IJCRR - 3(6), June, 2011
Pages: 98-104
Print Article
Download XML Download PDF
IN-VITRO ANTIBACTERIAL SCREENING OF THE EXTRACTS OF SWERTIA CHIRAYITA LINN. AGAINST MRSA (METHICILLIN RESISTANT Staphylococcus aureus)
Author: Abdul Latif, Sumbul Rehman, Shamim Ahmad, Asad U Khan
Category: Healthcare
Abstract:Aqueous and ethanolic extract of Chirayita (Swertia chirayita Linn.) were screened for their antibacteria activity Methicillin Resistant Staphylococcus aureus (MRSA). Kirby Bauer's Disk Diffusion method an Broth Serial Dilution method according to CLSI Guidelines by W.H.O were used. It was compared with the Standard drug-Vancomycin and Plane control-DMSO (the solvent used). The Resistant drug\? Methicillin was also used to confirm the resistance of MRSA strains used. The prepared plates were incubated and results were evaluated by measuring the Zone of Inhibition - ZOI (in mm.) of drug extract. MIC and MBC was also carried out against the resistant strain. All the experiments were conducted in triplicates and in sterilized conditions. The results were analyzed statistically using gpaid software and it was found that MRSA strain was sensitive to S.chirayita showing a significant ZOI as compared to the standard drug. Present investigation indicates that S.chirayita possesses antimicrobial properties and hence can be used for future natural plant based antimicrobial agents. Thus the study provides an in-vitro proof of its antibacterial activity against MRSA. However further investigations are needed to find out its pharmacological details and to make it fit for use, for the welfare of humankind. So, that it can be used
against the infectious diseases caused by the resistant strain of Staphylococcus aureus safely and effectively.
Keywords: Swertia chirayita Linn. MRSA, Methicillin, Vancomycin
Full Text:
INTRODUCTION
Over the last three decades Methicillin Resistant Staphylococcus aureus is responsible for causing several difficult-to-treat infections in humans. Also known as Multidrug resistant S.aureus or Oxacillin resistant S.aureus (ORSA) emerged as a noscomial pathogen in early 1960s [1] it is resistant to a large group of antibiotics called beta-lactams, which include the penicillin and the cephalosporins. A study led by the CDC (Centre for Disease Control) and published in the October 17, 2007 issue of the Journal of the American Medical Association estimated that MRSA would have been responsible for 94,360 serious infections and associated with 18,650 hospital stay-related deaths in the United States in 2005. These figures suggest that MRSA infections are responsible for more deaths in the U.S. each year than AIDS. In 2007, the CDC reported that MRSA causes 19,000 deaths every year in the US, which is more than HIV/AIDS cases [2]. The studies done so far about the increasing incidence of MRSA infection are motivating the researchers to find out new pharmacological components of ?natural origin‘. Antibiotics provide the main basis for the therapy of microbial (bacterial and fungal) infections. Since the discovery of these antibiotics and their uses as chemotherapeutic agents there was a belief in the medical fraternity that this would lead to the eventual eradication of infectious diseases. However, overuse of antibiotics has become the major factor for the emergence and dissemination of multi-drug resistant strains of several groups of microorganisms [3]. The worldwide emergence of Resistant strains of Staphylococcus aureus has become a major therapeutic problem. The increasing failure of chemotherapeutics and antibiotic resistance exhibited by pathogenic microbial infectious agents has led to the screening of several medicinal plants for their potential antimicrobial activity [4]. Researchers are increasingly turning their attention to herbal products, looking for new leads to develop better drugs against multidrug resistant microbe strains. A wide group of medicinal plant preparations are available that have been used over the centuries almost exclusively on the basis of empirical evidence [6]. The primary benefits of using plant-derived medicines are that they are relatively safer than synthetic alternatives, offering profound therapeutic benefits and more affordable treatment [7]. The World Health Organization estimates that 65%-80% of the world's population use traditional medicine as their primary form of health care. The use of herbal medicine, the dominant form of medical treatment in developing countries, has been increasing in developed countries in recent years [8]. Swertia chirayita (Family Gentianaceae) is used in traditional medicines since the period of Discorides. It is rich in a wide variety of secondary metabolites such as alkaloids and flavonoids, which have been found in vitro to have antimicrobial properties [6]. Phytochemical analysis reveals that it contains a yellow bitter acid Ophelic acid (C15H 20O 13), two bitter glucosides-chiratin (C26H 48O 15) and amarogentin (C32H 38O 16); gentiopicrin, two yellow crystalline phenols, xanthone-swerchirin [9-13]. It has been found to have anti-carcinogenic, antiviral, antihelminthic, antimalarial, antipyretic, anti-inflammatory, anti-arthritic activity [14-20]. There are several reports on the antimicrobial activity of its extracts [21]. These evidences contribute to support and quantify the importance of screening S. chirayita. The pharmacological potential of Swertia chirayita reveals that its antibacterial activity towards a large number of bacterial strains, but towards MRSA its activity has not been done so far, so this was an attempt to screen its in-vitro antibacterial activity against the resistant bacteria.
