IJCRR - 13(6), March, 2021
Pages: 09-13
Date of Publication: 20-Mar-2021
Print Article
Download XML Download PDF
mRNA Expression of Somatostatin Receptors (1-5) in MCF7 and MDA-MB231 Breast Cancer Cells
Author: Alkhansa Mahmoud, Maria Teresa Mancuso, Barbara Tanno, Md Zuki Abu Bakar, Hazilawati Hamzah, Mohd Hezmee Mohd Noor
Category: Healthcare
Abstract:Background: Breast cancer is one of the most common types of cancer and heterogeneous disease. Somatostatin receptors (SSTRs) are expressed in normal and tumour tissues including breast cancer cells. Objective: The aim of this study to evaluate the expression of SSTRs subtypes 1 \? 5 in MCF7 and MDA-MB231 human breast cancer cell lines and compare the levels between two cells. Methods: The detection of mRNA expression levels of SSTRs in MCF7 and MDA-MB231 was performed using quantitative�polymerase chain reaction (qPCR). Results: SSTR1, 2, 3 and 4 mRNA levels were significantly higher in MDA-MB-231 about MCF-7. The expression of SSTR 4 mRNA was highest in MDA-MB231 and MCF-7 cell lines, however, SSTR3 mRNA was least expressed in both cell lines. Conclusion: SSTRs subtypes (1 \? 5) were expressed in both MDA-MB231 and MCF7 cells. However, the levels of expression differ between both cell lines.
Keywords: Breast cancer, Cell lines, Somatostatin receptors and mRNA expression
Full Text:
INTRODUCTION
Breast cancer is the most common type of cancer and the second leading cause of cancer-related deaths in women worldwide.1,2 It is the most common cancer in both developed and developing countries,2but still diagnosed in late-stage due to lack of awareness and knowledge for most of risk factors, signs and symptoms of breast cancer.3 Breast cancer is considered a heterogeneous disease because of the changes in the mammary epithelial cells leading to aggressive cell proliferation.4 The three main biomarkers of interest in breast cancer include estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2). Currently, four molecular subtypes with different levels of expression of these receptors are luminal A, luminal B, HER2 enriched, and basal-like have been identified. Triple-negative breast cancer (TNBC), which is a part of the basal-like subgroup, is characterized by the lack of PR, ER, and HER2 expression.5 Different breast cancer subtypes extremely high mortality, poor drugs response and recurrence.6 At the most advanced stage of breast cancer, in particular, the hormone-independent cancers develop resistance to therapy and leading to increasing cases of mortality. Breast cancer cure and control includes surgery, radiation therapy and chemotherapy as well as hormone therapy.7The peptide hormones are expressed in tumour tissues affected cellular process regulation and proliferation which causing therapy resistance.8 Somatostatin (SST) is an endogenous peptide known to inhibit the cellular processes such inhibits the motility and acid secretion of GI, it’s effectively stopped bleeding in cases with acute upper GI bleeding,9 neurotransmissions, hormonal secretion and cell proliferation as well as induced apoptosis through SSTRs subtypes (1-5) encoded by five distinct SSTR genes on chromosomes 14, 16, 17, 20 and 22, respectively.10 Somatostatin receptors (SSTRs) are G-protein-coupled plasma membrane receptors, initially secreted as a long precursor molecule; it undergoes specific enzymatic degradation generation with two forms of SST peptides, SS-14 and SS-28, as their natural ligands.11 SSTRs1-5 expressed in normal and tumour tissues depending on cell type. In normal tissues, SSTRs are found mainly in the brain, pancreas, stomach and kidney, while in tumour tissues their expression depending on the type of tumour and biological characteristics.12 SSTRs have direct and indirect effects on tumour biology. The direct effect includes the inhibition of tumour proliferation and induction of pro-apoptotic pathways including both intrinsic and extrinsic