IJCRR - 5(11), June, 2013
Pages: 30-42
Date of Publication: 18-Jun-2013
Print Article
Download XML Download PDF
SIDEROPHORE PRODUCTION FROM AZOTOBACTER SP. AND ITS APPLICATION AS BIOCONTROL AGENT
Author: I. Muthuselvan, R. Balagurunathan
Category: General Sciences
Abstract:Screening and isolation of siderophore producing bacterial strains were carried out from soil samples collected from plant rhizosphere region. The isolate showed positive for siderophore on Chrome azurol sulfonate agar medium was selected for further studies. The organism was subjected to various biochemical tests and 16S rRNA analysis, which lead to its identification as Azotobacter sp. The Azotobacter sp. holds optimal growth temperature and pH at 30ºC and 8.5 respectively. It was also optimized for carbon and nitrogen with optimal growth in Malic acid and peptone respectively. The presence of magnesium and zinc with iron concentration of 4m concentration also favored the siderophore production. Siderophore production was carried out using optimized media and the harvested siderophore was partially purified by ion exchange chromatography. The antagonist activity of the siderophore producing Azotobacter sp. and partially purified siderophore were tested against fungal pathogens such as Fusariurm sp., Alternaria sp., Phytophthora sp., Rhizoctonia sp., Colletotrichum sp. and Curvularia sp. This result showed that the Azotobacter sp. under study is a good producer of siderophore, which can be beneficial for its antagonistic activity towards fungal pathogens.
Keywords: Chrome azurol sulfonate agar medium, 16S rRNA analysis, Amberlite IR120, Antagonism.
Full Text:
INTRODUCTION
Iron is essential for the growth of almost of the microorganisms. Although iron is abundant in nature, it is not readily available to microorganisms because of its extreme insolubility in aerobic water and soil environments (Neilands, 1972). To sequester iron from the environment, microorganisms excrete high-affinity ferric chelating compounds, termed as siderophores, which solubilize and transport the metal (Neilands, 1974). Nitrogenase and other proteins involved in nitrogen fixation require a high complement of iron (Brill, 1980). Therefore, it is likely that nitrogen fixing organisms evolved efficient ironacquisition mechanisms. Iron-deficient nitrogenfixing cultures of Azotobacter spp. produce 2,3- dihydroxybenzoic acid (DHB), 2-N,6-N-di-(2,3- dihydroxybenzoyl)-L-lysine (DHBL), and a yellow-green fluorescent peptide (YGFP) (Carbin and Bulen, 1969). Although,VB the chemical nature of these molecules and their physiological response to iron make them strong candidates for siderophores (Neilands, 1977). The alternative strategies for disease management include the use of bacteria that show beneficial effects on plants and these bacteria are known as Plant Growth Promoting Rhizobacteria (PGPR). The positive effects of PGPR are normally divided into two categories: growth promotion and biological control (Kleopper, 1997). Also, certain root colonizing bacteria can protect plants from soil-borne pathogens when used as inoculants (Slininger et al., 1996). Azotobacter strain was one of the most active PGPR and dominant bacteria in the rhizosphere and had been intensively used as nitrogen fixers and bio-control agents. Bio-control by using antagonistic microorganisms is a potential alternative to chemical compounds for crop protection against phytopathogens (ElKatatany et al., 2003). The mechanisms through which Azotobacter spp. control plant diseases involve competition for niches and nutrients antibiosis, predation, and induction of plant defense responses. In past years, Azotobacter sp. had drawn a worldwide attention because of production of secondary metabolites such as siderophore, antibiotics, enzymes and phytohormones and involving in nitrogen fixation. It has been observed that the antagonistic activity also conferred through the action of siderophores produced from Azotobacter spp. called Azotobactin. The Azotobactin possesses three different types of coordinating moieties, a hydroxamate, an R-hydroxy acid, and a catechol, making it a representative of several classes of siderophores simultaneously. The known interference of pyoverdine-mediated uptake of FeIII provides indirect evidence that azotobactin shows antibacterial effect against root colonizing bacterial and fungal pathogens (Schalk, 2008). Azotobactin is thus a good model compound to test whether the results obtained on the uptake of iron and can be extended as bio-control agent for the plant. The azotobactin seems to be released only under severe Fe limitation (Duhme et al., 1998). Therefore azotobactin is sometimes considered the “true” siderophore of this bacterium (Page et al., 1991). These had been implicated in reduction of plant pathogenic fungi and harmful rhizobacteria (Gupta et al., 2001). Azotobacter sp. by itself has utility as an extremely economical and eco-friendly biopesticide (Rachin and Ahmed, 2005). This work interested with the molecular identification through 16S rDNA sequencing of Azotobacter sp. isolated from soil, production of siderophore, process optimization (pH, cell mass, iron, sugars, organic acids, nitrogen sources, and metals), partial purification of siderophore and application study of siderophore, antagonistic activity towards plant fungal pathogens.
