IJCRR - 10(7), April, 2018
Pages: 22-27
Date of Publication: 14-Apr-2018
Print Article
Download XML Download PDF
Efficacy of Pemetrexed or Pemetrexed Plus Cisplatin/Carboplatin in Pretreated Patients with Advanced Non\?Small-Cell Lung Cancer
Author: Nurgul Yasar, Caglayan Geredeli
Category: Healthcare
Abstract:Introduction: In this study, the efficacy and safety of pemetrexed alone and platinum-pemetrexed combination chemotherapy were evaluated in patients who have demonstrated progression after the first-line treatments in advanced non-small cell lung cancer (NSCLC).
Material and Method: 263 patients, who were diagnosed NSCLC in years 2008 - 2014, were assessed retrospectively in single center. Patients were given pemetrexed 500 mg/m2, and the ones receiving combined treatment were given pemetrexed 500 mg/ m2 and cisplatin 75 mg/m2 or carboplatin area under the curve 5 according to the Calvert formula (AUC 5 ) once in every 21 days.
Results: One hundred ninety (72%) of the patients had received pemetrexed, seventy three (28%) of the patients had received platinum- pemetrexed. Median PFS (progression-free survival) was 2 months (95% CI, 1. 6 - 2.4 ) for pemetrexed arm versus 4 months (95% CI, 2.6 to 5.3 ) for platinum- pemetrexed arm (p=0.001). The HR ( hazard ration) for disease progression was 0.45 (95% CI, 0.25 to 0.65) in favor of the combination arm (p=0.001). Median OS (overall survival) was 7 months (95% CI, 5.9 - 8 ) for pemetrexed arm versus 10 months (95% CI, 8 to 11.9 ) platinum-pemetrexed arm, respectively (p=0.001). The 1-year survival
rate was 24% and 42% for pemetrexed arm and combination arm, respectively. Toxicities in both arm was manageable.
Conclusion: Our study has shown that adding platinum compound to second-line pemetrexed chemotherapy significantly increases ORR (overall response rate), PFS and OS in patients with advanced NSCLC after having received first-line platinumbased chemotherapy.
Keywords: NSCLC, Second line treatment, Pemetrexed
DOI: 10.7324/IJCRR.2018.1075
Full Text:
Introduction
Lung cancer, estimating for approximately 13% of total cancer cases, pursues one of the major causes of cancer-related death worldwide with an estimated 1.8 million new lung cancer cases occured in 2012(1). Non-small cell lung cancer (NSCLC) accounts for 80-85 % of all lung cancers, and 75%of patients are diagnosed at the advanced stages of disease (2). First-line treatment for patients with stage IIIB or stage IV NSCLC usually consists of platinum-based doublet chemotherapy was found to produce a survival benefit(3,4). Nonetheless, disease progression ultimately occurs for most patients and further treatment is reguired.(5). When compared with best supportive care, second-line chemotherapy with docetaxel, pemetrexed or other agents are associated with (OS) benefit and improvement of quality of life (5,6,7).
Pemetrexed, an analogue of folic acid, was approved for first-line, second-line and maintenance treatmentof advanced NSCLC (4,7). It inhibits three enzymes which necessary for de novo pyrimidine and purine synthesis:thymidylate synthase (TYMS), dihydrofolate reductase, and glycinamide ribonucleotide formyltransferase (4,7). In the first-line setting, pemetrexed/platinum combination is more effective than gemcitabine/platinum combination for advanced NSCLC patients with adenocarcinoma histologic subtype (4). In the second-line therapy, pemetrexed was associated with more favorable toxicity profile compared with docetaxel and comparable efficacy (median survival of 8.3 months vs 7.9 months)(7).
Combinations of chemotherapy agents have been competent in increasing efficacy over single agents in the first-line treatment(8,9). On the other hand, role of combinations is less clear in the second-line treatment (8,9). In this study, the efficacy and safety of pemetrexed alone and platinum-pemetrexed combination chemotherapy were evaluated retrospectively in patients who were non-responders or who have demonstrated progression after the first-line treatments in local advanced and metastatic NSCLC.