apoptosis pathway.13,14 Meanwhile, the indirect effects include the inhibition of hormones and growth factors.15,16 SSTRs mediate signal transduction pathways via inhibition of adenylyl cyclase (AC) and guanylyl cyclase (GC), protein phosphorylation and activation of mitogen-activated protein kinase (MAPK).17,18 However, the activation of signalling pathways affected cell cycle arrest through activation of cyclin-dependent kinase inhibitor (p27Kip1) and apoptosis. Cancer cells with positive SSTRs are less malignant with higher survival whereas the lack of SSTRs expression has been associated with the poorly differentiated and invasive tumour. However, several effects demonstrated subtype selectivity, and subtype-specific signalling has been reported.19 SSTR1, 2, 4 and 5 frequently interfere with the mitogen-activate protein kinase pathway to modulate cell proliferation, whereas SSTR3 was indicated to have an increased potential to induce apoptosis.4 Furthermore, SSTR2 is considered a prognosis factor because it is associated with low proliferative and invasive breast cancer. Also, SSTRs being frequently expressed in the same cell, and the existence of ligand-induced dimerization proposed for G?protein-coupled receptors.12 Therefore, somatostatin analogues (SSAs) have been used in the treatment of SSTR-positive tumours.14 However, the therapeutic results of SSAs treatment varied due to different SSTR expression patterns and reasons that are not understood.20 SSTRs levels have been investigated in different kinds of human cancer such as hepatocellular carcinoma, pancreatic cancer and breast cancer.21 SSTRs are highly expressed in neuroendocrine tumours but their levels of expression in breast cancer are not well documented.22 However, in breast cancer, SSTRs are expressed in different levels and are correlated with various histological markers in a receptor-specific manner.23 Previous studies reported that of SSTRs expressed variety in tumour tissues and cancer cell lines.24 In the present study, MCF-7 (estrogen-receptor-positive breast cancer cell line) and MDA-MB231 (estrogen receptor-negative breast cancer cell line) have been used in vitro as common breast cancer models to determine the expression of these receptors. The MCF7 cell line characterised by differentiated mammary epithelium including the ability to process estradiol via cytoplasmic estrogen receptors. While, MDA-MB231 is a highly aggressive, invasive but poorly differentiated triple-negative breast cancer (TNBC) cell line that lacks receptors for estrogen (ER) and progesterone (PR) expression, as well as human epidermal growth factor receptor 2 (HER2) amplification,25 and is known to be resistant to several anti-cancer agents. 26 The aim of this study was, therefore, to evaluate the mRNA expression for SSTRs (1-5) in human breast cancer cell lines MCF7 and MDA-MB231 using quantitative real-time polymerase chain reaction (qRT- PCR).
Materials and methods
Cell culture
MCF7 and MDA-MB231 human breast cancer cell lines were kindly donated by the National Cancer Institute Regina Elena Rome, Italy. Both cells were purchased from the American Type Culture Collection (Manassas, VA, USA). MCF7 and MDA-MB231 cells were grown in a humidified 37°C incubator in 5% CO2 and cultured in Dulbeccos modified essential medium /F12 complete media supplemented with 10% fetal bovine serum, 1% penicillin/streptomycin and 1% L-glutamine.
RNA extraction
Total RNA from MCF7 and MDA-MB231 cell lines was extracted by using EXIQON kit. The concentrations of total RNA were quantified by Nanodrop 2000 (Thermo Scientific, Hvidovre, Denmark), and all samples were stored at -20°C until analysis.
Reverse-Transcriptase Polymerase Chain Reaction (RT-PCR)
RNA was converted into complementary DNA (cDNA) by reverse transcriptase process according to the manufacturer's instruction. After addition oligo (dT), samples were incubated at 42°C for 1 hour in a thermal.
