IJCRR - 7(14), July, 2015
Pages: 49-53
Print Article
Download XML Download PDF
IL-3 AND GM-CSF INDUCES INCREASED RECRUITMENT OF LYMPHOCYTES IN PERIPHERAL BLOOD IN ENU INDUCED LEUKEMIA MOUSE MODEL
Author: Ashish Kumar Singha, Bhaskar Bhattacharjee, Debasish Maiti
Category: Healthcare
Abstract:Aim: The enrolment of blood progenitor cells from the bone marrow into peripheral blood or mobilization is required in leukemia following treatment with chemotherapy and/or cytokines. In this experiment, we examined the mobilization of lymphocytes from bone marrow to peripheral blood using interleukin-3 (IL-3) and granulocyte monocyte-colony stimulating factor (GM-CSF) in experimentally induced leukemia model. Methods: In our Laboratory, leukemia is induced in Balb/C mice by challenging with N'-N' Ethylnitrosourea (ENU) at the dose of 80mg/kg body twice in one week interval and confirmed by staining of peripheral blood and bone marrow smear. After confirmation of leukemia induction, animals were received rmIL-3 and rmGM-CSF in combination for consecutive four days. Leishman's staining of peripheral blood smear and isolation of lymphocytes by percoll gradient method used in this study to determine the cell count. Results: Total leukocyte count exhibits 6 folds higher in number in leukemia group compared to control group. After combination of treatment the total leukocyte count was same as leukemic control group until the sacrifice of animals. But the lymphocyte count increased around 2.6 fold after combination of IL-3 and GM-CSF treatment to leukemic mice. Conclusion: This data suggests the mobilization of lymphocytes from bone marrow to peripheral blood may be immunologically beneficial to combat against tumor cells
Keywords: Mobilization, Leukemia, ENU, interleukin-3, Granulocyte monocyte colony, Stimulating factor
Full Text:
INTRODUCTION Conventional cancer treatment strategies include chemotherapy and radiotherapy. The rationale for administration of high-dose chemotherapy and/or radiation to patients with therapy-sensitive tumours is to reduce tumour burden (Kapustay PM. 1997). Carriage of these therapies with respect to higher drug doses and intensified schedule are resulted by organ toxicities (e.g., bone marrow, heart, and lung) and pancytopenia (Walker F et al., 1994). Hematopoietic stem/progenitor cells (HSC) were formed from the bone marrow and transported generally as mature hematopoietic cells into peripheral blood. The alterations of cells transportation in following treatment with chemotherapy and/or cytokines is termed as mobilization (Lemoli RM et al., 2008). Numerous factors affect HSC mobilization: age, type and dose of cytokines used, in the autologous setting, the patient‘s diagnosis, mobilizing chemotherapy regimen, number and type of previous chemotherapy cycles or radiation, and interval from last chemotherapy cycle (SuarezAlvarez B et al., 2012). Leukemia is a type of cancer of the blood or bone marrow featured by an abnormal increase of immature leukocytes and clinically it is called blast cells. Generally, it starts in the bone marrow, the soft tissue inside most bones. All of the primary elements of the blood are derived from multipotent stem cells found primarily in the bone marrow. During the diseased condition, impair the ability of the bone marrow to produce red blood cells and platelets (Mackaness GB. 1964). Lymphocyte which are derived from the bone marrow and become matured in spleen or thymus, participates in immune system. It is to be believed that; different subpopulation of T cells performs different function in cancer. CD8+ T cells mainly provide antitumor activity (Yusuf Net al., 2008; Kmieciak M et. al., 2011). However, in leukemic condition most of the lymphocytes are in defected condition. CD4+ T cells are not involved directly in antitumor activity whereas pure CD8+ T cells alone can have the antitumor activity as well as can help to survive in marrow transplant condition (Palathumpat V et al., 1995). The number of normal CD8+ T cells present in the blood is important and if the number is not sufficient to fight against the cancer cells in leukemia, the condition of the patient will deteriorate. In diseased condition, as many normal activated CD8+ T cells will present in the blood, immune system can protect the disease progression and so mobilization of lymphocytes by some cytokines and growth factors (Fu P et al., 2006) are helpful. IL-3 stimulates the differentiation of multipotent hematopoietic stem cell into lymphoid lineage; IL-3 stimulates proliferation of all cells in the myeloid lineage, in conjunction with other cytokines like GM-CSF). GM-CSF stimulates stem cells to produce granulocytes and monocytes. Monocytes exit the circulation and migrate into tissue, whereupon they mature into macrophages and dendritic cells to fight against infection through inflammatory cascade. The function of IL-3 is quite similar to GM-CSF. Reports suggest that, combination dose of GM-CSF and IL-3 enhances megakaryocyte maturation and platelet count in primates (Stalh CP et al., 1992). It was also suggested that, treatment of G-CSF or GM-CSF with IL-3 exhibits increased rate of neutrophil and monocyte release in peripheral blood from bone marrow (Lord B et al., 1991). Treatment of IL-3 in patient with bone marrow failure, observes significant increase in leukocyte count (Ganser A et. al., 1990). It was reported that, GM-CSF were used as immunotherapeutic agent in alone or in combination with other factors and cytokines (Arellano M et al., 2008) to treat different type of solid cancer. IL-3 alone also can induce the lymphocyte differentiation and proliferation (Leite-de-Moraes MC et al., 2002; Dilloo D et al., 1996).
