IJCRR - 10(10), May, 2018
Pages: 01-09
Date of Publication: 30-May-2018
Print Article
Download XML Download PDF
Body Mass Index, Blood Pressure and Lipid profile in type 2 diabetes-Review
Author: Chitra R. Hinge, Sachin B. Ingle, Baban D. Adgaonkar
Category: Healthcare
Abstract:Diabetes mellitus type II (formerly noninsulin-dependent diabetes mellitus (NIDDM) or adult-onset diabetes) is a metabolic disorder that is characterized by hyperglycemia (high blood sugar) in the context of insulin resistance and relative lack of insulin. Obesity is thought to be the primary cause of type 2 diabetes in people who are genetically predisposed to the disease (although this is not the case in people of East-Asian ancestry).
Keywords: Type 2 diabetes, Obesity, Anthropometry, Lipid profile
Full Text:
INTRODUCTION
Diabetes mellitus (DM) is a syndrome of impaired carbohydrate, fat and protein metabolism caused by either lack of insulin secretion or decreased sensitivity of the tissues to insulin. Type I DM is also called as insulin dependent diabetes mellitus (IDDM) is caused by lack of insulin secretion.
Type II DM is also called as non insulin dependent diabetes mellitus (NIDDM) is initially caused decreased sensitivity of target tissues to metabolic effects of insulin. This reduced insulin sensitivity of insulin is often called as Insulin resistance1. Type II DM is the most common type of diabetes and usually associated with obesity. It usually develops after the age 402.
The incidence of type II diabetes mellitus (Type II DM) is rapidly increasing worldwide and it constitutes a major health problem in both developed and developing countries3. There is a great deal of evidence that both genetic and environmental factors are of importance in the pathogenesis of Type II DM. Obesity, in particular the central obesity, physical inactivity, and a diet rich in saturated fatty acids increases the risk of type II DM4.
Cardiovascular disease (CVD) is a major cause of morbidity and mortality in patients with type II DM having 2 to 4 times higher risk of developing CVD when compared to non diabetics. In diabetics cardiovascular complication occurs at an earlier age and often results in premature deaths5.
Patients with type II DM are frequently affected by atherosclerotic vascular disease. Multiple factors contribute to this accelerated atherosclerosis in type II DM. These factors include dyslipidemia, obesity, hypertension, and insulin resistance6-9.
Lipid abnormalities are more common in type II DM and are aggravated with poor glycaemic control. The classical dyslipidemia in type II DM is so called atherogenic dyslipidemia. This is a constellation of lipid abnormalities which includes increased serum triglycerides (TG), increased low-density lipoprotein cholesterol (LDL-C), and decreased high-density lipoprotein cholesterol (HDL-C) also known as “lipid triad” 10.
Lipid abnormalities play an important role in the causation of diabetic atherosclerosis. Elevated levels of TG, cholesterol, and LDL-C increases the risk of atherogenesis and high levels of HDL-C in contrast bear an inverse relationship to the risk of atherosclerosis and coronary heart disease (CHD) 11-12
Type II DM is associated with centralized and disharmonious distribution of fat. There is a significant association between regional fat distribution and CVD risk factors. Abdominal or central adiposity is considered the most important determinant of CVD and Type II DM13. Although imaging techniques can accurately determine total body fat and its distribution in human but are not suitable for use in large population studies because of cost, irradiation exposure, and limited availability14.
The use of simple anthropometric measurements seems to diagnose obesity in early stages due to its benefits in routine monitoring and assessment in patients. Some of the simple anthropometric measures used routinely includes Body mass index (BMI), Waist circumference (WC), Waist to hip ratio (WHR) 15‚16.
BMI is widely used for classification of obesity, but it does not account for the variations of fat distributions. Waist circumference is the best simple anthropometric index of abdominal visceral adipose tissue and also the best index for predicting CVD risks17.