MATERIAL AND METHODS
The herb was procured from the local market Baradari of Aligarh city and was properly identified by the Botanical literature available and then confirmed by Prof. S. H. Afaq from the Pharmacognosy section, Department of Ilmul Advia, Aligarh Muslim University, Aligarh. Voucher specimens (SC-0100/09-G) were preserved in the herbarium of Medicinal Plant Lab in the Department of Ilmul Advia, F/O Unani Medicine, Aligarh Muslim University, Aligarh for future reference.
Preparation of plant extract
Two different extract were prepared for analysis in the present study viz. aqueous extract and ethanolic extract as per W.H.O guidelines with some minor modifications [22].
For aqueous extract:
10 gm of the powdered drug and 150 ml of the Double Distilled Water (DDW) were put into a soxhlet apparatus. The solvent was boiled at 400C and refluxed for a period of 150 min (eleven extraction cycles).The extract was filtered and evaporated to dryness under reduced pressure in the Lyophilizer (Macro Scientific works, Delhi). It was redissolved in DMSO (Dimethyl Sulphoxide) to the desired concentration (20 mg/ml) for the study.
Similarly for the ethanolic extract: 10 gm of the powdered drug and 150 ml of the ethanol (Solvent) were put into a soxhlet apparatus and the same procedure was repeated as stated above.
Microorganisms used
Clinical strains of MRSA isolated from various sources viz. pus (Pus culture: PC), urine (Urine Culture: UC) and control strains N315, Mu50, ORSA (Oxacillin resistant Staphylococcus aureus) of the tested microorganisms were obtained from Department of Microbiology, JNMC and Hospital, AMU; Microbiology Section, Institute of Ophthalmology, JNMC and Hospital, AMU, Aligarh. The bacterial cultures were grown in Nutrient Broth (M002 Himedia Labs, Mumbai, India) and incubated at 370C for 24 hours, followed by frequent sub culturing to fresh media and were used as test bacteria. The bacterial cultures were checked to confirm the presence of sufficient number of bacterial cells on nutrient broth and maintained on nutrient agar slant.
Antimicrobial activity
Antimicrobial assay of the crude extracts was performed against pathogenic strains by Kirby Bauer‘s Disk diffusion method and Agar well method [23, 24]. The nutrient agar plates were swabbed with a suspension (106 cfu/ ml) of the bacterial strains. The wells of the equivalent size were prepared with the help of a cork borer and the drug (40µl) was poured in the respective well with the help of a micropipette. Finally, the resistant antibiotic–Methicillin disks (SD137, Himedia Labs, Mumbai, India) were placed on the prepared plates with sterile forceps and pressed properly to make complete contact with the surface of the medium. Later on these plates were kept at room temperature for 30 minutes (Pre-diffusion time) and then incubated at 370C for 24 hours. The antibiotic disks (6 mm) were used as Positive Control while the solvent used for diluting the test drug was used as the Negative Control. The diameters of the inhibition zone – Zone of Inhibition (ZOI) in mm was measured and is given in Table-1. The experiment was done in triplicate and the mean values were calculated.