MATERIAL AND METHODS
Isolation of Azotobacter sp. The rhizosphere soil samples from various plant roots were collected from different locations of cultivable lands around Chennai. The enrichment of the culture was made by inoculating one gram of sail sample into sterile Ashby’s mannitol broth (G/L: Mannitol – 20, K2HPO4 – 0.2, MgSO4 - 0.2, NaCl - 0.2, K2SO4, CaCO3 - 15, pH-7.4) and incubated at room temperature for three days for the formation of pellicle. The pellicle was collected aseptically and inoculated into same fresh broth incubated further in the same condition. This method was repeated thrice for the enrichment. After enrichment, the pellicle was streaked into sterile Ashby’s mannitol agar medium and incubated at 28°C for more than 48 hours. Screening for Siderophore production In order to screen the production of siderophore, the isolates were grown in Chrome azurol sulfonate agar medium. The inoculated agar plates were incubated at 37°C for 24 hours. The observation was made for the change of medium color from blue to reddish yellow to determine the siderophore production. The isolate which showed high intensity of color change was considered for further study.
Identification of the bacterial isolate
The selected bacterial isolate was subjected to identification by conventional methods such as Gram’s staining to check the Morphology and Gram’s reaction. The motility was checked using Hanging drop method. The further identification was done using routine biochemical tests based on the key provided by the Bergey’s manual of Systematic bacteriology. The molecular identification of the positive isolate was carried out through 16S ribosomal coding gene sequencing. DNA extraction was carried out by alkaline lysis and phenol chloroform extraction method. The PCR amplification of 16S ribosomal RNA coding sequence using universal primers and the reaction conditions as follows: 94°C (1 min) for denaturation, 51°C (30 sec) for annealing and 72°C (1 min) for extension. The amplified products were purified and cycle sequenced using Big Dye terminator cycle sequencing kit. The sequence was determined using ABI PRISM sequence analyzer. The obtained sequences were subjected to BLAST (Basic Local Alignment Research tool) analysis with online tool with the preexisting sequence available in NCBI/Genbank to confirm the sequences. The obtained 16S ribosomal RNA sequences of strain was aligned with various Azotobacter spp. obtained from GenBank, NCBI using Clustal-X Ver.2.0 aligning tool. The phylogenetic analysis was carried out using MEGA (Molecular Evolutionary Genetic Analysis) Ver.4.0 (Nei and Kumar, 2000). Siderophore production The culture inoculum was prepared using Luria Bertani broth contains 1% Peptone, 1% Sodium chloride and 3% Yeast extract. The isolate was inoculated into sterile LB broth and incubated at 37°C for overnight and 150 rpm agitation. The Siderophore production was carried out using Fedeficient mannitol medium containing g L-1 : K2HPO4, 6.0; KH2PO4, 3.0; MgSO4 0.2; NH4SO4, 1.0 and succinic acid, 4.016; pH-7.0 was inoculated with 1% of overnight inoculums and incubated at 30°C for 24-30 hours with constant shaking at 120 rpm. The culture broth was centrifuged at 10,000 rpm for 10 min and the cell free supernatant was subjected to siderophore assay. Partial purification Partial purification of siderophore was carried out by ion exchange chromatography using amberlite IR120 (Mayer et al., 1998). After the production of siderophore, the bacterial cells were harvested by centrifugation at 10000 rpm for 5 min and the supernatant was collected. The supernatant was considered as crude siderophore extract and pH was adjusted to pH 6.0. The crude extract was passed through an ion exchange chromatographic column containing Amberlite IR120 (Na+). The siderophore was later eluted with methanol (60%). The obtained partially purified siderophore was proceeded with further studies.