In India 50% of diabetics has hypertension (HTN). The frequency of hypertension in diabetic population is almost twice as compared to non-diabetic general population7. In hypertensive patient with DM, atherosclerosis gets accelerated and its consequences get manifested earlier18. Both HTN and type II DM are recognised as independent CVD risk factors19.
These risk factors have a great potential for prevention through modification of life style and dietary changes.
REVIEW OF LITERATURE
Type II DM is due to insulin resistance or reduced insulin sensitivity, combined with relatively reduced insulin secretion. There is a great deal of evidence that both genetic and environmental factors are of importance in the pathogenesis of Type II DM. Whereas the genetic factors are still poorly understood, numerous studies have shown that obesity (in particular, abdominal obesity), physical inactivity, a high-fat diet, and a diet rich in saturated fatty acids increases the risk of diabetes4. It is a chronic disease that requires long-term medical attention both to limit the development of its devastating complications and to manage them when they do occur.
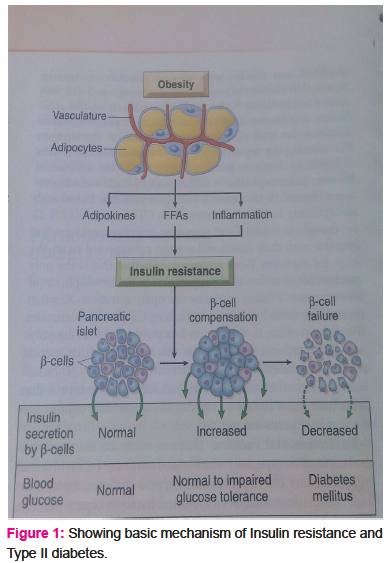
Insulin resistance associated with obesity is induced by adipokinase, free fatty acids and chronic inflammation in adipose tissue. The pancreatic beta cells compensate for insulin resistance by hypersecretion of insulin. However, at some point beta cell compensation is followed by beta cell failure and diabetes ensues20 (Fig1).
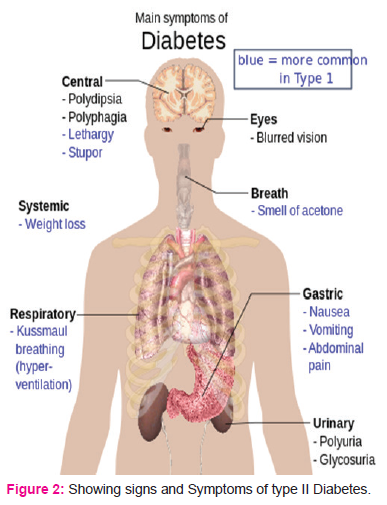
The classic symptoms of diabetes are polyuria (frequent urination), polydipsia (increased thirst), polyphagia (increased hunger), and weight loss. Other symptoms that are commonly present at diagnosis include a history of blurred vision, itchiness, peripheral neuropathy, recurrent vaginal infections, and fatigue. Many people, however, have no symptoms during the first few years and are diagnosed on routine testing. People with type II diabetes mellitus may rarely present with hyperosmolar hyperglycaemic state (a condition of very high blood sugar associated with a decreased level of consciousness and there is a great low blood pressure) (Fig 2)4.
World health organization (WHO) has predicted that India would experience the largest increase (48% increase in total population and 168% increase in population with >65 years of age) in type II DM and would have the greatest number of diabetic individuals in the world by the year 2030 (31.7 million in 2000 to 79.4 million in 2030) 21, 22.
CVD is the most prevalent complication of DM23. The age-adjusted cardiovascular mortality is at least 2-fold higher in diabetic men than in non-diabetic subjects in the presence of many numbers of major risk factors24. The survival after myocardial infarction is worse in diabetic men and women25.
In general population, women experiences relative protection from myocardial infarction and usually develop coronary artery disease (CAD) approximately 10 years later than men but diabetes blunts the cardiovascular benefits of female gender26.