Quantitative Real-time Polymerase Chain Reaction (qRT -PCR)
qRT-PCR was performed using q RT- PCR Systems (Bio-Rad) to detect the expression of SSTRs1-5 levels in MCF7 and MDA-MB231 breast cancer cell lines. qRT-PCR was performed using 2 μg of retro-transcribed RNA and normalized with GADPH. The quantity of mRNA relative to the reference gene was calculated by 2-ΔC? methods, the analysis type is Singleplex, and RQ min/max confidence level is 95.0. Samples were analysed using SYBR Green Supermix (Bio-Rad) according to the manufacturer’s instructions. The sample analysis was performed in triplicate and the experiments have been repeated in different batches of cell lines. Primer sequences were obtained from thermo-fisher used for SSTRs (1-5) are shown in
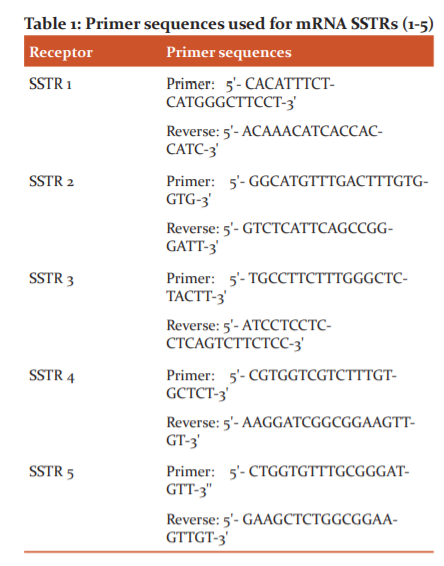
Statistical analysis
The SPSS software version 22 (IBM®) was used for the analysis. The mean and standard deviation of SSTRs expression levels in MCF7 and MDA-MB231 cell lines were compared using a T-test. P-values of < 0.05 were considered statistically significant.
Results
SSTR1-5 mRNA expression was determined in both MCF-7 and MDA-MB-231 breast cancer human cell lines (Figure 1) and the overall expression levels differ between the two cell lines. The SSTR1, 2, 3 and 4 mRNA levels were significantly higher in MDA-MB231 cell line in relation to MCF-7 cell line (P=0.02, 0.002, 0.001, 0.01) respectively. While no different significant of SSTR5 in MCF7 compared to MDA-MB231 (P=0.2). The expression of SSTR4 mRNA was highest in the MDA-MB231 cell line followed by SSTR2, SSTR1, SSTR5 and SSTR3 mRNA. In the MCF7 cell line, SSTR4 has the highest expression levels, followed by SSTR1, SSTR5, SSTR2 and SSTR3. SSTR3 mRNA was least expressed in both cell lines, while SSTR4 has highly expressed in both cell lines too. An arbitrary score was adopted to summarize the expression levels of SSTRs as in (Table 2).
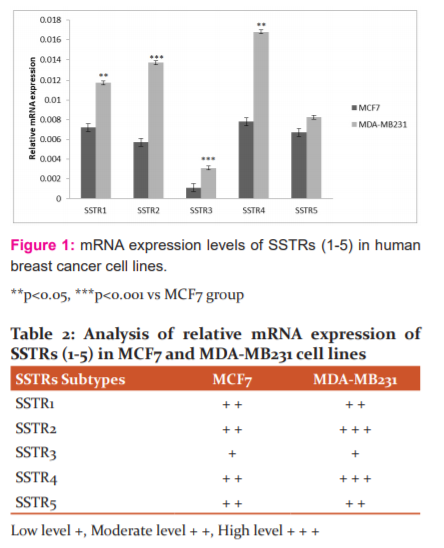
Discussion
In this study, our data showed that all the SSTRs1-5 were expressed in both MCF-7 and MDA-MB-231 breast cancer cell lines. Similar findings have been reported that all the SSTR subtypes were expressed in both MCF7 and MDA-MB231 and SSTR3 mRNA being the least expressed in both cell lines. SSTR4 was found to be express highly in MDA-MB231 cell lines while it is often reported that SSTR2 is predominant on breast cancer cells,27 however, our somatostatin receptors' expression levels differ from the literature, but several other studies were in agreement with our results.7 The expression of SSRT2, SSTR3 and SSTR4 were significantly higher in the MDA-MB231 cell line.28 The association between SSTRs and ER/PR positive receptors, it might be suggested that SSTR were overly expressed in MDA-MB231 cell lines that were correlated with poorly differentiated cancer cells. STTR1, 2, 3 and 4 have a key role in blocking tumour growth by inhibiting cell cycle progression and inducing apoptosis. Furthermore, SSA antagonist may have more clinical benefits for ER and PR negative tumours. The highly expressed SSTR4 and low expressed SSTR3 agreed with earlier reports.29,30 The high expression of SSTR3 and SSTR2 or SSTR4 in MCF7 cell lines were associated with apoptosis. Meanwhile, SST enhanced cytotoxicity via SSTR2 and SSTR3.31 The low-level expression of SSTR3 has made it a target for breast cancer therapy. The activation of SSTR3 in MCF7 and MDA-MB231 breast cancer cell lines by SST for cancer treatment is now been explored.31,32 However, the