Siderophore assay
Siderophore assay was carried out based on the CAS shuttle assay of Payne (1994). The culture extract (0.5 ml) was mixed with CAS reagent and 0.5ml of chrome azurol S reagent. The color obtained was measured using the spectrophotometer at 630 nm after 20 minutes of incubation. The blank was prepared using uninoculated broth medium by carrying same procedure. The siderophore content in the aliquot was calculated by using following formula:

Optimization of Physico-Chemical parameters for Siderophore Production
Siderophore production as a function of time
The siderophore production was carried out by submerged shake flask fermentation in using mannitol medium at 35°C, 100 rpm agitation. Culture was aseptically with drowned periodically at every 6 hours intervals up to 48 hours. The growth rate of the culture was estimated by spectrophotometrically at 360nm and siderophore content of the cells free culture filtrate at different intervals was estimated.
Effect of pH
The mannitol medium was prepared with different pH values ranging from of 4 to 10 in separate flasks. Each flask was seeded with 3% overnight inoculum of Azotobacter sp. and incubated at 35°C for 30 hours under shaking at 120 rpm. After incubation the growth and siderophore production was measured at 410 and 630 nm respectively.
Effect of sugars and organic acids The sterile mannitol basal medium was prepared without carbohydrate source and aliquoted into different conical flasks. Each flask were amended with different carbon sources like glucose, sucrose, dextrose and lactose, different organic acid like oxalic acid, malic acid and lactic acid, at the concentration of 0.1%. The flasks were inoculated with 3% of overnight inoculum and incubated at 30°C for 36 hours under shaking at 100 rpm. After incubation the growth and siderophore production was estimated. Effect of nitrogen sources The sterile basal nitrogen source free mannitol medium was prepared and amended with different concentrations of organic nitrogen sources like peptone and yeast extract, inorganic nitrogen source such as urea, ammonium nitrate and ammonium chloride added at the concentration range of 0.1%. The medium inoculated with 3% of Azotobacter sp. overnight inoculum and incubated at 35°C for 30 hours under shaking at 120 rpm. After incubation the siderophore production was estimated.
Effect of iron concentration
The mannitol medium was prepared with different iron concentration (FeCl3.6H20) in the range of 1- 100 μm. The medium inoculated with Azotobacter sp. and incubated at 30°C for 36 hours under shaking at 100 rpm. After incubation the growth and siderophore production was estimated. Effect of metal ions The sterile mannitol medium was prepared in several flasks and amended with 100μM /L concentration of different metal ions like Na2+, K+, Ca2+,Cu2+, Mg2+, Mn2+, and Zn2+. The flasks were inoculated and incubated at 35°C for 36 hours under shaking at 100 rpm. After incubation, siderophore production was estimated.
Antagonistic activity of Azotobacter sp. Dual culture technique
The antagonistic properties of Azotobacter sp. were tested against plant fungal pathogens such as Fusariurm sp., Alternaria sp., Phytophthora sp., Rhizoctonia sp., Colletotrichum sp. and Curvularia sp. by dual culture technique (Rabindran and Vidhyasekaran, 1996). Azotobacter sp. was streaked on Potato Dextrose Agar (Potato infusion-4 g L-l ; Dextrose-10g L-l ; Agar-18g L-l ) at one side of petri dish, approximately 1 cm away from the edge. Five mm of fungal mycelial agar disc of actively growing 8- 15 day-old-culture from PDA was taken and placed at the opposite side of petri dishes perpendicular to the bacterial streak. Petri dishes were then incubated at room temperature for 4 days. Petri dishes inoculated with fungal discs alone were served as control. The experiment was done in triplicates. Observations on width of inhibition zone and mycelial growth of test pathogens were recorded and percent inhibition of pathogen growth was calculated. Where, I=Inhibition of Mycelial growth, C=Growth of plant pathogen in the control plate (cm) and T=Growth of pathogen in dual cultures (cm).