MATERIALS AND METHODS
Chemicals required: N-ethyl-N’-nitrosourea (ENU) obtained from Sigma Aldrich, USA. RPMI-1640, Hank’s balanced salt solution (HBSS), Antibiotic solution from Himedia, India. Percoll from GE Healthcare, USA. rmIL-3, mrGM-CSF from ImmunoTools GmBH, Germany. Leishman’s stain from LOBA Chemicals. PBS and the other chemicals were obtained from SRL, India.
Maintenance of animals
Male Balb/C mice, 3-4 weeks old, were obtained from National Institute of Nutrition, Hyderabad, India. Mice were housed in a virus-free animal facility for the duration of the experiments as per guidelines of Institutional Animal Ethics Committee, Tripura University (Ethical Clearance Ref. No.: TU/IAEC/2014/VIII/3-2; Dated: 12-09-2014). These mice were kept and maintained specific pathogen free condition with proper humid (60-65%), and temperature 25-28o C in Tripura University Animal House. Food, dietary supplements and water were provided ad libitum.
Numbers of animal used
Four groups (two groups as control and two groups of leukemic mice) were used for entire experiment having six animals in each group. Group I: Normal control introduced with PBS only. Group II: Challenged with ENU for leukemia induction. Group III: Normal mice received only PBS and treated with rmGM-CSF and rmIL-3 in combination starting at 5days before sacrifice at 24 hr. of interval. Group IV: Mice challenged with ENU and after five months confirmed leukemia and treated with rmGM-CSF and rmIL3 in combination starting at 5 days before sacrifice at 24 hr. of interval.
Treatment of animals
Leukemia control mice are produced by treating with Nethyl-N’-nitrosourea (ENU) at 80 mg/Kg body weight at 3-4 weeks old condition twice in one week interval (Chatterjee R et al., 2015; Law S et al., 2001).
Cytokine supplementation
After confirmation of leukemia induction, all the mice from group III and IV were received rmIL-3 and rmGM-CSF in combination for 4 consecutive days(ImmunoTools GmBH, Germany) (Stalh CP et al., 1992, Lord BI et al., 1991).
Sample collection
All the mice were sacrificed as per guidelines of Institutional Animal Ethics Committee. Mice were dissected and peripheral blood was collected using a sterile syringe into a 2 ml micro centrifuge tube containing heparin solution. The spleen was collected in 1X PBS for T cell isolation. The bones from upper and lower legs were dissected and kept it into 1X PBS for bone marrow smear.
Blood smear and bone marrow smear preparation
After five months of first injection of ENU, a drop of peripheral blood were drawn in a clear, fresh and grease free slide for blood film smear. The blood and bone marrow smear were stained with Leishman’s stain for differential analysis of blood cells. Appearance of blast cells confirmed the leukemia induction in animal and was again confirmed by taking bone marrow smear after sacrificing the animal.