The incidence of death from cardiovascular causes in diabetic subjects without a history of myocardial infarction during a 7-year follow-up was similar to the incidence observed in non-diabetic subjects with a history of myocardial infarction 27.
Coronary angiography is one of the most reliable procedures adopted to diagnose CAD and angiographic data on Indian patients with suspected CAD had revealed that triple vessel disease (TVD) was much higher in diabetes as compared to non-diabetics 28.
These observations highlight the high prevalence of undiagnosed CVD in type II DM and the gravity of cardiovascular events in this population. The problem of undiagnosed disease is the result of lack of awareness by the patients of the strong association between diabetes and CVD. Multiple factors contribute to the accelerated atherosclerosis in diabetes. These factors include excess prevalence of risks such as dyslipidemia and advanced glycosylated end products, obesity, hypertension, and, the state of insulin resistance29 (Fig. 3).
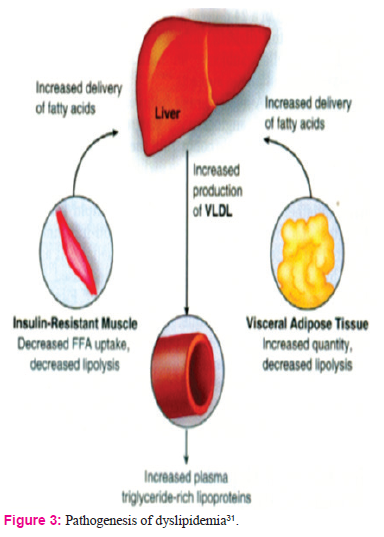
In type II DM there is a global dysfunction of lipoprotein metabolism. There is increased delivery of free fatty acids (FFA) to liver due to excess adipose efflux and impaired skeletal muscle uptake increases hepatic production of very low density lipoprotein (VLDL) and cholesterol ester synthesis. Over production of triglyceride-rich lipoproteins and impaired clearance by lipoprotein lipase leads to hypertriglyceridemia in diabetes30. Triglyceride level tends to vary inversely with HDL levels as cholesterol ester transfer protein mediates exchange of cholesterol from HDL to VLDL.
The combination of elevated triglycerides and low HDL is more common than elevated total and LDL cholesterol in diabetic patients with CAD. Increased concentration of small, dense LDL in diabetic person results from abnormal cholesterol and triglyceride transfer between VLDL, and LDL and depends on increased levels of VLDL, particularly when triglyceride concentration are higher than 130mg/dl31.
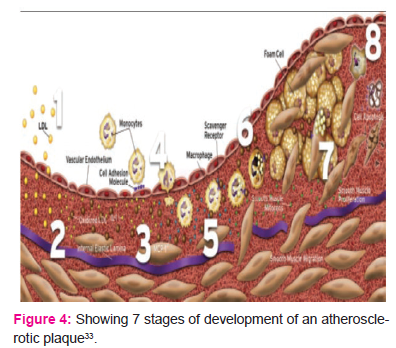
Small dense LDL-C is proatherogenic, first LDL-C moves into sub endothelium and is oxidized by macrophages and smooth muscle cells (stage 1 and 2). Release of growth factors cytokines attracts additional monocytes (stage 3 and 4). Foam cell accumulation and proliferation results in growth of the plaque (stage 6, 7, and 8) 20, 32 (Fig.4).
Atherosclerosis is the process underlying CVD, which includes coronary heart disease (CHD), myocardial infarction (MI), ischemic stroke, and peripheral vascular disease (PVD) 34. Atherosclerosis is the primary cause of death in patients with type II DM and it seems to be closely related to a specific cluster of lipid abnormalities, including low levels of HDL-C, increased numbers of small dense LDL-C, and elevated triglyceride levels11.
The classical dyslipidemia in type II DM is so called atherogenic dyslipidemia. This is a constellation of lipid abnormalities also known as “lipid triad” 10. This risk is even greater when the lipid triad is accompanied by insulin resistance, a procoagulant state, and hypertension–a condition known as the cardiovascular dysmetabolic syndrome. Each of these abnormalities is associated with an increased risk for cardiovascular morbidity and mortality.