Materials and methods
We performed a retrospective screening of 263 patients who had pathologically or cytologically confirmed as stage IIIB to IV lung adenocarcinoma in SB Okmeydani Training and Researh Hospital between November 2008 and May 2014. These patients had failed prior chemotherapy regimen and received pemetrexed alone or platinum-pemetrexed combination chemoterapy rejimens in the second-line setting. Data were collected on baseline characteristics including age, gender, Eastern Cooperative Group (ECOG) performance status (PS), histology, stage, single agent or combined administration of treatment, third or further-line treatment intake and length of progression-free period after the first-line treatment.
Pemetrexed 500 mg/m2 administered as an intravenous (IV) bolus infusion of 10 minutes duration every 3 weeks. Folic acid and vitamin B12 supplementation was mandatory for all patients. Dexamethasone 4 mg tablet, three times a day for 3 days, was administered routinely to prevent allergic reactions. Cisplatin 75 mg/m2 administration intravenously over a 2-h infusion or carboplatin area under the curve 5 according to the Calvert formula (AUC 5 ) a 30-min infusion after pemetrexed administration every 3 weeks.
Patients underwent baseline computed tomography (CT) at the beginning of second-line chemotherapy and computed tomography repeated every 2-3 cycles of chemotherapy. Evaluation of treatment response was based on the Response Evaluation Criteria in Solid Tumors (RECIST) (10). Patients achieving complete response and partial response were recognized to be responders. Adverse events (AEs) were classified according to National Cancer Institute Common Terminology Criteria for Advers Events (CTCAE), version 3.
statistical analysis
The baseline characteristics of the patients were analyzed using descriptive statistics. PFS of second-line chemotherapy was measured from the date of initiation of second-line chemotherapy to the date of disease progression or any cause of death. OS for second-line chemotherapy was calculated from the date of initiation of second-line chemotherapy to the date of death from any cause. The Kaplan-Meier method was used to estimate PFS and OS. The difference between the survival curves of the treatment groups was tested using the log-rank test. In multivariate analysis with the Cox proportional hazards model, including age, gender, ECOG performance status, stage, the length of progression-free period after first- line treatment and the number of course for first and second-line chemotherapy, were used to estimate for PFS and OS. The chi-square test or Fisher exact test was used to compare the ORRs and adverse events (AEs) between two groups. A two-sided p value of ≤0.05 was considered statistically significant. All statistical analyses were performed using SPSS statistics 17.0 (SPSS, Inc, Chicago, IL).
Results
From November 2008 to May 2014, 263 patient files were screened onto this study. The median follow-up period was 7 months (range, 1 to 74 months). Two hundred thirty-seven patients (90%) died during the follow-up. Patients characteristics were listed in Table 1.Two hundred twelve (81%)males and 51 (19%) females were included. Median age at diagnosis was 55 years (range,28-83), 85% had PS of 0 to 1, 91% had stage IV disease, and 94% of the patients were diagnosed with adenocarcinoma.
The length of progression-free period after first-line of treatment was 6 months (range, 1- 88 months) for patient groups. The median time off platinum treatment was 6 months (range, 1 to 49 months) in pemetrexed arm and 7 months (range, 6 to 88 months) in platinum-pemetrexed arm. Median number of courses was five for the first-line setting. One hundred ninety (72%) of the patients had received pemetrexed, seventy three (28%) of the patients had received platinum- pemetrexed as second-line treatment. None of the patients had received pemetrexed as part of first-line treatment. Median number of cycles was four in patient groups for the second-line treatment. Two patients stopped treatment because of an adverse event. Three percent of patients had a dose reduction for pemetrexed arm, and 4%%of patients had a dose reduction for platinum-pemetrexed arm.
