IJCRR - 2(7), July, 2010
Pages: 27-38
Print Article
Download XML Download PDF
WATER REQUIREMENT, DEFICIT IRRIGATION AND CROP COEFFICIENTS FOR PEPPER
Author: J.D. Owusu-Sekyere, S. Twum
Category: Technology
Abstract:An experiment was conducted to determine the seasonal water requirement of pepper, the crop coefficient under full water requirement as well the effect of deficit irrigation on pepper growth and development under a rain shelter. The treatments imposed were T1, application of 100% crop water requirement, T2, was 80%, T3 60% and T4 40 % of crop water requirement. It was determined that pepper requires about 526.7 mm of water over the growth season. The crop coefficient under full water supply was found to be: 0.43, 0.75, 1.37 and 0.94 for initial, development, mid-season and the late season stages respectively. It was also found out that a water deficit of 20% lead to growth and development that was not significantly different from pepper production under full water requirement.
Keywords: Deficit irrigation, crop coefficient, water requirement, pepper
Full Text:
Abstract
An experiment was conducted to determine the seasonal water requirement of pepper, the crop coefficient under full water requirement as well the effect of deficit irrigation on pepper growth and development under a rain shelter. The treatments imposed were T1, application of 100% crop water requirement, T2, was 80%, T3 60% and T4 40 % of crop water requirement. It was determined that pepper requires about 526.7 mm of water over the growth season. The crop coefficient under full water supply was found to be: 0.43, 0.75, 1.37 and 0.94 for initial, development, mid-season and the late season stages respectively. It was also found out that a water deficit of 20% lead to growth and development that was not significantly different from pepper production under full water requirement. Keywords: Deficit irrigation, crop coefficient, water requirement, pepper
Introduction
Scarce water resources and growing competition for water has reduced its availability for irrigation. At the same time, the need to meet the growing demand for food requires increased crop production from less water. Achieving greater efficiency of water use will be a primary challenge for the near future and will include the employment of techniques and practices that deliver a more accurate supply of water to crops. In this context, deficit irrigation can play an important role in increasing water use efficiency (FAO, 2003). Deficit irrigation is accomplished by allowing planned plant stress during one or more periods of the growing season. Adequate water is supplied during critical growth stages to maximise water use efficiency. It is economically justified when reducing water applications below full irrigation causes production costs to decrease faster than revenue declines therefore irrigation water management in an era of water scarcity will have to be carried out most efficiently, aiming at saving water and at maximizing its productivity. AVRDC (2006) has placed emphasis on water use efficiency in pepper production because in recent times, the demand for pepper has increased steadily due to its diversified uses. The pepper fruit is mainly used as spices in almost every home (Cobbley and Steele, 1979). It is a common ingredient in the herbal preparation for douching and also in local ointment for rheumatic pains, sprains and twisted joints. Pharmaceutical companies use it in analgesic creams that are applied to release pain of arthritis, shingles, cluster headache and ailments (Levetin and Mcmahon, 1999). Powdered forms of pepper are used in preparing hot sauce, pickles curry powder and condiment (Cobbley and Steele, 1976). Many countries are going into commercial cultivation of hot pepper to meet the growing demand (AVRDC, 2006). Present world production is about 21.3 million tons fresh fruits from 1.6 million hectares and Ghana ranks 15th in the world and 2rd in Africa for hot pepper production. Hot pepper is thus becoming an important export commodity for Ghana. The most recent data available from 2000-2001 showed that Ghana?s pepper export rose from 2.8 thousand tons to 5.2 thousand tons (FAOSTAT, 2001). Water is one of the critical inputs for obtaining maximum production of a particular crop. According to Evans et al. (1993), to avoid moisture stress in the root zone of plants, it is important to establish crop coefficients (Kc) and measurements or calculations of potential evapotranspiration to estimate water use by hot pepper to adopt efficient and strategic use of water while obtaining optimum crop yield. Grimes and Williams (1990) added that crop coefficients for pepper have been developed in several different locations elsewhere in the world with different values and their corresponding water requirements. However, the challenge is whether these values are compatible with our environmental conditions in Ghana. To answer this question, an experiment was conducted to determine the crop coefficient values and water requirement as well as deficit irrigation on the growth and development of pepper at the University of Cape Coast School of Agriculture Teaching and Research Farm.
Materials and Method Study Area
The experiment was conducted during the minor rainy season which normally starts in September to Mid November at the School of Agriculture research farm of the University of Cape Coast, located in Cape Coast which lies on latitude 05- 06 degrees north and longitude 01-15 degrees south at an altitude of 1.1m. The soil is described as sandy loan with characteristics as neutral to slightly acid in reaction and with a pH of 6.5. This site lies within the costal savannah vegetation zone of Ghana.
Experimental design and field layout
The randomized complete block design was used. There were four treatments (100%, 80%, 60%, and 40%) which were replicated three times (R1-R3). This gave a total of forty-eight (48) transplants. The experiment was carried out using 48 nursery bags each filled with sandy loam to about 6Kg. The experiment was conducted under a rain shelter in order to exclude the influence of rainfall, and it was conducted between September 2009 and February 2010.