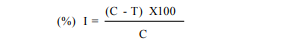
Bio control assay of partial purified siderophore
Sterile potato Dextrose Agar was prepared and solidified in petri dishes. The well size 6mm diameter was made aseptically in the agar plate. Forty µl of partially purified siderophore was added to the well and allowed to diffuse radially for 1 hour at low temperature. The inoculum size of 6x103 spores of plant pathogenic fungi was swabbed over the surface of the PDA plates and incubated at room temperature for 48 hours. After incubation, the zone of inhibition of the mycelia growth was measured.
RESULTS AND DISCUSSION
There were 26 bacterial isolates that produced siderophore were isolated and siderophore production was confirmed by the color change from blue to reddish yellow (Milagres et al., 1999). In order to obtain nitrogen fixer among the siderophore producers, The isolates were short listed by inoculating the isolates using Derx’s medium. Among the short listed isolates which showed double benefit organisms that producing siderophore and nitrogen fixing capcity, potent siderophore producer was used used for futher study. The siderophore production is quiet common phenomenon exhibited by various organisms like Pseudomonas spp., Bacillus spp.,, Clinical isolates like E. coli, Klebsiella etc. (Syed and Vidhale, 2011) for the acquisition of iron complex from the soil. The production of antagonisms against fungal pathogen through siderophore production by nitrogen fixing organisms giving dual benefit for the plants (Frank et al., 1983).
Identification of Bacteria
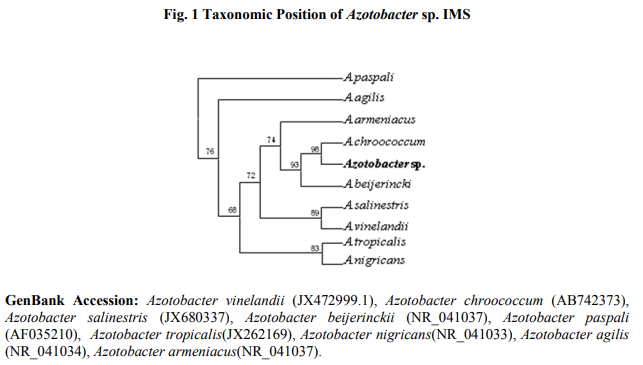
The potent isolate was Gram negative short oval shape rod in nature and motile. The biochemical test showed that the isolate could be considered as Azotobactersp. (Table 1). For the confirmation of identification, molecular identification was carried out 16S ribosomal coding gene identification. The amplified gene product showed 1 Kb in size in agarose gel electrophoresis whereas showed 995 nucleotide base pair in size after sequencing. The Blast result revealed that obtained nucleotide base pairs showed 100% identify with Azotobacter chroococcum (Accession AB742373) 16S ribosomal genes sequence (Table 2). Furthermore, the sequence was aligned and phylogenetic tree was constructed using related sequences. The sequence paired with Azotobacter chroococcum of Indian isolate with bootstrap value of 99% (Fig. 1). So the isolate was confirmed as Azotobacter chroococcum and designated as IMS. Widely, the biochemical test of identification is found to be preliminary identification in which the genus will be identified and species can be identified certain extent. Whereas the molecular identifications through 16S ribosomal RNA coding sequence analysis is widely accepted method among the modern techniques for prokaryotes. The prokaryotic 16rRNA sequence consisted of maximum of 1.6 kb in size and unique sequence distributed among the prokaryotes facilitates to identify the species. The complete sequence of 16S rRNA comprises of approximately 1.5 kb has been so far reported (Gimmestad et al., 2006).
Growth rate and siderophore prodution
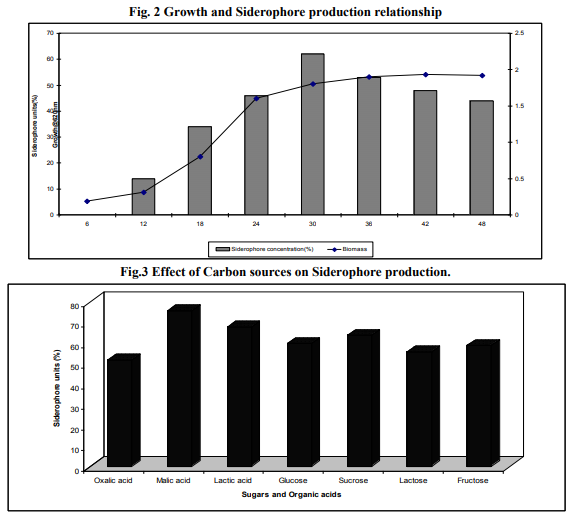
The organism did not exhibit any traces of siderophore synthesis during beginning hours of incubation whereas the bud in the vegetative growth was observed. The strain started siderophore production after 10 hours of inoculation (Fig. 2). The maximum of siderophore production was observed (65% of siderophore units) at 30 hours of incubation and started decreasing beyond 42 hours of incubation. The highest production of siderophores occurred in Nitrogen fixing Azotobacter sp. at a period of 18 hours coinciding with the stationary growth phase (Frank et al., 1983).