Majority of Indian type II DM are dyslipidemic at baseline. The most common pattern of dyslipidemia is high LDL-C and low HDL-C among both males and females contributing to 22.7% and 33% patients of diabetic dyslipidemia, respectively35, 36.
The most prevalent problem among males is high LDL –C while among females low HDL –C emerged as a bigger threat. In Indian subjects with DM the lipid profile and pattern is greatly influenced by the ethnic origin, food habit, nutritional status and lifestyle influences. There has been a quantum increase in the incidence of CAD amongst urbanites while the picture in rural India has changed very little, suggesting the major impact of lifestyle modifications on lipid profiles and the deleterious effect of the latter in causing accelerated and more extensive CAD as evident angiographically37.
Hypercholesterolemia, hypertriglyceridemia, elevated LDL-C, and low HDL-C are generally accepted as strong risk factors for cardiovascular disease (CVD) and mortality38-40.
Several evidence has contributed to our current understanding of the relationship between increase in plasma cholesterol and development of CHD. Premature atherosclerosis results from high cholesterol levels, even in the absence of other cardiovascular risk factors. Large population surveys have shown that plasma cholesterol level is predictive of CHD41. In the Framingham study individuals below 50 years, cholesterol level was directly related to cardiovascular mortality. The study highlights the profound effects of lipoprotein abnormalities on incidence of CAD in diabetics compared to non diabetics42.
In a large prospective study, over 350,000 men aged 35 to 57 years were followed for 6 years. A curvilinear relationship between plasma cholesterol and coronary death rate was observed. If a risk ratio of 1 is assigned for a cholesterol level of 200 mg/dl, then at 250 mg/dl, the risk is doubled. This relation between cholesterol and CHD is not lost in the presence of other risk factors such as diabetes. The presence of diabetes further increased the risk of a given cholesterol level43. Because most cholesterol in plasma is transported in LDL and this is responsible for the correlation between plasma cholesterol and CHD. On the contrary some studies showed no significant difference in the lipids and lipoprotein profiles of diabetics and that of control5.
High-density lipoprotein cholesterol has been repeatedly shown to be an independent inverse predictor of CVD risk in epidemiological and observational studies, and patients with low HDL-C levels have been suggested to have a comparable CVD risk as those with high LDL-C levels44-45. The distribution of HDL-C levels varies with age, sex, race, and education. Women have higher levels of HDL-C than men.
Alcohol consumption is directly related to HDL-C levels and an inverse relation between smoking and HDL-C levels has been reported. Low HDL-C represents a highly prevalent and potentially modifiable risk factor for CVD prevention in type II DM46-47.
The rise in triglyceride rich lipoprotein concentrations which normally occur after a fat containing meal is found to be greater in type II DM patients than in people without diabetes48.
Exaggerated postprandial spikes in glucose and lipid levels can lead to an excess of free radicals and trigger a cascade of endothelial dysfunction and sympathetic hyperactivity- this state is known as postprandial dysmetabolism, is an
References:
-
Chitra R Hinge (Ingle), Baban D Adgaonkar and Sachin B Ingle, Study of waist circumference and lipid profile in type 2 diabetes as compared to healthy individuals Biomedicine 2016;36(3):82-89
-
Ganong’s Review of medical physiology. 24th edition; 2012:449-450.
-
Paul Z and Robert JS. Diabetes- A world wide problem. In: Kahn CR, Weis GC, George LK, Jacobson AM, Mose AC, and Smith JR, editors. Joslin’s Diabetes Mellitus. 14th ed. Place: Bi publication PVT LTD; 2006:525-529.
-
Harrisons Principles of internal medicine. 18th edition;2012:2968-3000.