Planting
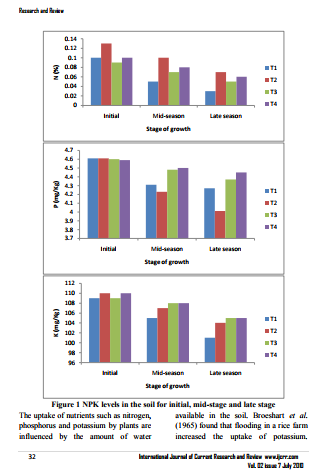
Seeds of Legon 18 variety of hot pepper were nursed on September 15, 2009 and transplanted after 30 days on October 15, 2009. A week before transplanting, watering at the nursery was reduced in order to harden the seedlings so as to make them withstand transplanting shock. Prior to transplanting, the nursery was watered until near to soil saturation to enhance easy uprooting of seedlings and to prevent damage to the roots of the seedlings. Transplanting was done two days after watering the soil in the bags to saturation. A seedling was put per nursery bag. There were 12 plants per treatment. Weeds were removed as soon as they appeared. Treatment was imposed r after five days when the plants had established. The treatments imposed were 100% of crop water application (T 1), 80% of crop water application (T 2), 60% of crop water application (T 3), and 40% of crop water application (T 4). Growth stages Four growth stages were considered. They were the initial stage, developmental stage, mid-season stage, and late season stage. The initial stage excluding seedlings at the nursery lasted for 15 days (October 15 – October 30, 2009). The developmental growth stage lasted for 30 days (October 30, 2009– November 29, 2009). The mid-season growth stage (flowering and fruiting) stage lasted for 50 days (November 29, 2009 – January19, 2010) and the late season stage lasted for 26 days (January19, 2010 – February 14, 2010). This stage was later characterized by senescence and drying of leaves after the harvesting was over
Irrigation regime
A two-day interval irrigation regime was adopted and the amount of water to be applied each two-day interval was derived from the computed loss in weight of each set up over the two days. The equivalent in volume basis was found and applied to the plants as the various treatments demanded. Irrigation days amounted to 60.5 days out of the 121 days of the growing period
Determination of crop coefficient (Kc) and water requirement (ETc) and reference evapotranspiration (ETo)
ETc was determined by weighing, and ETo determined by measuring water loss from an evaporation pan. Kc was determined using the following formula: Kc = Etc/ETo (1) Soil Analysis Soil samples were taken nursery bag in a treatment were thoroughly mixed together. The samples were divided into four and two opposite quadrants were taken out. This was repeated and each time, another opposite quadrants was taken off until a substantial amount was obtained. The sample was then dried for four days after which it was grounded and then analysed for the amount of nitrogen, phosphorous and potassium. Soils at three growth stages (initial, midseason and late season) were considered for analysis. Other Data collected a. Plant height: This was measured using a rule; three plants were selected from each treatment replication. b. Leaf area: Five leaves from different parts of the plants were selected on each replication. The longest part along the petiole line of the leaf and the widest breath across the leaf were noted and measured as the length and breath of the leaf by using a 30 cm metre rule. The product was multiplied by a factor of 0.75 to get the leaf area. c. Mean number of fruits per treatment: The number of fruits per treatment was determined by counting the number of harvested fruits. Mean fruit size: Mean fruit size per plant was determined by using a veneer calliper to transversely measure the breath. d. Mean fruit weight: The number of fruits produced by each of the selected plants under each treatment was weighed by the use of an electronic analytical balance. Reference crop
evapotranspiration rate and rainfall reading: Evaporation rate and amount of rainfall readings were obtained from the US Class A evaporation pan and a rain gauge respectively situated at the farm where the experiment was conducted. To obtain the reference crop evaporation. 0.8 was chosen as the pan factor because the experimental location had a moderate wind speed of 2-3 ms-1 and high humidity of 75- 79%. tical analysisStatis Data collected were subjected to analysis of variance and the means were separated by Duncan?s Multiple Range Test at a probability level of 0.0f According to Iwena (2002), hot pepper requires 1000 to 1500mm of water during the growing season. According to FAO (1999) however, when the crop is grown extensively under rain fed conditions, high yields are obtained with rainfall of 600mm to 900mm, well distributed over the growing season. Huguez and Philippe (1998) also indicate that the total water requirements are 750mm to 900mm and up to 1250mm for long growing periods and several pickings. Agodzo et al. (2003) indicate that the crop water requirements range between 300mm - 700mm depending on the climatic condition and the season of the crop and the location. Grimes and Williams (1990) also asserted that water requirement for hot pepper per growing season ranges between 400 mm and 500 mm depending on the season of planting and the climatic conditions prevailing in the area. The results obtained from this study show that when the crop is given its full water requirement, 526.7 mm of water is required, but a figure of 212.4 is required when deficit irrigation of up to 40% of the crop water requirement is applied. Comparing these values to those
