drugs. Avic. 2002; (7):356-373.
-
Farmanova NT, Pulatova LT. To the issue of studying the quality and safety indicators of a biologically active additive of choleretic action. III International Scientific and Practical Conference «Harmonization of Approaches to Pharmaceutical Development». 2020:177-178.
-
Farmanova NT, Nurullaeva DKh. On the question of studying the chemical composition of the biologically active additive "Avena-uz". J Izv GSTU. 2020; 4(4):288-289.
-
Johnson M, Olufunmilayo LA, Anthony DO, Olusoji EO. Hepatoprotective Effect of Ethanolic Leaf Extract of Vernoniaamygdalina and Azadirachtaindica against Acetaminophen-Induced Hepatotoxicity in Sprague-Dawley Male Albino Rats. Am J Pharmac Sci. 2015; 3(3):79-86.
-
Rane J, Kadhai R, Bakal RL. Liver diseases and herbal drugs: A review. J Innov Pharm Bio Sci. 2016;13 (2):2349-2759.
-
Irani R, Khaled KL. Quantitative Analysis of Nutrients in the Gum Exudates of Acacia nilotica. Int J Cur Res Rev. 2020;12(08):11.
-
Okamoto T, Kajino K, Hino O. Hepatoprotective drugs for the treatment of virus-induced chronic hepatitis: from hyper carcinogenic state to hypo-carcinogenic state. J Pharm. 2001 Nov;87(3):177-80. doi: 10.1254/jjp.87.177. PMID: 11885965.
-
Wang FS, Fan JG, Zhang Z, Gao B, Wang HY. The Global Burden of Liver Disease: The Major Impact of China. Hepat. 2014; 60(6):2099-2108.
-
Kugaler Ganesan Parthiban, Balakrishnan Senthil Kumar, Ponnaiyah Ramajayam Institute of Science & Technology, Thanjavur, Tamil Nadu Rangasamy Manivannan and Natesan Senthil Kumar. Herbal drug comprehensive approach for treating liver disease and focus towards herbal medicine. Int J Cur Res Rev. 2021;13(03):60.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License