mechanism of SSTR3 in apoptosis and cell cycle arrest is still unclear.30 Besides, estrogen and progesterone receptors in MCF7 are important in breast cancer prognosis and development33, and the positive estrogen effect on SSTR2 expression on regulation in breast cancer cells development has been documented.34 Furthermore, many SST analogues have been synthesized for activation SSTRs while SST agonist is currently under development to control cancer cell proliferation.35,36 Several studies demonstrated that SSTRs expression in breast cancer is down-regulated either in more aggressive and less differentiated tumours37 or in anti-estrogen agents.38 In this study, the high levels of SSTRs expression were documented in aggressive tumours and thus, MDA-MB231 may be considered as a target for therapeutic strategy. Meanwhile, the activation of the expression levels of SSTR1, SSTR2, SSTR3 and or SSTR4 might enhance apoptotic activity in MCF7 cells. Several previous studies have investigated that SSTR expression may be able to be explored for further insights into the therapeutic of breast cancer. Besides, the antiproliferative role of SST and its analogues have also been demonstrated. Several in vitro studies have investigated the anti-proliferative effect of somatostatin analogues in breast cancer cells. Previous studies have also shown that SSTR2 overexpression produces an anti-proliferative role in the estrogen-dependent MCF-7 cells by inducing apoptosis and decreasing EGFR expression.12 These results highlighted the SSTRs -targeted therapy in which the evaluation that SSTR1-5 is expressed in both breast cancer cell lines MCF7 and MDA-MB231. These findings recommended more understanding of the role of SSTRs functions in breast tumour biology to improve therapy in estrogen receptors positive and estrogen receptors negative breast cancers.
Conclusion
SSTRs (1-5) were expressed in both MDA-MB231 and MCF7 cell lines, but the level of expression differed between both cell lines. The activation of SSTRs receptors in ER+, PR+ tumour may be considered. SSTRs overexpression in aggressive tumours (ER-, PR-) may be considered as a target for therapeutic strategy. Future research is warranted to study the functions of SSTRs.
Abbreviations
SSTRS: Somatostatin receptors; qRT-PCR: Quantitative polymerase chain reaction; mRNA: Messenger RNA; ER: Estrogen receptors; PR: Progesterone receptors.
Acknowledgements
Authors acknowledge the Programme for Training and Research in Italian Laboratories (TRIL) fellowship. The authors would like to thanks Dr G. Bossi for kindly gifted MCF7 and MDA-MB231 human breast cancer cell lines. The Authors acknowledge the immense help received from the scholars whose articles are cited and included in references to this manuscript. The authors are also grateful to authors/editors/publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
Conflict of interest
Nothing to report
Source of funding
International Centre for Theoretical Physics (ICTP) - Training and Research in Italian Laboratories (TRIL).
Conflicts of interest
The authors have no conflict of interest.
References:
-
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin 2018;68(1):7–30.
-
Park’s textbook of Preventive And Social Medicine. K. Park. Twenty-second edition; 2013: Non- Communicable Diseases; Breast cancer; page. No; 359.
-
Rajini S, Kamesh C, Senthil VS. Knowledge of breast cancer and its risk factors among rural women of Puducherry – a cross-sectional study. JCRR 2015;07(19):60-64.