-
Ugwa CE, Ezeanyika LUS, Daikwo MA and Amana R. Lipid profile of a population of diabetic patients attending Nigerian petroleum corporation clinic, Abuja. African Journal of Biochemistry Research 2009 Mar 20; 3(3):066-069.
-
Steinberger MD, Stephen R. Daniel. Obesity, insulin resistance, Diabetes and cardiovascular risk in children. Circulation 2003;107:1448-1453.
-
Kannel WB, Cupples LA, Ramaswami R, Stokes J, Kreger BE, Higgins M. Regional obesity and risk of cardiovascular disease; the Framingham Study. J Clin Epidemiol 1991;44:183-190.
-
Arya SN. Hypertension in diabetic patients-Emerging trends. J Indian Acad Clin Med 2003 April; 4(2):96-102.
-
Ishikawa-Takata K, Ohta T, Moritaki K, Gotou T and Inoue S. Obesity, weight change and risk for hypertension, diabetes and hypercholesterolemia in Japanese men. Euro J Clin Nutr 2002; 56:601-607.
-
Rizzo M and Berneis K. Lipid triad or atherogenic lipoprotein phenotype: A role in cardiovascular prevention. J Atherosclerosis Thromb 2005 June 29; 12(5):237-239.
-
Ballantyne CM. Hyperlipidemia: Diagnostic and Therapeutic Perspectives. The J Clin Endocrinol Metab 2000; 85(6):2089-2112.
-
Yalamanchali Himabindu, Manne Sriharibabu, Katam Reddy, Alekhya et al.Correlations between anthropometry and lipid profile in type II diabetics. Indian J. endocrinol metab 2013;17(4):727-729.
-
Williams SR, Jones E, Bell W, Davies B, and Bourne MW. Body habitus and coronary heart disease in men: A review with reference to methods of body habitus assessment. Eur Heart J 1997; 18(3): 376-393.
-
Ness-AR, and Caroline MA. Waist circumference measurement in clinical practice. Nutr Clin Pract 2008 Aug; 23(4):397-404.
-
Vazquez G, Duval S, Jacobs DR, and Silventoinen K. Comparison of body mass index, waist circumference, and waist/hip ratio in predicting incident diabetes: A meta analysis. Epidemiological reviews 2007 May 10; 42(6):1-14.
-
Raja Reddy P, Jayarama N. and Mahesh V.Study of anthropometric variables and lipid profile in Diabetics and Nondiabetics in rural Kolar. International Journal of basic and applied medical sciences 2013;3(2):263-266.
-
Janssen I, Katzmarzyk PT, and Robert R. Body mass index, waist circumference and health risk. Arch Intern Med 2002 Oct 14; 162: 2074-2079.
-
Maiti R, and Agrawal NK. Atherosclerosis in diabetes- Role of inflammation. Indian J Med Sci 2007; 61(5):292-306.
-
Goyal RK, Gandhi TP, and Satia MC. Role of hypertension control in diabetes mellitus and agents of choice. Indian Journal of pharmacology 1993; 25:181-187.
-
Text book of Robbins and Cotran Pathological basis of disease. 8th edition;2012:1131-1145.
-
Vijayakumar G, Arun R and Kutty VR. High prevalence of type II Diabetes Mellitus and other metabolic disorder in rural central kerala. J Assoc Phys India 2009 Aug; 57:563-567.
-
Das SK. Genetic epidemiology of adult onset type II diabetes in Asian Indian population: Past, Present and Future. Int J Hum genet 2006; 6(1):1-13.
-
Arghag D and Moordian. Cardiovascular diseases in type II diabetes mellitus. Arch Intern Med 2003; 163:33-40.
-
Stamler J, Vaccaro O, Neaton JD, Wentworth D. Diabetes, other risk factors, and 12-yr cardiovascular mortality for men screened in the Multiple Risk Factor Intervention Trial. Diabetes Care 1993; 16:434-444.