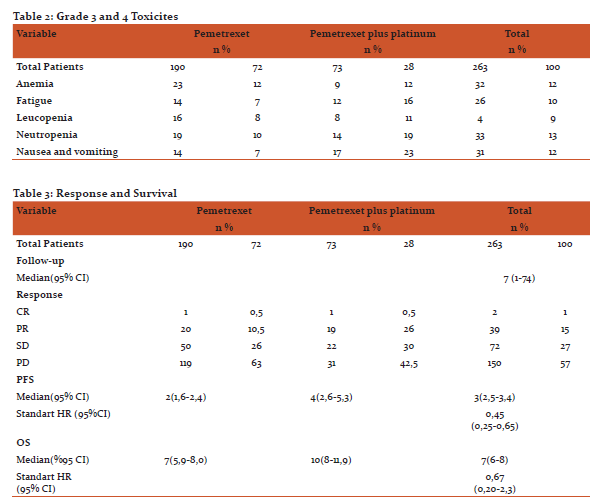
With the exception of hematologic toxicity, fatigue, nasuea and vomiting, the frequency of treatment-related toxicity exceeding CTCAE grade 2 was less than 5% for all categories (Table 2). Neutropenia (12,5%), nasuea and vomiting (12%) and fatique (10%) were all more frequently observed in the platinum-pemetrexed arm, significantly (p<0,05) .In particular, the incidence of hospitalization cause of febrile neutropenia was 3,4% in both treatment arms.
Two complete response were observed, whereas 39 patients (15%) had a confirmed partial response as the best response (Table 3). Nineteen (26%) of these patients were the combination of platinum and pemetrexed, whereas 20 (10% ) patients received pemetrexed alone (p=0,001). The median duration of response was 3 months. More patients in the pemetrexed arm had disease progression as compared with the combination arm (119 patients [63%] versus 31 patients [42%],(p=0,003) ).
Median PFS of the patients treated with pemetrexed alone was 2 months (95% CI, 1.6 to 2.4 months) and median PFS was 4 months (95% CI, 2.6 to 5.3 months) for patients treated with platinum and pemetrexed (Figure1). The HR for disease progression was 0.45 (95% CI, 0.25 to 0.65) in favor of the combination arm (p=0,001).Median overall survival was 7 months (95% CI, 5.9 to 8 months) versus 10 months (95% CI,8 to 11.9 months) for pemetrexed arm and platinum-pemetrexed arm, respectively (p=0,001, HR =0.67 [95%CI,0.20 to2.3]) (Table 3 figure 2). The 1-year survival rate was 24% and 42% for pemetrexed arm and combination arm, respectively.
Log-rank tests stratified by the treatment arm indicated that performance status and gender were significantly related to OS (p=0,034, p=0,032 ). Female gender had a better prognosis than male gender. . This model indicated that survival was worse for patients with higher performance status (p=0, 001). In addition, multivariate analysis, age, gender, stage, progression-free interval of more than 6 months after first- line were not found to be effective on survival.
Discussion
Several prospective trials and meta-analyses clearly defined the role of combination chemotherapy particularly for platinum-based rejimens in the first-line treatment of advanced NSCLC (11,12). Unfortunately, these patients show progression after the first-line treatment and most of them are the candidates for the second-line treatment (5,7,1). In the second-line treatment, docetaxel, pemetrexed and erlotinib have been registered by the European Medicines Agency (EMA) and US Food and Drug Administration for treatment of advanced NSCLC (5,6,7,13). Although these treatments are efficient for the second-line, these patients show progression readily and survival of patients with advanced NSCLC is poor (5,7). In this retrospective study for the second-line treatment, we aimed to invastigate the efficiency of pemetrexed alone and platinum-pemetrexed combination rejimens for the patients received platin-based combination chemotherapy before.