Determination of minimum inhibitory concentration (MIC)
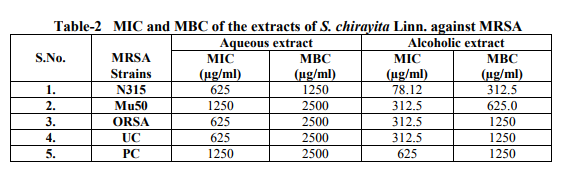
Minimum inhibitory concentration of various extracts against tested microorganisms was determined by broth dilution method [25]. 96- well microtitre plates were used, 50 µl of standardized suspension of a strain (106 cfu/ml) (cfu-colonies forming unit) was added to each tube containing extracts at various concentrations. The plates were incubated at 37oC for 24h and observed for visible growth. The minimum inhibitory concentration (MIC) is taken as the lowest concentration of the extracts at which there is turbidity after incubation, the values are given in Table-2. Minimum Bactericidal Concentration (MBC) was further determined from the same isolates MBC is the minimal concentration of drug needed to kill most (99.9%) of the viable organisms after incubation for 24 hours.
STATISTICAL ANALYSIS OF DATA
All the values have been expressed as Mean ± SEM (Standard error of mean). Statistical significance was determined by one way ANOVA (Analysis of variance) using g-paid software for calculation.
RESULT
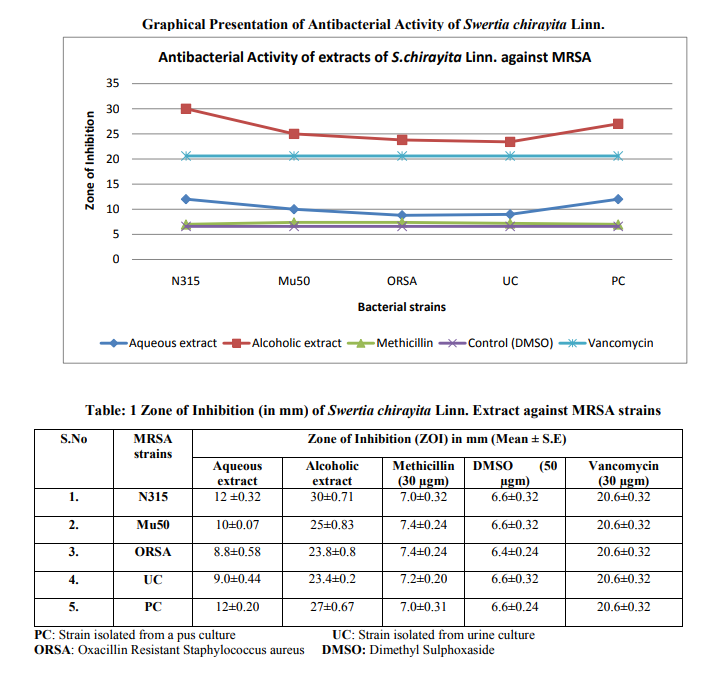
The aqueous and ethanolic extracts of S.chirayita exhibit varying degree of inhibitory effect against all tested pathogenic strains (Table-1). The MIC of crude extracts of the drug was determined at the concentrations ranging from 19.53 to 5000 µgm/ml (Table-2). The aqueous extract produced zone of Inhibition in the range of 8.8 to 12.0 mm for all the tested strains while ethanolic extract produced comparably larger zone against them in the range of 23 to 30 mm, the results were found to be significant with p-value>0.0001. The minimum inhibitory concentration varies in the range of 625 to 1250 µgm/ml for the aqueous extract while it was lesser towards ethanolic extract in the range of 78.12 to 312.5 µgm/ml of the S.chirayita extract. Aqueous extract of the test drug sample was found to have lesser activity as compared to ethanolic extract, but both extract showed larger zone as compared to the inhibitory zone of Vancomycin, which is considered as effective drug for MRSA at present. DMSO- the solvent used to dissolve the drug was also tested; it has not shown any inhibitory effect towards the tested bacteria. This confirms that the inhibitory activity of the extract was only due to the drug used and not due to the solvent in which it was dissolved. Further the resistance to Methicillin was maintained on the nutrient plate, as the antibiotic disk does not produce any inhibitory effect, this confirms the resistance of the tested strains.