Total count
Small amount of blood were used for total count using Neubeur’s hemocytometer chamber. Remaining blood sample were used for analysis using Blood Analyzer (Cellenium 19, Trivitron)
Isolation of splenic lymphocytes
Non-adherent cells from the splenic suspension removed after macrophage isolation was layered at the top of the percoll layer. Percoll gradient were made using 0.25M sucrose solution and iso-osmolar percoll solution, according to the instruction of manufacturer (GE Healthcare USA). After centrifugation of percoll gradients with cell suspension the layer at upper phase contains lymphocyte cell population taken in separate fresh tube (Depamede NS 2010). After isolation small amount of cells were taken in 1.5 ml microfuge tube to check the purity and was confirmed by using anti CD8 primary antibody (SC-18860 FITC, Santacruz Biotech Inc.) followed by FITC conjugated goat antimouse secondary antibody. The isolated cells were 95% pure CD-8+ cell. In brief, cells were fixed with 4% paraformaldehyde and kept in 1% gelatin coated slide. Cells were washed twice with PBST and blocked with 3% BSA. The cells were incubated with anti CD8 antibody conjugated FITC followed by washing twice and count the cells under the fluorescence microscope
Statistical analysis
All the readings were taken 3-5 times repeats of same experiments. Data were expressed as means ± SEM. Data were analyzed Student’s t test for test of significant using GraphPad Prism software. All statistical test were considered as (***p < 0.001, **p < 0.01, *p < 0.05).
RESULTS
Leukemia induction
After 5 months of the ENU challenge, mice showed some secondary infection including hair fall, foot and mouth infection, red colour of lower portion of nose. Two mice out of twelve treated mice were dead within five days of ENU introduction. Peripheral blood smear from tail vein showed appearance of blast cells with Leishman’s stain in ENU chal lenged mice which were absent in control group is confirmedfor leukemia induction. Further it is supported by bone marrow smear of ENU challenged group showed presence of blast cells which is one of the conventional diagnosis processes for the leukemia patient.
Total Count
The other conventional diagnostic process of leukemia is to count the total leukocyte number. In ENU challenged mice the total count increased around six fold compared to control mice (Figure 1). After treatment with IL-3 and GM-CSF in combination for 4 days, the total count of leukocyte taken every 24 hrs after treatment was not changed significantly.
Induction of lymphocyte count in peripheral blood after cytokine treatment:
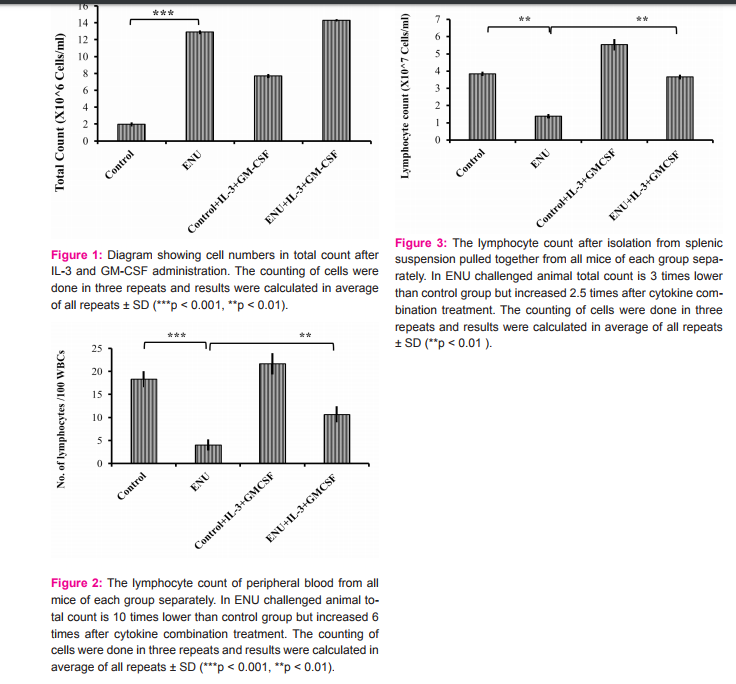
After complete treatment of IL-3 and GM-CSF in ENU challenged group, number of lymphocyte (Figure 2) per 100 leukocytes were increased. Splenic lymphocytes (Figure 3) were isolated in percoll gradient method from all the groups of animals after the experiment was over. The total splenic lymphocyte count was almost four fold decreased in ENU induced leukemia animal which was restored again after treatment with IL-3 and GM-CSF in combination for consecutive four days.