-
Sprafka JM, Burke Gl, Folsom AR, McGovern PG, Hahn LP. Trends in prevalence of diabetes mellitus in patients with myocardial infarction and effect of diabetes on survival: the Minnesota Heart Survey. Diabetes Care 1991; 14:537-543.
-
Donna MP and Tasneem ZN. Cardiovascular disease in women: Sex difference in presentation, risk factors and evaluation. Curr Cardiol Rep 2005; 7:166-172.
-
Steven MH, Seppo L, Tapani R, kalevi P, and Markku L. Mortality from coronary heart disease in subjects with type II diabetes and in non diabetic subjects with or without prior myocardial infarction. N Engl J Med 1998; 339:229-234.
-
Das S. Lipids, diabetes and coronary artery disease in Indians. Int J Diab Dev Countries 2004; 24:87-95.
-
James HO, Maia D, and Carl LJ. Primary and secondary prevention of cardiovascular diseases: A practical evidence- based approach. Mayo Clin Proc 2009 Aug; 84(8):741-757.
-
Barbara HV. Lipoprotein metabolism in Diabetes mellitus. J Lipid Res 1987; 28:613-628.
-
Douglas PZ, Libby P, Robert OB, Douglas LM, editors. Braunwald’s heart disease – A Text book of Cardiovascular Medicine. 8th ed; Philadelphia: Elsevier Saunders; 2008: 985-1002.
-
Kenneth CW and Allen Cooper D. Postprandial lipoproteins and atherosclerosis. Front Biosci 2001 march 1; 6:332-354.
-
Libby P and Pierre T. Pathophysiology of coronary artery disease. Circulation 2005; 111:3481-3488.
-
Boyle JJ. Macrophage activation in atherosclerosis: pathogenesis and pharmacology of plague rupture. Curr Vasc Pharmacol 2005; 3:63-68.
-
V. Mohan and R Pradeepa. Epidemiology of Diabetes in different regions of India. Health administrator 2009;22(1,2):1-18
-
Sandeep Singh, Ajaypal Singh, Manish Kishore Multani, Ashish Purohit. Clinical and biochemical profile of Indians with type II diabetes mellitus: A problem lurking for India. Tropical Journal of medical Research 2014;17(6):91-98.
-
Lt Gen Mehta SR, Col Kashyap AS, and Lt Col Das S. Diabetes Mellitus in India: The Modern Scourge. Medical Journal Armed Forces India 2009; 65:50-54.
-
Evans M, Roberts A, Davies S and Rees A. Medical lipid-regulating therapy: current evidence, ongoing trials and future developments. Drugs 2004; 64:1181- 1196.
-
Dean BB, Borenstein JE, Henning JM, Knight K and Merz CN. Can change in high-density lipoprotein cholesterol levels reduce cardiovascular risk? Am Heart J 2004 Jun; 147(6):939-941.
-
Walldius G and Jungner I. Apolipoprotein B and apolipoprotein A1: risk indicator of coronary heart disease and targets for lipid modifying therapy. Journal of Internal Medicine 2004; 255:188.
-
Steven HM, Seppo L, Tapani R, Kalevi P, and Markku L. Mortality from coronary heart disease in subjects with type II diabetes Mellitus and nondiabetic subjects with and without prior myocardial infarction. N Engl J Med 1998 July 23; 339(4):229-234.
-
Preis SR, Pencina MJ, Hwang SJ, Ralph BD, Savage PJ, Levy D et al. Trends in cardiovascular disease risk factors in individuals with and without diabetes mellitus in the Framingham Heart study. Circulation 2009; 120:212-220.
-
Turner RC, Millins H, Neil HW, Stratton M, Mathews DR and Holman RR. Risk factor for coronary artery disease in non insulin dependent diabetes mellitus: United Kingdom prospective diabetes study (UKPDS). BMJ. 1998; 316:823-828.
-
Richard GW and James MB. Prevalence and treatment of low HDL cholesterol among primary care patients with type II diabetes. Diabetes Care 2007; 30:479-484.