DISCUSSION
Medicinal plants are naturally gifted with invaluable bioactive compounds which form the backbone of traditional medicines. Many infectious diseases have been known to be treated with herbal remedies throughout the history of mankind [26]. In the present investigation, in vitro antimicrobial efficacy of the crude extracts of S.chirayita was quantitatively assessed on the basis of inhibition zone and minimum inhibitory concentration. The study shows that the crude ethanolic extract of S.chirayita showed more pronounced antimicrobial activity as compared to aqueous extracts, and when efficacy of either extract was compared to Standard drug ?Vancomycin‘ it was found to be quite efficacious showing a greater ZOI. The result of the present investigation suggests that S.chirayita is a potent natural source of biologically active compounds from herbal medicines, which may potentially prove to be efficient natural antimicrobial agents. This antibacterial property of S.chirayita can be attributed to the presence of the biologically active constituents present in it like amarogentin, swerchirin, triterpenoids, xanthones, ophelic acid, gentiopicrin [27]. This study also supports the previous antibacterial activity of the chirayita extract towards gram positive bacterial strains [28] and verifies the claims of traditional medicine for their use in various infectious diseases [29-31]. However exactly which constituent is helping in killing these dreadful bacteria and in which manner, this part of the study is further need to be explored. And more investigations are needed in this direction before its use in clinics, as the present study is just an in-vitro proof about its antibacterial efficacy; many studies regarding its clinical use are still needed to be done.
CONCLUSION
With the emergence and widespread occurrence of multi drug resistant bacteria focus has now been shifted in exploring natural compounds that may combat drug resistance problem. Clearly strategic planning for search on fundamental and safe integration of efficacious medicine into conventional medical practice is needed. On the other hand the drugs used in traditional system of medicine could be systematically explored for novel bioactive compounds which can inhibit drug resistance bacteria or which can enhance activity of antibiotics. Such compounds or herbal medicine may find application in combination with antibiotic for the treatment of bacterial diseases. The study suggests that S.chirayita exhibit antimicrobial properties against MRSA which is an emerging cause of a number of infectious diseases and has developed resistance to the synthetic antibiotics. The potential antimicrobial activity of S.chirayita towards the infectious micro-organism explains the basis for its use in future in combating the disease caused by such dreadful bacteria.
References:
1. Tyagi A, Kapil A, Singh P: Incidence of Methicillin Resistant Stahylococcus aureus (MRSA) in Pus Samples at a Tertiary Care Hospital: AIIMS: New Delhi: JIACM: 2008; 9 (1): 33 - 35
2. en.wikipedia.orgi date of access : 12-12- 2010.
3. Khan R, Islam B, Akram M, Shakil S, Ahmad A, Ali SM, Siddiqui M and Khan AU: Antimicrobial Activity of Five Herbal Extracts Against MultiDrug Resistant (MDR) Strains of Bacteria and Fungus of Clinical Origin: Molecules 2009;14: 586- 597
4. Chahal K. J., Sarin R., and Manvimalwal: Efficacy of clerodendrum inerme l. (garden quinine) against some human pathogenic strains: International Journal of Pharma and Bio Sciences 2010; 1(4): 219 - 223
5. Cos P, Vlietinck AJ, Berghe DV, Maes L: Anti-infective potential of natural products: How to develop a stronger in vitro ?proofof-concept‘: Journal of Ethnopharmacology 2006; 106: 290 - 302
6. Bandow JE, Brotz H, Leichert LIO, Labischinski H and Hecker M: Proteomic approach to understanding antibiotic action: Amicro. Agents. Chemother. 2003; 47: 948 - 955