Production optimization
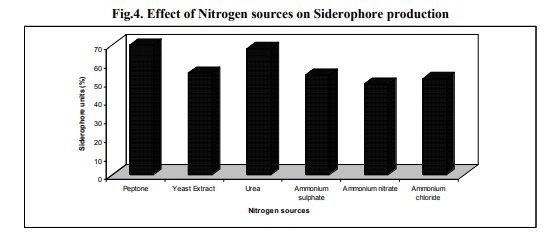
Of the different level of pH tested, production of siderophore by Azotobacter sp. was optimum at pH 7.0 with the yield of 62. In the pH values below and above the neutral the production was also somewhat significant (Table.3). The most of the previous reports of Azotobacter sp. have been stated that the siderophore has been produced in large quantity at neutral pH (Frank et al., 1983). The other nitrogen fixing bacteria Rhizobium strains isolated from root nodules of Sesbania sesban (L.) Merr. produced siderophore in culture after 4 h of incubation and at neutral pH (Sridevi and Malaliah, 2007) Since the production of siderophore depended largely on the pH of the medium. It was very important to maintain the pH of the fermentation broth between 7.0-7.5, which is the optimum for siderophore production in Azotobacter sp. The pH of the medium increased from 7 to 8.5 during the growth period in accordance with the siderophore concentration which suggest that alkalinity is important to avoid siderophore destruction - The mannitol medium with different organic acids oxalic acid, malic acid and lactic acid, at the concentration of 4.0 g L-1 showed considerable amount of siderophore production by Azotobacter sp. in which the quantity was high in malic acid with the yield of 71% (Fig. 3). The presence of sugars such as sucrose increased the growth of the organism but not siderophore production. Similar results were obtained in Azotobacter sp. (Anne et al., 2000) and in Pseudomonas fluorescens, Pseudomonas putida and Alcaligens faecalis (Sayyed et al., 2005). In contrast Nocardia levis MK-VL_113 utilized sucrose and tryptone as good carbon and nitrogen sources for the elaboration of bioactive metabolites (Kavitha and Vijayalakshmi, 2009). Mannitol and sucrose at the concentration 2% stimulated growth and siderophore production in Rhizobium strains (Sridevi and Malaliah, 2007). Nitrogen sources such as peptone, yeast extract, urea, ammonium nitrate and ammonium chloride was added to the mannitol medium at the concentration of 1 g L-1. Among these, the organic nitrogen peptone and urea enhanced the siderophore of Azotobacter sp. with the yield of maximum 70% respectively. In other nitrogen sources siderophore production was almost same as control (Fig. 4). Some of the amino acids as nitrogen sources tested stimulated the siderophorogenesis in Azotobacter sp. (Anne et al., 2000). Arginine and praline stimulated growth and siderophore production in Rhizobium strains (Sridevi and Malaliah, 2007). The Nocardia levis MK-VL113 utilized tryptone as a good nitrogen source for the elaboration of bioactive metabolites. The presence of urea metabolism is known to increase the pH due to hydrolysis of urea into ammonium ion intracellularly (Sama et al., 2009).