In advanced NSCLC patients who received first-line setting pemetrexed-platinum combination chemotherapy, approximately 28-45% ORR and 4.8-6.9 months of PFS were announced by the studies had carried out before (14,15). Meta analysis of Di Maio et al (16) included 847 eligible patient showed that in comparison of combination chemotherapy and single-agent therapy for the second-line treatment of advanced NSCLC, combination chemotherapy increased RR (p=0.0004) and PFS(p=0.0009) significantly but not OS (p=0.32). OS was not significantly different between arms (p= 0.32)(16) (16). Median OS was 37.3 and 34.7 weeks in the doublet and single-agent arms, respectively. HR was 0.92 (95% CI, 0.79 to 1.08). Response rate was 15.1% with doublet and 7.3% with single-agent (p=.0004) (16). Median progression-free survival was 14 weeks for doublet and 11.7 weeks for single agent (p = .0009; HR, 0.79; 95% CI, 0.68 to 0.91). According to this meta analysis, toxicity was higher in combination rejimen, as well (16).
Smit et al (17) reported that median PFS was 2.8 month for patients second line treated with pemetrexed alone and 4.2 months for those treated with carboplatin and pemetrexed and the HR for disease progression was 0.67 (95% CI, 0.51 to 0.89) in favor of the combination arm (p=.005). Whereas, median overall survival was 7.6 months to 8.0 months (HR, 0.85; 95% CI, 0.63 to 1.2; p=not significant) (17). Our results (PFS was 2 month and OS was 4 month) are quite similar to those of Smit et al with 2.8 months of PFS and 4.2 months of OS. Ardizonni et al(GOIRC 02-2006 Trial and NVALT7) pooled analysis (18) recently published that median OS was not different between the two treatment arms: 8.2 months for pemetrexed alone (95% CI, 6.9 to 9.1 month) as compared with 8.7 months for carboplatin- pemetrexed (95% CI, 7.5 to 10.5 months; HR, 0.90; 95% CI, 0.74 to 1.10; p=0.316). However, RR was higher (15% v 9%) in the carboplatin-pemetrexed arm, with an OR of 1.72 (95% CI, 0.97 to 3.02; p=0.062).(18) A non istatistically significant increase in PFS from 3 months (95% CI, 2.7 to 3.3) to 3.9 months (95% CI, 3.4 to 4.4), favoring the combined chemotherapy arm, was also observed, with an HR of 0.85 (95% CI, 0.70 to 1.02; p=0.07)(18). Our PFS results (PFS was 2 month ) was shorter than to those of Ardizonni et al with 2.8 months of PFS.
In our study we demostrated that median PFS was 2 months (95% CI, 1.6 to 2.4 months) and 4 months (95% CI, 2.6 to 5.3 months) for patients treated with pemetrexed alone and platinum- pemetrexed combination, respectively. HR for disease progression was 0.45 (95% CI, 0.25 to 0.65) in favor of the combination arm (p=0,001). Median overall survival was 7 months (95% CI, 5.9 to 8 months) versus 10 months (95% CI,8 to 11.9 months) for pemetrexed arm and platinum-pemetrexed arm, respectively (p=0,001, HR =0.67[95%CI, 0.20 to2.3]). One-year survival rate was 24% and 42% for pemetrexed arm and combination arm, respectively. In our study, both PFS and OS was statistically different in favour of the combination arm. Although our study was retrospective, the number of cencored case was low due to 90% of the patients death at the end of the study. Additionally, our patient group consisted of only the patients who had non-squamous histology and 85% of the patients had 0-1 PS. Partial response rate was higher in combination arm and progression rate was higher in pemetrexed arm significantly (p=0.001, p=0.003, respectively).
In comparison of pemetrexed arm and platinum-pemetrexed arm, in the means of grade 2 or more toxicity; although, fatique, neutropenia, nasuea and vomiting were observed more in combination arm, there was no difference in febrile neutropenia and quiting due to side affects.
Pemetrexed is a chemotherapy agent used in advanced NSCLC patients for the first-line, second-line and maintenance treatments, which has favourable toxicity profile (7,13,14,15). Contrarily, since its survival in advanced NSCLC is disappointing, more effective treatment options are being invastigated continously. Studies of pemetrexed combination with other chemotherapy agents except platinium compounds like targeted agents and immunotherapy agents are being carried out (19-25). While some of these studies are promising, some of them are fail. It was demostrated that the EGFR mutant patients treated with first-line platinum-pemetrexed combinations, ORR was significantly higher than that of the wild-type patients treated with similar regimens (43 vs. 21 %, p=0.039) (26). Additionally, in another study erlotinib statistically decreased TS expression and activity, maybe means that it increases the sensitivity of pemetrexed (27). Pemetrexed can be used for second-line treatment with good response rate in patients having TKIs resistance (27). All these studies shows that pemetrexed usage is going to continue in advanced NSCLC treatment.