-
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 2010;127(12):2893-2917.
-
Kondov B, Milenkovikj Z, Kondov G, Petrushevska G, Basheska N, Bogdanovska-Todorovska M, et al. Presentation of the Molecular Subtypes of Breast Cancer Detected By Immunohistochemistry in Surgically Treated Patients. Maced J Med Sci 2018;6(6):961-967.
-
Cheang MC, Voduc KD, Tu D, Jiang S, Leung S, Chia SK, et al. Responsiveness of intrinsic subtypes to adjuvant anthracycline substitution in the NCIC.CTG MA.5 randomized trial. Clin Cancer Res 2012;18:2402–2412.
-
Hsu YL, Kuo PL, Lin LT, Lin CC. Asiatic acid, a triterpene, induces apoptosis and cell cycle arrest through activation of the extracellular signal-regulated kinase and p38 mitogen-activated protein kinase pathways in human breast cancer cells. J Pharmacol Exp Ther 2005;313:333-344.
-
Lahlou H, Saint-Laurent N, Esteve JP. sst2 Somatostatin receptor inhibits cell proliferation through Ras-, Rap1-, and B-Raf-dependent ERK2 activation. J Biol Chem 2003;278:39356-39371.
-
Geredeli C. Comparison of Somatostatin and Famotidine for the Treatment of Nonvariceal Acute Upper Gastrointestinal Bleeding. Int J Curr Res Rev 2018;10(08):25-27.
-
Günther T, Tulip G, Dournaud P, Bousquet C, Csaba Z, Kreienkamp HJ, et al. International union of basic and clinical pharmacology. CV. Somatostatin receptors: Structure, function, ligands, and new nomenclature. Pharmacol Rev 2018;70:763?835.
-
Day R, Dong W, Panetta R, Kraicer J, Greenwood MT, Patel YC. Expression of mRNA for somatostatin receptor (sstr) types 2 and 5 in individual rat pituitary cells. A double-labelling in situ hybridization analysis. Endocrinology 1995;136:5232?5235.
-
Corleto VD, Nasoni S, Panzuto F, Cassetta S, Delle Fave G. Somatostatin receptor subtypes: basic pharmacology and tissue distribution. Dig Liver Dis 2004;36 Suppl 1:S8-16.
-
He Y, Yuan XM, Lei P, Wu S, Xing W, Lan XL, et al. The antiproliferative effects of somatostatin receptor subtype 2 in breast cancer cells. Acta Pharmacol Sin 2009; 30:1053-1059.
-
Watt HL, Kharmate GD, Kumar U. Somatostatin receptors 1 and 5 heterodimerize with epidermal growth factor receptor: agonist-dependent modulation of the downstream MAPK signalling pathway in breast cancer cells. Cell Signal 2009;21:428-439.
-
Grant M, Alturaihi H, Jaquet P, Collier B, Kumar U. Cell growth inhibition and functioning of human somatostatin receptor type 2 are modulated by receptor heterodimerization. Mol Endocrinol 2008;22:2278-2292.
-
Ferrante E, Pellegrini C, Bondioni S, Peverelli E, Locatelli M, Gelmini P, et al. Octreotide promotes apoptosis in human somatotroph tumour cells by activating somatostatin receptor type 2. Endocr Relat Can 2006;13:955-962.
-
Somvanshi PK, Billova S, Kharmate G, Rajput PS, Kumar U. C-tail mediated modulation of somatostatin receptor type-4 homo- and heterodimerizations and signaling. Cell Signal 2009;21:1396-1414.
-
Florio T. Somatostatin/somatostatin receptor signalling: Phosphotyrosine phosphatases. Mol Cell Endocrinol 2008;286:40?48.
-
Lahlou H, Guillermet J, Hortala M, Vernejoul F, Pyronnet S, Bousquet C, et al. Molecular signalling of somatostatin receptors. Ann N Y Acad Sci 2004;1014:121-131.