-
Haseeb AK. Clinical significance of HbA1c as a marker of circulating lipids in male and females tye II diabetic patients. Acta Diabetol 2007; 44:193-200.
-
Shai l, Robinson F, Basil R, Margaret C, Richard M, Dale W, Clifford J et al. High density lipoprotein cholesterol levels among us adults by selected demographic and socioeconomic variables. Am J Epidemiol 1976; 129(2):281-294.
-
Kumiko O, Michael K, Yoshimichi S, Paul W, Ichiro I and George C. Factors associated with high density lipoprotein cholesterol in Japanese and American Telephone Executives. Am J Epidemiol 1991; 134(2): 137-148.
-
Atonio C. The post-prandial state and cardiovascular disease: relevance to diabetes mellitus. Diabetes Metab Res Rev 2000; 16:125-132.
-
Breuer HW. Hypertriglyceridemia: a review of clinical relevance and treatment options: focus on cerivastatin. Curr med Res Opin 2001; 17(1):60-73.
-
Gotto AM. Triglyceride as a risk factor for coronary disease. Am J Cardiol 1998 Nov 5; 82(9A): 22-25.
-
Angela RA, Claudia DN, Lucrezia DM, Lidia P, Ciro I, Silvana C et al. Exogenous and endogenous postprandial lipid abnormalities in type II diabetic patients with optimal blood glucose control and optimal fasting triglyceride levels. J Clin Endo Metab 2004; 89(5):2153-2159.
-
Lewis GF, O’Meara NM, Soltys PA, Blackman JD, Iverius PH, Pugh WL et al. Fasting hypertriglyceridemia in non insulin dependent diabetes mellitus is an important predictor of postprandial lipid and lipoprotein abnormalities. J Clin Endo Metab 1990 June 7; 72(4):934-944.
-
Chen YD, Swami S, Skowronski R, Coulston A and Reaven GM. Difference in postprandial lipemia between patients with normal glucose tolerance and noninsulin-dependent diabetes mellitus. . J Clin Endo Metab 1993; 76:172-177.
-
Syvanne M, Hilden H and Taskinen MR. Abnormal metabolism of postprandial lipoproteins in patients with non-insulin-dependent diabetes mellitus is not related to coronary artery disease. J Lipid Res 1994; 35:15-26.
-
Gissette Reyes-Soffer, Steve Holleran, Wahida Karmally, Colleen Ngai, Neim-Tzuchan, Margarita Torres et al. Measures of postprandial lipoproteins are not associated with coronary artery disease in patients with type II diabetes mellitus. J Lipid Res 2009 Sept; 50:1901-1909.
-
Goran W and Ingmar J. Rationale for using apolipoprotein B and apolipoprotein A-1 as indicators of cardiac risk and as targets for lipid-lowering therapy. Eur Heart J 2005; 26: 210-212.
-
Sarah P, Richard P, Alison P, Robert C, Sarah L, Alison O et al. The joint effects of apolipoprotein B, apolipoprotein A1, LDL cholesterol, and HDL cholesterol on risk: 3510 cases of acute myocardial infarction and 9805 cases Eur Heart J 2009; 30:2137-2146.
-
Ana MW, Antonio P, Edgar Z, and Jordi OL. Non-HDL Cholesterol and Apolipoprotein B in the dyslipiemic classification of type II Diabetic patients. Diabetes Care 2003 July; 26(7):2048-2051.
-
Gosta F, Samar B and Anders L. Apolipoprotein A1 is a stronger prognostic merker than are HDL and LDL Cholesterol for cardiovascular disease and mortality in elderly men. Journal of Gerontology 2006; 61A(12):1262-1266.
-
Youfa W, Eeric BR, Meir JS, Walter CW and Frank BH. Comparison of abdominal adiposity and overall obesity in predicting risk of type II diabetes mellitus among men. Am J Clin Nutr 2005; 81:555-563.
