IJCRR - 8(2), January, 2016
Pages: 23-28
Print Article
Download XML Download PDF
ASSESSMENT OF BIOCHEMICAL RISK FACTORS OF CARDIO METABOLIC SYNDROME IN PATIENTS OF HYPOTHYROIDISM
Author: Meghana K. Padwal, Bapu Kamble, Pinky Sawlani, Rajani R. Melinkeri
Category: Healthcare
Abstract:Introduction: Hypothyroidism is the second most prevalent endocrinal disorder in India. Most of existing data supports that thyroid disease is associated with increased cardiovascular risk which is mainly attributed to hemodynamic alteration as well as to a high risk of atherosclerosis, dylipidemia and insulin resistance. Aim: To investigate relationship between thyroid function and biochemical risk factors of cardiovascular diseases. Material and Methods: The present study was a cross-sectional, prospective. The study population was comprised of a total of ninety (90) participants (30 new cases of overt hypothyroidism, 30 new cases of subclinical hypothyroidism and 30 age and gender matched controls). Estimation of serum Thyroid profile and Insulin (fT3 , fT4 and TSH) was done by CLIA while Glucose and Lipid profile were estimated by biochemistry autoanalyzer. Insulin Resistance was calculated by HOMA IR score. Statistical Analysis: Data obtained was statistically analyzed by using student \"t\" test. Results: We found statistically significant rise in the levels of serum cholesterol triglycerides VLDL and LDL in the cases of overt hypothyroidism than controls. HDL levels are low in overt hypothyroidism. We also found statistically significant rise in the levels of blood glucose, serum insulin and HOMA IR score in cases of overt hypothyroidism than controls. Conclusion: Overt Hypothyroidism is at the risk of developing cardiovascular diseases and type II DM.
Keywords: Hypothyroidism, Cardiovascular diseases, Type II DM
Full Text:
INTRODUCTION
The thyroid gland synthesizes and releases two iodoamino acid hormones: 3,5,3’-triiodothyronine (T3 ) and 3,5,3’,5’ thyroxine (T4 ). Thyroxine (T4 ). More than 99% of the circulating T3 and T4 is protein bound. (1) The biologically active component of T4 and T3 in plasma is the free fraction (fT3 and fT4 respectively); that are not bound to proteins. Assay of free thyroid hormone levels is done routinely to avoid problems in interpretation of thyroid hormone levels caused by fluctuations in binding proteins.(2,3) Thyroid hormones may be considered the accelerator pedal of metabolism.(4,5,6) Thyroid disorders are among the commonest endocrine disorders worldwide. It has been estimated that about 42 million people in India suffer from thyroid diseases.(7) Hypothyroidism is the second most prevalent endocrinal disorder in India. ‘Hypothyroidism’ is the reduced production of thyroid hormone.(8) It is a clinical entity resulting from the deficiency of thyroid hormones or more rarely from its impaired activity at tissue levels.(9) Hypothyroidism is classified as Primary, Secondary and Tertiary by its association with the indicated organ dysfunction. (10) According to symptoms and clinical features; it is further sub classified into:
i. Subclinical Hypothyroidism (SCH): It is defined as biochemical evidence of thyroid hormone deficiency i.e. elevated serum Thyroid Stimulating Hormone (TSH) level with a normal serum free Thyroxine concentration(11) in patients who have few or no apparent clinical features of hypothyroidism. Subclinical hypothyroidism can progress to Overt Hypothyroidism.(12)
ii. Overt Hypothyroidism(OH): It is defined as an elevated serum Thyroid Stimulating Hormone (TSH) concentration and reduced free thyroid hormones with symptoms and clinical features of hypothyroidism. It is also referred as Clinical Hypothyroidism.(13) Most of existing data supports that thyroid disease is also associated with increased cardiovascular risk which is mainly attributed to hemodynamic alteration as well as to a high risk of atherosclerosis. (14-15) Dyslipidemia induces insulin resistance, oxidative stress, via vicious cycle. It has been also observed that Insulin resistance, hypertension, inflammation, oxidative stress and coagulation defect are also promoted by thyroid disease, independently of dyslipidemia.(15,16)
AIM AND OBJECTIVES: Assessment of biochemical risk factors of cardiovascular diseases in patients with hypothyroidism
MATERIAL AND METHODS: The present study was a cross-sectional, prospective study. It was conducted in the department of biochemistry; of university medical college with a tertiary care hospital. The duration was November 2011- June 2013.The study protocol was approved by Institutional Ethics Committee. The study group of a total of ninety (90) participants was divided into three groups:
1. Group A: Control- Euthyroid: Thirty (30) age and gender matched normal; healthy euthyroid individuals.
2. Group B: Subclinical hypothyroidism (SCH): Thirty (30) clinically diagnosed, new cases of subclinical hypothyroidism on the basis of clinical examination and their fT3, fT4 and TSH levels.
3. Group C: Overt hypothyroidism (OH): Thirty (30) clinically diagnosed, new cases of overt hypothyroidism on the basis of clinical examination and their fT3, fT4 and TSH levels.
Exclusion Criteria for cases and controls: Participants suffering from DM, CVDs, Renal, Liver diseases, Malignancy, other systemic disease that alters thyroid status or participants taking thyroid hormone supplementation, β-blockers, Multivitamins, Steroids, alcohol and smokers. All participants after their written informed consent underwent detailed physical and clinical examination.
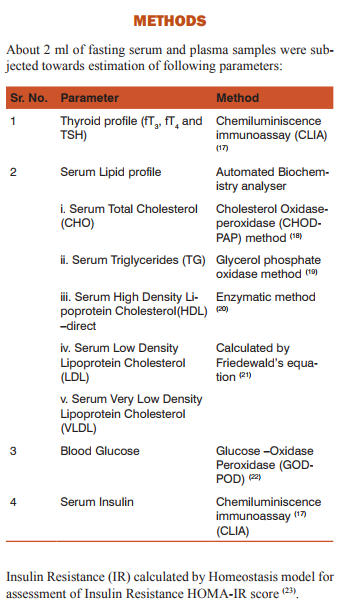
Statistical Analysis: Microsoft Excel 2007 was used to calculate Z Test for finding the statistical significance between the means of Serum lipid profile, Blood glucose, Serum Insulin and HOMA-IR score in all the three groups.
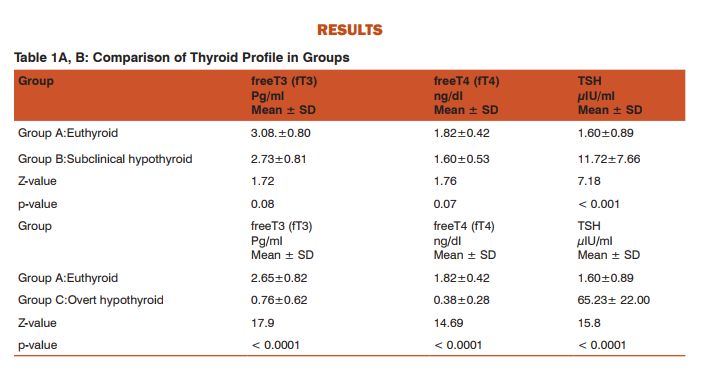

DISCUSSION Table 1 A,B shows thyroid profile in groups. We observed that there was no statistically significant difference in the levels of fT3 and fT4 in subclinical hypothyroid group as compared with euthyroid group. However, the levels of TSH were significantly increased in subclinical hypothyroid group as compared with euthyroid group. Vanderpump MP et al. (1995)(24) and Surks MI et al. (2004)(25) stated that subclinical hypothyroidism is a state of biochemical hypothyroidism rather than clinical hypothyroidism. We noted that the levels of fT3 and fT4 had been significantly decreased while levels of TSH were significantly increased in subclinical and overt hypothyroid group as compared to euthyroid group.