There was several limitations in our study. First, this was a retrospective study and population of the study may not represent the average population. Second, the number of the patients in pemetrexed group and combination chemotherapy group were not the same. Third, treatments after second-line is not specified. Fourth, Although very few patients had received anti-EGFR therapy after the second-line treatment, situation of EGFR mutation in patients was not known. Fifth, the choice of cisplatin or carboplatin, and response evaluation are dependent to the clinical practice of oncology physicians. This study design has a lack of standardization and quality control.
In conclusion, our study has shown that adding platinum compound to second-line pemetrexed chemotherapy significantly increases ORR, PFS and OS in patients with advanced NSCLC after having received first-line platinum-based chemotherapy and the toxicity profile of pemetrexed/ platinum combination rejimen is manageable. It is needed that further randomized prospective studies with larger samples on efficiency of platinum-pemetrexed combination or less toxic pemetrexed- based combinations in second-line chemotherapy for advanced NSCLC.
Acknowledgements
Author acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The author is also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
Source of funding: No funding source
Conflict of interest: No conflict of interest


References:
.Torre LA, Bray F, Siegel R et al.. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108.
3. Schiller JH, Harrington D, Belani CP, et al. Eastern Cooperative Oncology Group. Comparison of four chemotherapy regimens for advanced nonsmall-cell lung cancer. N Engl J Med 2002;346:92–98.
4. Scagliotti GV, De Marinis F, Rinaldi M, et al.; Italian Lung Cancer Project. Phase III randomized trial comparing three platinum-based doublets in advanced non-small-cell lung cancer. J Clin Oncol 2002;20:4285–4291.
5. Shepherd FA, Dancey J, Ramlau R, et al: Prospective randomized trial of docetaxel versus best supportive care in patients with non-smallcell lung cancer previously treated with platinumbasedchemotherapy. J Clin Oncol 2000 ;18:2095-2103.
6. Shepherd FA, Pereira J, Ciuleanu TE, et al: Erlotinib in previously treated non small cell lung cancer. N Engl J Med 2005; 353:123-132
7. Hanna N, Shepherd FA, Fossella FV, et al: Randomized phase III trial of pemetrexed versus docetaxel in patients with non small cell lung cancer previously treated with chemotherapy. J Clin Oncol 2004; 22:1589-1597
8. R, HerndonJ, ListM, et al. Single-agent versus combination chemotherapy in advanced non-small cell lung cancer (NSCLC): a CALGB randomized trial of efficacy, quality of life, and cost-effectiveness. Eur J Cancer 2001; 37: S153–S154.OpenUrl
9.Le ChevalierT, BrisgandD,DouillardJY, et al.Randomized study of vinorelbine and cisplatin versus vindesine and cisplatin versus vinorelbine alone in advanced non-small-cell lung cancer: results of a European multicenter trial including 612 patients. J Clin Oncol 1994; 12: 360–367.
10. Eisenhauer EA, Therasse P, Bogaerts J et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2): 228–47.
11. Azzoli CG, Baker S Jr, Temin S, et al: American Society of Clinical Oncology clinical practice guideline update on chemotherapy for stage IV non–small-cell lung cancer. J Clin Oncol 2009; 27:6251- 6266.
12. Delbaldo C, Michiels S, Syz N, et al: Benefits of adding a drug to a single-agent or a 2-agent chemotherapy regimen in advanced non-small-cell lung cancer: A meta-analysis. JAMA 2004; 292:470-484.
13. Smit EF, Mattson K, von Pawel J, et al: ALIMTA (pemetrexed disodium) as second line treatment of non-small cell lung cancer: A phase II study. Ann Oncol 2003; 14:455-460.