-
Kathryn RM, Vincent CJ, Charles HH, Ellen WE, Graham AC, Meir SJ et al. Abdominal adiposity and coronary heart disease in women. JAMA 1998; 280(21):1843-1848.
-
Ian J, Steven H, David AB, Donald K and Robert R. Body mass index and waist circumference independendly contribute to prediction of nonabdominal, abdominal subcutaneous, and visceral fat. Am J Clin Nutr 2002; 75:683-688.
-
Philipp SE. Adipose tissue: From lipid storage compartment to endocrine organ. Diabetes 2006 June; 55:1537-1545.
-
Ridker PM, Rifai N and Cook NR. Non HDL cholesterol, Apolipoproteins A! And B100, Standard lipid measures, lipid ratios and CRP as risk factors for cardiovascular disease in women. JAMA 2005; 294:326-333.
-
Luigi F, Cristopher E, Maria T, Philipp S and Samuel K. Visceral fat adipokinine secretion is associated with systemic inflammation in obese humans. Diabetes 2007 April; 56:1010-1013.
-
Eva R, Braian W, Stefan S, Dawn L, Mats E, Owe J et al. Tissue specific changes in peripheral cortisol metabolism in obese women: increased adipose 11β-hydroxysteroid dehydrogenase type1 activity. J Clin Endo Metab 2002; 87(7):3330-3336.
-
Karin BM, Sander G and Bernard R. Does body mass index adequately captures the relation of body composition and body size to health outcomes. Am J Epidemiol 1998; 147(2):167-172.
-
Amalia G. Abdominal fat: does it predict the development of type II diabetes. Am J Clin Nutr 2008; 87:1118-1119.
-
Eric R, Meir S, Edward G, Alberto A, Donna S, Graham C, and Walter W. Body size and fat distribution as predictor of coronary heart disease among middle aged and older US men. Am J Epidemiol 1995; 141(12):1117-1127.
-
Helke MF, Fahim A and Gerald R. Body mass index and waist circumference both contribute to difference in insulin mediated glucose disposal in nondiabetic adults. Am J Clin Nutr 2006; 83:47-51.
-
Ho SC, Chen YM, Woo JL, Leung SSF, Lam TH, and Jannus ED. Association between simple anthropometric indices and cardiovascular risk factors. Int J Obes 2001; 25:1689-1697.
-
Bektas MY, Erkan MS and Ersa Y. Which anthropometric measurements are most closely related to elevated blood pressure? Family Practice 2005; 22:541-547.
-
Carl EM. Combined high blood pressure and glucose in type II diabetes: double jeopardy. BMJ. 1998; 317(12):693-694.
-
Paul P, Isabelle L, Pascale M, Eric D, Carole B, Jean B et al. Impact of waist circumference on relationship between blood pressure and insulin. Hypertension 2005; 45:363-367.
-
Dorothy B, Jean PH, Ethan SA, David S and Sidney A. Evidence for an increase risk for hypertension with centrally located body fat and effect of race and sex on this risk. Am J Epidemiol 1984; 119(4):526-540.
-
Johan S, Eric V, Riserus Ul, Lisa B, Bjorn Z, Cristian B, et al. Risk associated with the metabolic syndrome versus the sum of its individual components. Diabetes Care 2006; 29(7):1673-1674.
-
Anderson PJ, Critchley JAJH, chan JCN, Cockram CS, Lee ZSK, Thomas GN and Tomlinson B. Factor analysis of metabolic syndrome: obesity vs. insulin resistance as the central abnormality. Int J Obes 2001; 25:1782-1788.
-
Eriksson J, Lindstrom J, Valle T, Aunolaz S, Hamalainenz H, IIanne-Parikkas P, et al. Prevention of type II diabetes in subjects with impaired glucose tolerance: the Diabetes Prevention Study (DPS) in Finland. Diabetologia 1999; 42:793-801.
-
Trinder, P. Ann Clin biochem. 1969;6(24).
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License