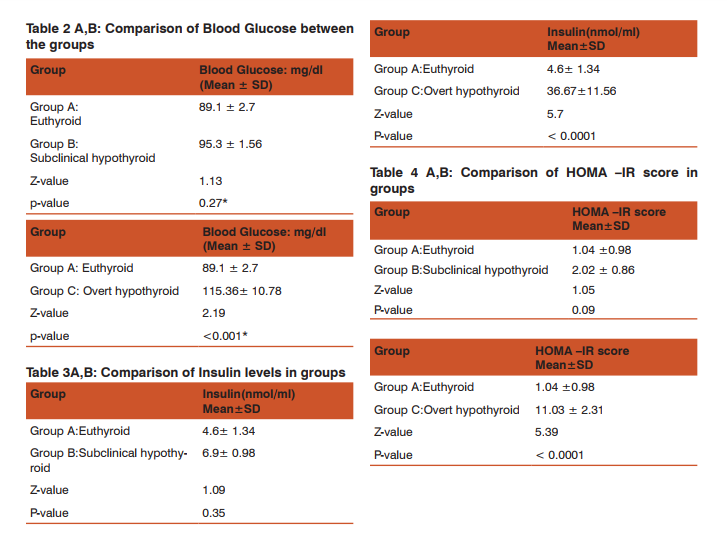
Table 2 A,B shows the levels of Blood Glucose in all the groups. We found statistically highly significantly rise in Blood Glucose level in the overt hypothyroid group (115.36 ± 10.78) as compared with euthyroid (89.1 ±2.7) and subclinical hypothyroid (95.3 ±1.56). Table 3 A,B shows high levels of Serum Insulin in all the groups. We found that statistically significant rise in Serum Insulin overt hypothyroid group (36.67 ± 11.56) as compared with euthyroid (4.60 ±1.34) and subclinical hypothyroid (6.90 ±0.98). Table 4 A,B shows the levels HOMA-IR Score in all the groups. We found statistically highly significant rise in HOMA-IR Score in overt hypothyroid group (11.03 ±2.31) as compared with euthyroid (1.04 ± 0.98) and subclinical hypothyroid (2.02 ±0.86). Table 5 A,B shows the Lipid Profile in groups. We found significant dyslipidemia in overt hypothyroid group.
There was significant increase in Total Cholesterol (211.30 ±40.22), Triglyceride (155.30±96.39) LDL(170.9 ±33.37) and VLDL (31.00±19.22) levels as compared in subclinical hypothyroid group where it was Total Cholesterol (180.06±43.21), Triglycerides (133.64±79.60), LDL (154.22±40.48), VLDL (25.83±16.38) and in euthyroid group it was Total Cholesterol (170.11±40.26), Triglycerides (106.63±42.03), LDL (148.78 ±34.08), VLDL (21.32±8.40). There was also statistically significant decrease in HDL levels in overt hypothyroid group HDL (32.5±8.74) as compared with subclinical hypothyroid group (43.27±12.84) and euthyroid group (46.44 ± 12.11). We calculated the correlation of biochemical risk factors of cardiovascular diseases with TSH levels.
There is statistically significant positive correlation between TSH levels and BGL (r=0.41, p=< 0.0001), Insulin (r=0.50 p=< 0.0001), HOMA- IR (r=0.51 p=< 0.0001),Total Cholesterol (r=0.39, p=0.0001),TG(r=0.20, p=0.04) levels. We also found statistically significant negative correlation between TSH levels and HDL levels. (r= - 0.33, p=< 0.001) However we could not find any significant correlation between TSH levels and LDL levels. (r=0.20, p =0.96). Therefore it can be clearly seen from our results that there is dyslipidemia and insulin resistance associated with overt hypothyroidism, however the same was not observed in subclinical hypothyroid group. The levels of TSH `showed a strong positive correlation with biochemical markers of cardiovascular diseases in our study.