DISCUSSION
Leukemia model is developed in our laboratory with ENU treatment according to the protocol published earlier (Chatterjee R et al., 2015). Blast cell appearance in peripheral blood as well as bone marrow smears confirmed the leukemia disease. The total cell count was increased, which are the symptoms, reminiscent of leukemia disease. But, after treatment of IL-3 and GM-CSF the total count remained same. Facilitating the normal and active cell count in peripheral blood may combat better way against the leukemic cells and after treatment with IL-3 and GM-CSF lymphocytes count was significantly increase compared to untreated leukemia . This might indicates the mobilization of lymphocytes from bone marrow to peripheral blood supported by lymphocyte count in peripheral blood. Cytotoxic T lymphocytes (CTLs) are the antigen specific effector cells of the immune system with the ability to lyse the virus infected cells, abnormal cells or parasites in a contact-dependent manner (Groscurth P et al., 1998). Numerous studies revealed that in solid tumor, the cytotoxic activity of CTL is decreased (Aerts J G et al., 2013, Hariharan K et al., 1995). During the disease condition, the number of functional normal cytotoxic T lymphocytes are (CTL) decreased in lung cancer (Aerts J G et al., 2013). These results also support our data that in diseased condition, the decreased number of CTL might be due to the killing activity of tumor cells and/or due to a hostile environment created by blast cells. It may happen that these cytokines may not activate CTL activity directly. Rather through antigen presenting cells these cytokines activate CTL activity. All of the CTL cells present in peripheral blood in leukemia condition may not function normally due to the hostile environment. Some tropic factors including cytokines like IL-3 and GM-CSF may responsible for differentiation and proliferation of normal hematopoietic stem cells of lymphoid lineage (Hoggatt J et al., 2011). CONCLUSION In animal leukemia model induced by carcinogen ENU, the number of cytotoxic T lymphocytes (CTL) was significantly reduced without affecting the total leukocyte count in the peripheral blood. The number of lymphocyte was again induced significantly towards normal with the treatment of IL-3 and GM-CSF in combination for consecutive 4 days. These experimental results may possibly suggest an immune-therapeutic approach for leukemia treatment.
ACKNOWLEDGEMENT
We are thankful to DBT, Govt. of India for funding as State Biotech Hub for this study. We also gratefully acknowledge to Dr. Manashi Saha (Roy), Dept of Pathology, Agartala Government College and Hospital for confirming the leukaemia. Authors acknowledge the immense help received from the scholars whose articles are cited and included in reference of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature has been received and discussed.
Ethical permission
All the animal experiments were done with proper permission and according to the guidelines of Institutional Animal Ethics Committee (IAEC) of Tripura University.
Conflict of interest
The authors declare that there is no conflict of interest for this study
References:
1. Aerts J G, and. Hegmans J P. Tumor-specific cytotoxic T cells are crucial for efficacy of immunomodulatory antibodies in patients with lung cancer. Cancer Res. 2013; 73(8): 2381-2388.
2. Arellano M, Lonial S. Clinical uses of GM-CSF, a critical appraisal and update. Biologics: Targets and Therapy 2008: 2(1): 13–27.
3. Chatterjee R, Chattopadhyay S, Sanyal S, Daw S, Law S. Pathophysiological Scenario of Hematopoietic Disorders: A Comparative Study of Aplastic Anemia, Myelodysplastic Syndrome and Leukemia in Experimental Animals. Proc Zool Soc. 2015.
4. Depamede NS. Assessment of the purity and characteristics of rat splenic T cells isolated by one-step discontinuous gradient of percoll. JITV 2010; 15(2): 157-164.
5. Dilloo D, Dirksen U, Schneider M, Zessack N, Buttlies B, Levitt L, Burdach S. Differential production of interleukin-3 in human T lymphocytes following either CD3 or CD2 receptor activation. Exp Hematol. 1996; 24(4): 537-43.
6. Fu P, Bagai RK, Meyerson H, Kane D, Fox RM, Creger RJ, et al. Pre-mobilization therapy blood CD34 cell count predicts the likelihood of successful hematopoietic stem cell mobilization. Bone Marrow Transplantation 2006; 38: 189–196.