7. Kumar VR, Kumar S, Shashidhara, Anitha : Modulation of CNS Activity By Traditional Medicines: A Review On Neuro Pharmacognosy: International Journal of Current Research and Review2011; 3 (1):39-48 8. Lewis K, Ausubel FM : Prospects of plant derived antibacterials: Nat. Biotechnol. 2006; 24: 1504-1507
9. Anonymous: The Wealth of India: A dictionary of Indian Raw materials and Industrial Products: CSIR: New Delhi1976; X: 77-81
10. Bhattacharjee SK and De LC: Medicinal Herbs and Flowers: Aavishkar Publishers: Jaipur 2005; 236- 237
11. Chopra RN, Chopra JC, Handa KL and Kapur LD: Indigenous drugs of India: U N Dhur and Sons Pvt. Ltd: Calcutta 1958: 292- 293
12. Nadkarni KM: The Indian Materia Medica: Bombay Prakashans Pvt. Ltd 2000; I: 1184
13. Wallis TE: Textbook of Pharmacognosy: CBS Publishers: Delhi 1985: 317-319
14. Saha, Prosenjit MS, Das A, Das PC and Das S: Evaluation of anticarcinogenous activity of Swertia chirayita-Buch.-Ham., an Indian Medicinal Plant, on DMBA-induced mouse skin carcinogenesis model: Phytotherapy Research.2004; 18 (5):373-378
15. Verma H, Patil PR, Kolhapure RM, Gopalkrishna V : Antiviral activity of the Indian medicinal plant extract, Swertia chirata against herpes simplex viruses: A study by in-vitro and molecular approach: Indian Journal of Medical Microbiology 2008;26 (4): 322-326
16. Zafar I, Lateef M, Khan MN, Jabbar A and Akhtar MS: Antihelminthic activity of Swertia chirata against gastrointestinal nematodes of sheep: Fitoterapia 2006; 77 (6): 463-465
17. Panda, Ashok K, Tripathy PC and Nandy A : Clinical evaluation of Swertia chirata in the treatment of P.vivax malaria: Aryavaidyan 2005;19 (1): 39-42
18. Bhargava S, Rao SP, Bhargava P, Shukla S: Antipyretic potential of Swertia chirata Buch Ham. Root extract: Scientia Pharmaceutica 2009; 77: 617-623\
19. Kumar M.L.R.S., Yadav A.K., Saxena A. and Paul B.N. : Modulation of Interleukin- 1β, Interleukin-4, Interleukin-6, Interleukin- 10, Tumor Necrosis Factor-α and Interferon- γ by aqueous extracts of Swertia chirayita: Indian Journal of Pharmacology 2002; 34: 141-155
20. Kumar MLRS, Paul BN, Asthana R, Saxena A, Mehrotra S and Rajan G : Swertia chirayita Mediated Modulation of Interleukin-1β Interleukin-6, Interleukin-10, Interferon-γ, and Tumour Necrosis Factor-α in Arthritic Mice: Immunopharmacology and Immunotoxicolog 2003; 25 (4): 573-583
21. Bonjar S: Evaluation of Antibacterial Properties of Some Medicinal Plants Used in Iran. :J.Ethnopharmacol 2004; 94: 301-305
22. Anonymous: Quality control methods for medicinal plant materials: Word Health Organization: Geneva 1998: 25-28
23. National Committee for Clinical Laboratory Standards: Methods for Disk Susceptibility Tests for Bacteria that Grow Aerobically: NCCLS Document M2-A7: National Committee for Clinical Laboratory Standards: Wayne, USA: 2000; 7
24. Ananthanarayan R and Paniker‘s: Textbook of Microbiology:8th edition:Publishers University Press: Hyderabad 2009: 618
25. Jennifer MA: Determination of Minimum Inhibitory Concentrations: Journal of Antimicrobial Chemotherapy 2001; 48, (SI): 5 -16
26. Chahal KJ, Sarin R, and Manvimalwal: Efficacy of clerodendrum inerme l. (garden quinine) against some human pathogenic strains: International Journal of Pharma and Bio Sciences 2010; 1(4): 219 - 223
27. Evans WC and Trease: Pharmacognosy: 15th edition: Elsevier publications: Delhi 2009: 417
28. Alam KD, Ali MS, Parvin S, Mahjabeen S, Akbar MA and Ahmed R: In vitro antimicrobial activities of different fractions of Swertia chirata ethanolic extract: Pak. J. Biol. Sci. 2009; 12: 1334-1337
29. Ghani HN: Khazainul advia: Idara Kitabul Shifa: New Delhi 1921; II: 595-596
30. Momin KM: Tohfatul Mominin (Persian) Matba Hasni 1272: 75
31. Nadkarni KM: The Indian Materia Medica: Bombay Prakashans Pvt. Ltd. 2000; I: 1184
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License