Togini et al (26) observed increase in total cholesterol, LDL, TG levels and decrease in HDL levels in overt hypothyroid group. XuC et al(27) observed that even after adjusting the confounding factors such as age, sex, smoking status, fasting plasma glucose levels and thyroid hormones , a significant positive impact of TSH on the serum total cholesterol (TC) level was revealed ( r = 0.095, p = 0.035 ). Similarly Benetti-Pinto CL et al(28) also observed no change in BGL, Insulin, HOMA IR, cholesterol, TG, HDL levels in sub clinically hypothyroids females. However they reported significantly elevated LDL levels. Garduno-Garcia Jde J et al (29) concluded that though subclinical hypothyroidism was not associated with derangements in the markers of metabolic syndrome (as per National Cholesterol Education Programme ATP III Criteria).
Despite the low thyroid function in hypothyroidism can predispose to cardio- metabolic syndrome. Uzunlula et al (30) and Shantha et al (31) also could not found any significant difference in lipid profile in euthyroid and sub clinical hypothyroid patients. Purvi Purohit (32) noted increase in serum insulin level and HOMA-IR score in hypothyroidism as well as hyperthyroidism. However, increased TSH level were positively correlated with IR. Their study concluded that there was presence of triad of metabolic syndrome in hypothyroid subjects. (i.e hypertension, dyslipidemia increased fasting plasma glucose). Khan et al (33) reported significant correlation TG and HDL level with HOMA-IR values in human subjects with Insulin Resistance syndrome. Chen G et al(34) could not found difference in Glucose, insulin HOMA-IR level in hypothyroid patient compared to euthyroid patients.
Thyroid hormone increases LDL receptor expression, increases Cholesterol Ester Transport Protein (CETP) concentrations and increases hepatic lipase (HL) concentrations. Thyroid hormone also increases hepatic cholesterol synthesis by inducing hydroxyl methylglutaryl coenzyme A (HMG CoA) reductase and decreases intestinal cholesterol absorption(35, 36). Overall effect of this leads to increased cholesterol synthesis and decreased degradation.
It is known that T3 and insulin have synergetic role in Glucose homeostasis, since hormone possess similar action site in regulation of Glucose metabolism at both cellular and molecular level (37,38,39). It could therefore be hypothesized that reduced intracellular content of T3 could lead to impaired insulin stimulated Glucose disposal. Interestingly even subtle decreases in level of thyroid hormone within the pathological range have been shown to correlate inversely with HOMA-index. Insulin resistance and metabolic syndrome are important cardiovascular risk factor, even in non-diabetic individuals (40). It has been found that individuals with Insulin resistance and raised TSH have higher LDL concentration while in those with normal insulin sensitivity, TSH level do not affect LDL (41).
CONCLUSIONS
• Overt hypothyroidism is associated with significant dyslipidemia.
• There is no derangement in this biochemical risk factor of cardiovascular disease in subclinical hypothyroidism.
• Thyroid stimulating hormone (TSH) showed significant positive correlation with total cholesterol, triglyceride, BGL, Insulin and HOMA- IR score and negative correlation with HDL level. Therefore estimation of TSH level can serve as prognostic marker of future cardio-vascular risk.
References:
1. Harper’s Biochemistry (1998): Published by a Lange Medical Book. Thyroid Hormones. Ed. 25th : 561.
2. K Boelaert and J A Franklyn. Thyroid hormone in health and disease. Journal of Endocrinology. 2005;187: 1–15.
3. HARRISON'S PRINCIPLES OF Internal Medicine (1958): Published by McGraw-Hill Companies. 15th Ed. Disorders of Thyroid Gland. Vol. (2):2063.
4. John W Baynes and Marek H Dominiczak (1999): Published by ELSEVIER MOSBY Medical Biochemistry. The hypothalamicpituitary-adrenal axis. Ed.3rd : 535.
5. Nishiki k et al. Evaluation of oxidative phosphorylation in hearts from enthyroid, hypothyroid, and hyperthyroid rats. Am J Physiol. 1978; (235) ;212-219.
6. Pereira B, Rosa LF, Safi DA et al. Control of Superoxide dismutase, Catalase and glutathione peroxidase activities in rat lymphoid organs by thyroid hormones. J Endocrinol. 1994;(140) :73-77.