7. Ganser A, Siepelt G, Lindeman A, Ottmann OG, Falk S, Eder S, et al. Effect of recombinant human interleukin 3 in patients with myelodysplastic syndrome. Blood 1990; 76: 455.
8. Groscurth P, Filgueira L et al. Killing mechanisms of cytotoxic T lymphocytes. News Physiol Sci. 1998; 13: 17-21.
9. Hariharan K, Braslawsky G, Black A, Raychaudhuri S, Hanna N. The induction of cytotoxic T cells and tumor regression by soluble antigen formulation. Cancer Res. 1995; 55: 3486-3489.
10. Hoggatt J, Pelus LM. Mobilization of hematopoietic stem cells from the bone marrow niche to the blood compartment. Stem Cell Research and Therapy 2011; 2:13.
11. Kapustay PM. Blood cell transplantation: concepts and concerns. Semin Oncol Nurs. 1997; 13(3):151-163.
12. Kmieciak M, Worschech A, Nikizad H, et al. CD4+ T cells inhibit the neu-specific CD8+ T-cell exhaustion during the priming phase of immune responses against breast cancer. Breast Cancer Res Treat, 2011. 126(2): 385-94.
13. Law S, Maiti D, Palit A, Majumder D, Basu K, Chaudhuri S, Chaudhuri S. Facilitation of functional compartmentalization of bone marrow cells in leukemic mice by biological response modifiers: an immunotherapeutic approach. Immunology Letters 2001; 76:145–152.
14. Leite-de-Moraes MC, Lisbonne M, Arnould A, Machavoine F, Herbelin A, Dy M, Schneider E. Ligand-activated natural killer T lymphocytes promptly produce IL-3 and GM-CSF in vivo: relevance to peripheral myeloid recruitment. Eur J Immunol. 2002; 32(7): 1897-904.
15. Lemoli RM, D’Addio A, Hematopoietic stem cell mobilization. Haematologica 2008; 93(3): 321-324
16. Lord B, Molineux G, Pojda Z, Souza L, Mermod J, Dexter T et al. Myeloid cell kinetics in mice treated with recombinant interleukin-3, granulocyte colony-stimulating factor (CSF), or granulocyte-macrophage CSF in vivo. Blood 1991; 77: 2154–2159.
17. Mackaness GB. The immunological basis of acquired cellular resistance. J Exp Med 1964; 120: 105–20.
18. Ogiu T, Odashima S. Induction of rat leukemias and thymic lymphoma by N-nitrosourea. Acta Pathol Jpn 1982; 32 Suppl 1: 223- 35.
19. Palathumpat V, Dejbakhsh-Jones S, Strober S. The role of purified CD8+ T cells in graft-versus-leukemia activity and engraftment after allogeneic bone marrow transplantation. Transplantation 1995; 60(4): 355-61.
20. Stahl CP, Winton EF, Monroe MC, Hoff E, Holman RC, Myers L, et al. Differential effects of sequential, simultaneous, and single agent interleukin-3 and granulocyte-macrophage colonystimulating factor on megakaryocyte maturation and platelet response in primates. Blood 1992; 80: 2479-2485.
21. Suárez-Álvarez B, López-Vázquez A, López-Larrea C. Mobilization And Homing Of Hematopoietic Stem Cells. Stem Cell Transplantation, edited by Carlos López-Larrea, Antonio LópezVázquez and Beatriz Suárez-Álvarez. Landes Bioscience / Springer Science+Business Media, LLC dual imprint / Springer series: Advances in Experimental Medicine and Biology 2012; 741.
22. Walker F, Roethke SK, Martin G. An overview of the rationale, process, and nursing implications of peripheral blood stem cell transplantation. Cancer Nurs. 1994; 17(2):141-148.
23. Yusuf N, Nasti TH, Katiyar SK, Jacobs MK, Seibert MD, Ginsburg AC, et al. Antagonistic roles of CD4+ and CD8+ T-cells in 7,12-dimethylbenz(a)anthracene cutaneous carcinogenesis. Cancer Res 2008; 68(10): 3924-30
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License