14. Scagliotti GV, Parikh P, von Pawel J, Biesma B, Vansteenkiste J, Manegold C, et al. Phase III study comparing cisplatin plus gemcitabine with cisplatin plus pemetrexed in chemotherapy-naive patients with advanced-stage non-small-cell lung cancer. J Clin Oncol. 2008;26(21):3543–51
15. Zinner RG, Fossella FV, Gladish GW et al. Phase II study of pemetrexed in combination with carboplatin in the first-line treatment of advanced nonsmall cell lung cancer. Cancer. 2005;104(11):2449–56.
16. Di Maio M, Chiodini P, Georgoulias V, et al.Meta-analysis of single-agent chemotherapy compared with combination chemotherapy as second-linetreatment of advanced non-small-cell lung cancer. J Clin Oncol 2009; 27:1836-1843.
17.Smit EF, Burgers SA, Biesma B, et al: Randomized phase II and pharmacogenetic study of pemetrexed compared with pemetrexed plus carboplatin in pretreated patients with advanced non–small-cell lung cancer. J Clin Oncol 2009; 27:2038-2045.
18. Ardizzoni A, Tiseo M, Boni L et al. Pemetrexed versus pemetrexed and carboplatin as second-line chemotherapy in advanced non-small-cell lung cancer: results of the GOIRC 02-2006 randomized phase II study and pooled analysis with the NVALT7trial. J Clin Oncol. 2012 20;30(36):4501-7.
19. Kim YH, Nishimura T, Ozasa H et al. Phase II study of pemetrexed and erlotinib in pretreated nonsquamous, non-small-cell lung cancer patients without an EGFR mutation. Chemotherapy. 2013;59(6):414-9.
20. Yoshimura N, Kudoh S, Mitsuoka S, Yoshimoto N et al.Phase II study of a combination regimen of gefitinib and pemetrexed as first-line treatment in patients with advanced non-small cell lung cancer harboring a sensitive EGFR mutation Lung Cancer. 2015 ;90(1):65-70.
21. Daga H, Takeda K, Okada H et al. Phase I study of nintedanib in combination with pemetrexed as second-line treatment of Japanese patients with advanced non-small cell lung cancer.Cancer Chemother Pharmacol. 2015;76(6):1225-33.
22. Zhou CZ, Qin YY, Xie ZH et al.Efficacy of third-line pemetrexed monotherapy versus pemetrexed combination with bevacizumab in patients with advanced EGFR mutation-positive lung adenocarcinoma Chin J Cancer Res. 2014;26(6):705-10.
23. Vynnychenko I, Bondarenko I, Shparyk Y et al.An open-label, multicenter, randomized phase Ib/II study of eribulin mesylate administered in combination with pemetrexed versus pemetrexed alone as second-line therapy in patients with advanced nonsquamous non-small-cell lung cancer.Clin LungCancer. 2015;16(2):92-9.
24..Choi MK, Hong JY, Chang et al. Safety and efficacy of gemcitabine or pemetrexed in combination with a platinum in patients with non-small-cell lung cancer and prior interstitial lung disease.Cancer Chemother Pharmacol. 2014;73(6):1217-25.
25.. Heist RS, Wang X, Hodgson L et al; Alliance for Clinical Trials in Oncology.CALGB 30704 (Alliance): A randomized phase II study to assess the efficacy of pemetrexed or sunitinib or pemetrexed plus sunitinib in the second-line treatment of advanced non-small-cell lung cancer J Thorac Oncol. 2014;9(2):214-21.
26. Li S, Zhou F, Ren S, Zhou C et al. Response to pemetrexed rechallenge after acquired resistance of EGFR-TKI in a patient with advanced NSCLC. Lung Cancer. 2014;84(2):203-5.
27.Xiangli Jiang , Bo Yang , Jiuqin Lu et al.Pemetrexed-based chemotherapy in advanced lung adenocarcinoma patients with different EGFR genotypes. Tumor Biol. 2015; 36:861–869.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License