7. Desai PM. Disorders of the Thyroid Gland in India. Indian J Pediatr. 1997; (64) : 11-20.
8. Krysiak R, Okopien B, Herman ZS. Subclinical thyroid disorders. Pol MerkurLekarski. 2006; (21): 573-8.
9. Williams Textbook of Endocrinology (2008): Published by Saunders Elseiver, Hypothyroidism ; Ed. 11th : 377.
10. Kostoglou-Athanassiou I, Ntalles K. Hypothyroidism – new aspects of an old disease. HIPPOKRATIA. 2010; (14,2) : 82-87.
11. Cooper DS. Subclinical hypothyroidism. N Engl J Med. 2001; (345) :260-265.
12. Huber G, Staub J-J, Meier C, et al. Prospective study of the spontaneous course of subclinical hypothyroidism: Prognostic value of thyrotropin, thyroid reserve and thyroid antibodies. J ClinEndocrinolMetab. 2002; (87): 3221-3226.
13. Hollowel JG, Stehling NW, Flanders D, et al. Serum TSH, T4 and thyroid antibodies in the United states population (1998- 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab. 2002; (887) : 489-499.
14. Klein and K. Ojamaa. Thyroid hormone and the cardiovascular system. The New England Journal of Medicine. (vol. 344, no. 7): 501-509.
15. Fazio, E. A. Palmieri, G. Lombardi, and B. Biondi. “Effects of thyroid hormone on the cardiovascular system,” Recent progress in hormone research.(vol. 59) : 31-50.
16. Bakker SJL, Ter Maaten JC, Popp-Snijders C, Slaets JPJ, Heine RJ, Gans ROB. The relationship between thyrotropin and low density lipoprotein cholesterol is modified by insulin sensitivity in healthy euthyroid subjects. J Clin Endocrinol Metab 2001; 86: 1206-11.
17. A.K. Campbell. Detection and Quantification of chemiluminescence, in Chemiluminescence principles and applications in Biology and medicine.EllisHorwood.1988; 68-126.
18. Nader Rifai and G. Russell Warnick. Lipids, Lipoproteins, apolipoproteins and other cardiovascular risk factors. Teitz Textbook of Clinical Chemistry. 4th Edition. Section IV. Chapter 26: 943- 48.
19. Stein, E. A. and Mayers, G. L. Lipids, Lipoproteines and Apolipoproteines in Tietz Textbook of clinical Chemistry. Burtis C. A. and Ashwood E. R. (Ed). WB Saunders Company, Second Edition. 1994; (23) : 1002-93.
20. Dominiczak M, McNamara J. The system of cardiovascular prevention. 103-125; Nauk M, Warnick G. Measurement of HighDensity-Lipoproteine Cholesterol. In: Handbook of Lipoprotein Testing (eds. Rifai, Warnick and Dominiczak), 2nd edition.
21. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972; (18) : 499–502.
22. Trinder, P. Ann. Clin. Biochem. 1969; (6):24.
23. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fastingplasma glucose and insulin concentrations in man. Diabetologia.1985; (28) : 412-9.
24. Vanderpump MP, Tunbridge WM, French JM, et al. The incidence of thyroid disorders in the community: a twenty year follow-up of the Whickham survey. Clin Endocrinol (oxf). 1995; 43: 55-68.
25. Surks M.I., Ortiz, E., Daniels, G.H., Sawin, C.T., Col, N.F., Cobin, R.H.: Subclinical thyroid disease: Scientific review and guidelines for diagnosis and management. J. A. M. A. 2004; 291(2):228-238.
26. Tognini S, Polini A, Pasqualetti G, Ursino S, Caraccio N, Ferdeghini M, Monzani F.Age and gender substantially influence the relationship between thyroid status and the lipoprotein profile: results from a large cross-sectional study. Thyroid. 2012 Nov;22(11):1096-103.
27. Xu C, Yang X, Liu W, Yuan H, Yu C, Gao L, Zhao J. Thyroid stimulating hormone, independent of thyroid hormone, can elevate the serum total cholesterol level in patients with coronary heart disease: a cross-sectional design. NutrMetab (Lond). 2012 May 23;9(1):44.