-
Li M, Zhang R, Li F, Wan GH, Kim HJ, Becnel L, et al. Transfection of SSTR?1 and SSTR?2 Inhibits Panc?1 Cell proliferation and renders Panc?1 cells responsive to a somatostatin analogue. J Am Coll Surg 2005;201:571?578.
-
Watt HL, Kharmate G, Kumar U. Biology of somatostatin in breast cancer. Mol Cell Endocrinol 2008; 286:251-261.
-
Csaba Z, Dournaud P. Cellular biology of somatostatin receptors. Neuropeptides 2001; 35:1-23.
-
Papotti M, Kumar U, Volante M, Pecchioni C, Patel YC. Immunohistochemical detection of somatostatin receptor types 1-5 medullary carcinoma of the thyroid. Clin Endocrinol (Oxf) 2001,54:641-649.
-
Kumar U, Grigorakis SI, Watt HL, Sasi R, Snell L, Watson P, et al. omatostatin receptors in primary human breast can-cer: quantitative analysis of mRNA for subtypes 1-5 andcorrelation with receptor protein expression and tumourpathology. Breast Cancer Res Treat 2005,92:175-186.
-
Marguerite M. Vantangoli, Samantha J. Madnick, Susan M. Huse, Paula Weston, and Kim Boekelheide. MCF-7 Human Breast Cancer Cells Form Differentiated Microtissues in Scaffold-Free Hydrogels. PLoS One 2015;10(8):e0135426.
-
Ali R, Samman N, Al Zahrani H, Nehdi A, Rahman S, Khan AL, et al. Isolation and characterization of a new naturally immortalized human breast carcinoma cell line, KAIMRC1. BMC Cancer 2017;17:803.
-
He Y, Yuan XM, Lei P, Wu S, Xing W, The antiproliferative effects of somatostatin receptor subtype 2 in breast cancer cells. Acta Pharmacol Sin 2009;30,1053-1059
-
Watt HL, Kumar U. Colocalization of somatostatin receptors and epidermal growth factor receptors in breast cancer cells. Cancer Cell Int 2006;6:5.
-
Rivera JA, Alturaihi H, Kumar U. Differential regulation of somatostatin receptors 1 and 2 mRNA and protein expression by tamoxifen and estradiol in breast cancer cells. J Carcinogen 2005;4:10.
-
Yip CH, Rhodes A. Estrogen and progesterone receptors in breast cancer. Future Oncol 2014;10(14):2293-2301.
-
Burns KA, Korach KS. Estrogen receptors and human disease: an update. Arch Toxicol 2012;86:1491-1504.
-
War SA, Kumar U. Coexpression of human somatostatin receptor-2 (SSTR2) and SSTR3 modulates antiproliferative signaling and apoptosis. J Mol Signal 2012;7:5.
-
Pilichowska M, Kimura N, Schindler M, et al. Expression of somatostatin type 2A receptor correlates with estrogen receptor in human breast carcinoma. Endocr Pathol 2000;11:57-67.
-
Duran-Prado M, Gahete MD, Hergueta-Redondo M, Martínez-Fuentes AJ, Córdoba-Chacón J, Palacios J, et al. The new truncated somatostatin receptor variant sst5TMD4 is associated with poor prognosis in breast cancer and increases malignancy in MCF-7 cells. Oncogene 2012;31:2049-2061.
-
Reubi JC, Schonbrunn A. Illuminating somatostatin analogue action at neuroendocrine tumour receptors. Trends Pharmacol Sci 2013;34:676-688.
-
Cameron Smith M, Orlando C, Serio M, Maggi M. Somatostatin receptors and breast cancer. J Endocrinol Invest 2003;26:125–130.
-
Van Den Bossche B, Van Belle S, De Winter F, Signore A, van de Wiele C. Early prediction of endocrine therapy effect in advanced breast cancer patients using 99mTc-depreotide scintigraphy. J Nucl Med 2006;47:6–13.
-
Susini C, Buscail L. Rationale for the use of somatostatin analogs as antitumour agents. Ann Oncol 2006;17:1733–1742.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License