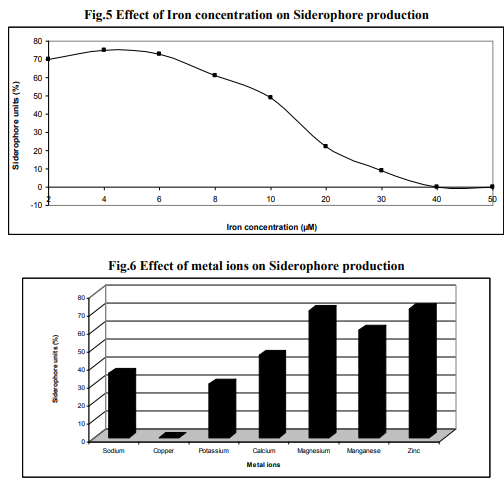
Effect of iron concentration
The iron concentration is an essential parameter which determines the production of siderophore. The mannitol medium was prepared with different iron concentration (FeCl3.6H20) in the range of 1- 100 µm. The presence of low concentration (1- 3µM) of iron enhanced the siderophore production of Azotobacter sp. up to 82%.The iron concentration between 5µM to 30 µM decreased the production and beyond this level the production was inhibited though growth occurred (Fig. 5). Increase in growth with increase in iron concentration reflects the iron requirement for the growth of the organism for cellular process (Syyed, et al., 2005). Siderophores are ironspecific compounds which are secreted under low iron stress and found that production of siderophore in the medium employed was inversely proportional to the iron concentration in the medium (Budzikkiewicz, 1993). The addition of limited iron in the media can increase the growth of the culture, which can lead to an increased production of siderophore in Rhizobium strains (Sridevi and Malaliah, 2007). The increase of Fe (III) concentration had a negative effect in siderophore production, especially above 10 µM (Villigas et al., 2002).
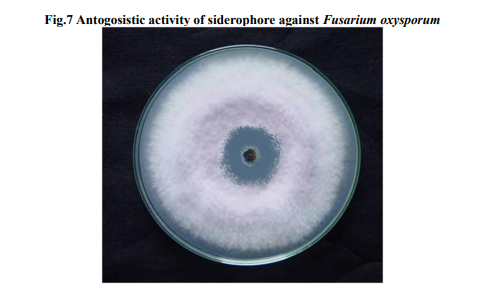
Metal ions
Inorder to find out the effect of other minerals towards siderophore, the production was carried out in mannitol medium amended with different mineral ions. The metal ions Mg2+ and Zn2+in the medium enhanced the siderophore production. The other metal ions tested inhibitory effect on siderophore production (Fig.6). The siderophore and cofactors produced by Azotobacter sp. is an iron chelating substance with the ability to bind to different metal ions was increased, Molybdenum and Cadmium (Neilands, 1974). Whereas, synthesis of metabolites azotichelin and aminochelin was inhibited by Mn2+ and Zn2+ in Azotobacter vinelandii (Page, 1995). Lead increased the siderophore production in Pseudomonas fluorescens, Pseudomonas putida while Mn2+, Hg2+ and Co2+ decreased the production (Sayyed et al., 2005).
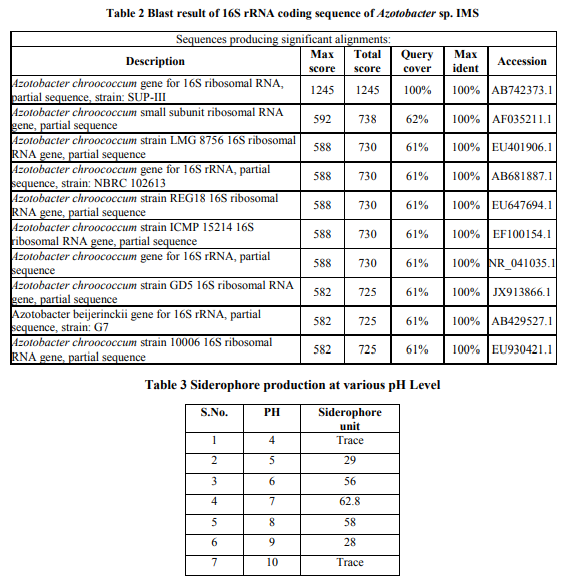
Antagonistic activity
Azotobacter sp. was evaluated for its ability to control plant root fungal pathogens such as Fusariurm sp., Alternaria sp., Phytophthora sp., Rhizoctonia sp., Colletotrichum sp. and Curvularia sp. by dual culture technique and assay of cell free supernatant. It was found that the Azotobacter sp. Lp1 inhibited all the tested pathogens in both the methods of evaluation. The percentage of inhibition was increased from 40% to 50.5% in partially purified siderophore (Fig. 7). This proved the ability of siderophore as biocontrol agent in our study. Azotobacter sp. was found to effectively inhibited 20-40% of the tested fungi in dual culture technique and 25-50% in purified siderophore assay (Table.4). Azotobacter sp. produces azotobactin type siderophore under iron starving conditions with high stability and affinity for iron that restricts growth of microorganism with low iron competition ability such as phytopathogenic fungi (Kloepper, 1977). Purified siderophore of A. calcoaceticus at 500 µg/mL concentrations inhibited the growth of phyto-pathogens up to 30.00%, suggested that both siderophore rich