28. Benetti-Pinto CL, Berini Piccolo VR, Garmes HM, TeatinJuliato CR.Subclinical hypothyroidism in young women with polycystic ovary syndrome: an analysis of clinical, hormonal, and metabolic parameters. Fertil Steril. 2013 Feb;99(2):588-92.
29. Garduño-Garcia Jde J, Alvirde-Garcia U, López-Carrasco G, Padilla Mendoza ME, Mehta R, Arellano-Campos O, Choza R, Sauque L, Garay-Sevilla ME, Malacara JM, Gomez-Perez FJ, Aguilar-Salinas CA. TSH and free thyroxine concentrations are associated with differing metabolic markers in euthyroid subjects. Eur J Endocrinol. 2010 Aug;163(2):273-8.
30. Uzunlulu M, Yorulmaz E, Oguz A. Prevalence of subclinical hypothyroidism in patients with metabolic syndrome. Endocr J. 2007 Feb;54(1):71-6.
31. Shantha GP, Kumar AA, Jeyachandran V et al. Association between primary hypothyroidism and metabolic syndrome and the role of C reactive protein: a cross- sectional study from South India. Thyroid Res. 2009 Mar 9;2(1):2.
32. Purohit P, Mathur R Hypertension association with serum lipoproteins, insulin, insulin resistance and C-Peptide: unexplored forte of cardiovascular risk in hypothyroidism. N Am J Med Sci. 2013 Mar;5(3):195-201.
33. Khan SH, Khan FA, Ijaz A, Sattar A, Dilawar M, Hashim R. Spectrum of lipid and lipoprotein indices in human subjects with insulin resistance syndrome. J Ayub Med Coll Abbottabad. 2008 Jul-Sep;20(3):17-22.
34. Chen G, Wu J, Lin Y, Huang B, Yao J, Jiang Q, Wen J, Lin L. Associations between cardiovascular risk, insulin resistance, betacell function and thyroid dysfunction: a cross-sectional study in She ethnic minority group of Fujian Province in China. Eur J Endocrinol. 2010: Nov;163(5):775-82.
35. 26. Lam KS, Chan MK, Yeung RT. High-density lipoprotein cholesterol, hepatic lipase and lipoprotein lipase activities in thyroid dysfunction—effects of treatment. 1986. Q J Med. 59:513–521.
36. Kuusi T, Taskinen MR, Nikkilä EA. Lipoproteins, lipolytic enzymes and hormonal status in hypothyroid women at different levels of substitution. J Clin Endocrinol Metab. 1988. 66:51–56.
37. Bakker SJ, terMaaten JC, Popp-Snijders C, Slaets JP, Heine RJ, Gans RO. The relationship between thyrotropin and low density lipoprotein cholesterol is modified by insulin sensitivity in healthy euthyroid subjects. J Clin Endocrinol Metab 2001;86:1206-1211.
38. Kim SR, Tull ES, Talbott EO, Vogt MT and Kuller LH. A hypothesis of synergism: the interrelationship of T3 and insulin to disturbances in metabolic homeostasis. Medical Hypotheses. 2002: 59; 660–666.
39. Hanley AJ, Williams K, Stern MP, Haffner SM. Homeostasis model assessment of insulin resistance in relation to the incidence of cardiovascular disease: the San Antonio Heart Study. Diabetes Care 2002;25:1177-1184.
40. Miura S, Iitaka M, Suzuki S, Fukasawa N, Kitahama S, Kawakami Y, Sakatsume Y, Yamanaka K, Kawasaki S, Kinoshita S, Katayama S, Shibosawa T, Ishii J. Decrease in serum levels of thyroid hormone in patients with coronary heart disease. Endocr J. 1996 Dec;43(6):657-63.
41. Dimitriadis G, Mitrou P, Lambadiari V, Boutati E, Maratou E, Panagiotakos DB, Koukkou E, Tzanela M, Thalassinos N and Raptis SA. Insulin action in adipose tissue and muscle in hypothyroidism. Journal of Clinical Endocrinology and Metabolism. 2006. 91; 4930–4937.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License