supernatant as well as pure siderophore has the inhibitory potential against phyto-pathogenic fungi (Prashanth et al., 2009). The isolates of Azotobacter spp. were found to effectively inhibit the mycelia growth of both fungal pathogen in dual cultures with rhizospheric bacteria and soil borne pathogens (Sapna et al., 2012). Siderophore mediated antagonism was observed against common phytopathogens viz., A. flavus, A. niger, C. capsicum and F. oxysporum by A. calcoaceticus isolated from wheat rhizosphere (Sapna et al., 2012). CONCLUSION The production of microbial metabolites and their applications in various fields were gaining attention. Siderophores facilitates the bacteria to take up iron under iron-limited environment. In the present study, siderophore producing bacteria was isolated and the production conditions were optimized for their better production. A further study in this field can bring about their applications in various fields.
References:
1. Gimmestad M, Magnus Steigedal, Helga Ertesvåg, Soledad Moreno, Bjørn Erik Christensen, Guadalupe Espín, and Svein Valla. Identification and Characterization of an Azotobacter vinelandii Type I Secretion System Responsible for Export of the AlgEType Mannuronan C-5-Epimerases.J Bacteriol 2006; 188(15): 5551-5560.
2. Neilands JB. Evolution of biological iron binding centers. Struct Bonding 1972; 11: 145-170.
3. Neilands JB. Iron and its role in microbial physiology. In: Neilands JB, editor. Microbial iron metabolism: a comprehensive treatise. New York: Academic Press; 1974. P. 3-34.
4. Brill WJ. Biochemical genetics of nitrogen fixation. Microbiol Rev 1980; 44: 449-467.
5. Corbin JL and WA Bulen WA. The isolation and identification of 2,3-dihydroxybenzoic acid and 2-N,6-Ndi (2,3-dihydroxybenzoyl)- L-lysine formed by iron-deficient Azotobacter vinelandii. Biochem 1969; 8: 757-762.
6. Neilands JB. Siderophores: biochemical ecology and mechanisms of iron transport in enterobacteria. In: Raymond KN, editor. Advances in chemistry, no.162, bioinorganic chemistry II. Washington DC: American Chemical Society; 1977. P. 3-32.
7. Kloepper JW. Current status and future trends in bio-control research and development in the U.S. In: International Symposium on Clean Agriculture, Sapporo, OECD: 49-52
8. Slininger PJ, Van Cauwenberge JE, Bothast RJ, Weller DM, Thomashow LS,Cook RJ. Effect of growth culture physiological state, metabolites and formulation on the viability, phytotoxicity and efficacy of the take-all biocontrol agent Pseudomonas fluorescens 2-79 stored encapsulated on wheat seeds. Appl Microbiol Biotechnol 1996; 45: 391-398
9. El-Katatany M, Hetta A, Sliaban G, ElKomy H. Improvement of cell wall degrading enzymes production by alginate encapsulated Trichoderma spp. Food Technol Biotechnol 2003; 41: 219-225.
10. Gupta C, Dubey R, Kang S, Maheshwari D. Antibiosis-mediatednecrotrophic effect of Pseudomonas GRC2 against two fungal plant pathogens.Curr Sci 2001; 81: 91-94.
11. Rachid D, Ahmed B. Effect of iron and growth inhibitors on siderophores production by Pseudomonas fluorescens. Afr J Biotechnol 2005; 4: 697-702.
12. Schalk IJ. Metal trafficking via siderophores in Gram-negative bacteria: specificities and characteristics of the pyoverdine pathway. J Inorg Biochem 2008; 102: 1159-1169.
13. Duhme AK, Hider RC, Naldrett MJ, Pau RN. The stability of the molybdenum-azotochelin complex and its effect on siderophore production in Azotobacter vinelandii. J Biol Inorg Chem 1998; 3: 520-526.
14. Page WJ, Collinson SK, Demange P, Dell A, Abdallah MA. Azotobacter vinelandii strains of disparate origin produce azotobactin siderophores with identical structures. Biometals 1991; 4: 217-222.
15. Nei M and Kumar S. 2000. Molecular Evolution and Phylogenetics. Oxford University Press, New York.
16. Meyer JM, Stintzi A, Coulanges V, Shivaji S, Voss JA, Taraz K, Budzikiewicz H. Siderotyping of the fluorescent pseudomonads: Pseudomonas fluorescens and Pseudomonas putida strains from Antartica. Microbiol 1998; 144: 3119-3126.
17. Payne SM. Detection, isolation and chacterization of siderophores. MethodsEnzymol 1994; 235: 329.
18. Rabindran R and Vidhyasekaran P. Development of a formulation of Pseudomonas fluorescens PfALR2 for management of rice sheath blight. Crop Protection 1996; 15: 715-721.
19. Milagres AMF, Machuca, A, Nepoleo. Ddetection of siderophore producing fungi and bacteria by modified CAS agar assay. Microbial methods 1999; 37: 1-6
20. Syed Sajeed Ali and Vidhale NN. Evaluation of siderophore produced by different clinical isolate Pseudomonas aeruginosa. Inter J Microbiol Res 2011; 3(3): 131-135. 21. Frank AF, Jack TS, Thomas Emery. Siderophores Produced by Continuous Culture vinelandii OP in Iron-Limited Nitrogen-Fixing Azotobacter. Appl Environ Microbiol 1983; 46(6): 1297-1300.
22. Sridevi M and Mallaiah KV. Production of catechol-type of siderophores by Rhizobium strains from Sesbania sesban (L.) Merr. Asian J Biol Sci 2007; 3(1): 187-194.
23. Anne ET, Manisha Mehrotra, Derek Ottem, William JP. Dual regulation of catecholate siderophore biosynthesis in Azotobacter vinelandii by iron and oxidative stress. Microbiol 2000; 146: 1617-1626.
24. Sayyed RZ, Badgujar MD, Sonawane HM, Mhaske MM, Chinchokar SB. Production of Microbial iron chelators (siderophores) by fluorescent Pseudomonads. Ind J Biotechnol 2005; 4: 484-490.
25. Kavitha and Vijayalakshmi M. Cultural parameters affecting the production of bioactive metabolites by Nocardia levis MKVL 113. JAppl Sci Res 2009; 5(12): 2138- 2147.
26. Sarma MVRK, Krishna Saharan, Lalit Kumar, Ashwani Gautam, Avhijeet Kapoor, Nishant. Siderophoregenic Acinetobacter L; calcoaceticus isolated from wheat rhizosphere with strong PGPR activity. Malay J Microbiol 2009; 5(1): 6-12.
27. Budzikkiewicz H. Secondary metabolites from fluorescent Pseudomonas. FEMS Microbiology Reviews 1993;104: 209-228.
28. Villegas M,, Pilar Villa ED, Alina Frías. Evaluation of the siderophores production by Pseudomonas aeruginosa PSS. Rev Latinoam Microbiol 2002; 44(3-4): 112-117.
29. Prashant SD, Rane Makarand R, Chaudhari Bhushan L, Chincholkar Sudhir B. Siderophoregenic Acinetobacter L; isolated from wheat rhizosphere with strong PGPR activity. Malay J Microbiol 2009; 5(1): 6-12
30. Sapna Chauhana, Kunal Wadhwab, Manjula Vasudevaa & Neeru Narulab. Potential of Azotobacter spp. as biocontrol agents against Rhizoctonia solani and Fusarium oxysporum in cotton (Gossypium hirsutum), guar (Cyamopsis tetragonoloba) and tomato (Lycopersicum esculentum). Arch Agro Soil Sci 2012; 58(12): 1365-1385.

|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License