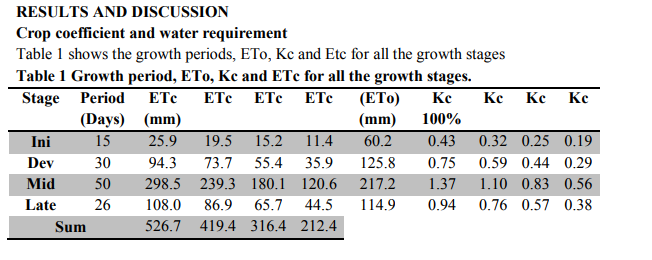
NPK levels Soil NPK levels for the initial, mid-stage as well as the last stage are shown in Figure 1. obtained by other experimenters, it can be concluded that water requirement for pepper in the Cape Coast area compares well with results obtained Agodzo et al. (2003). This figure is however lower than those obtained with the other researchers. In terms of the crop coefficient, Freeira and Goncalves (2005) obtained 0.3, 1.22 and 0.65. According to FAO (1999) Kc is 0.4 following transplanting, 1.1 during full cover and 0.9 at time of harvest. The crop coefficient (Kc) is affected by a number of factors, which include: the type of crop, stage of growth of the crop and the cropping pattern (Allen and Smith, 1998). Doorenbos and Pruitt (2000) indicated that plant height and total growing season influence crop coefficient values. The higher the plant height and the longer the growing season the higher the crop coefficient values and vice versa. In this study, Kc was 0.43, 0.75, 1.37 and 0.94 for initial, development, midseason and the late season stages respectively. These values compare quite well with those obtained by FAO (1999) where the water requirement was 600 mm for the growing period of 120 days.
Adequate amount of water in the soil tend to enhance aeration and this according to Cline and Erickson (1956), would improve potassium and nitrogen uptake. Shapiro et al. (1956) indicated that translocation of phosphorus increases when there is improvement in aeration. The results obtained from the experiment indicate that T1 utilised the most N, but T2 the most P. With regards to K, utilisation was greatest under T1.
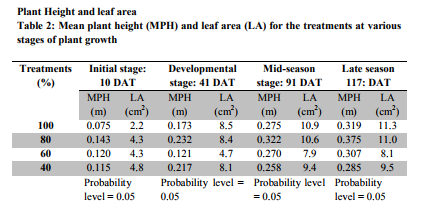
At the end of the growth period, mean plant height was in the order T2>T1>T3>T4 and mean plant height in the order T1>T2>T4>T3. Interestingly however, in both cases the results were not significantly different at the 5% probability level in spite of the large differences in amount of water applied. Pepper leaves photosynthesize more efficiently when water is abundant, resulting in a higher percentage of large, heavy, marketable fruits (Alvino et al., 1994). Under water stress, the products of photosynthesis are fewer; fruit growth and development are inhibited, and yield is decreased (Bray, 1997). Chlorophyll destruction is quickened by moisture stress (Alberte et al., 1997). More severe and prolonged water stress may result in poor flower-cluster development and reduced pistil and pollen viability and subsequent fruit set (Falcetti et al., 1995). Following fruit set, severe water stress may cause flower abortion and cluster abscission, possibly associated with hormone changes (During, 1986). Uncorrected water stress during this stage of development may result in reduced canopy development and, consequently, insufficient leaf area to adequately support fruit development and maturation. Interestingly however, there were no significant differences in leaf area in spite of the water stress imposed.
Yield components
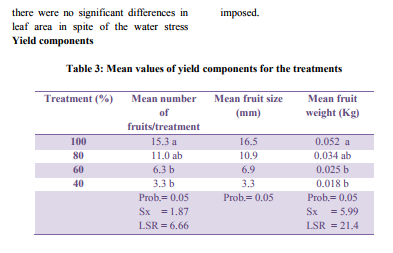
Treatment means followed by the same letter are not significantly different at 5% probability level. The relationships between crop yield and water use are complicated. Yield may depend on e timing of water application or on the amount applied. Information on optimal scheduling of limited amounts of water to maximize yields of high quality crops are essential if irrigation water is to be used most efficiently (Anaç et al., 1997). Timing, duration and the degree of water stress all affect crop yield.
Mean number of fruits
It can be seen from Table 3 that T1 produced the highest mean number of fruits (15.3) followed by T2 11.0 fruits, then T3, 6.3 fruits, and lastly T4 with 3.3 fruits . There was no significant difference between T1 and T2, but T1 differed significantly from T3 and T4. T2 was however not significantly different from T3 and T4. According to Fisher et al. (1985) in an experiment conducted, highest yields were obtained from highest regulated irrigation regime and lowest yield was obtained from the lowest irrigation water applied. Reducing irrigation water application by 40% resulted in 30% decrease in marketable fruit yield. As long as soil moisture is maintained throughout the growing season the roots will be able to maintain an adequate flow of water to the leaves to maintain growth. At the midseason and the late season stages, T1 one utilized available nitrogen effectively (see Figure 1) which may have influenced influenced the highest number of fruits formed since nitrogen is a component of amino acids and proteins and so forms essential part of protoplasm, enzymes which is stored stored food for fruit development. Factors that could be responsible for the low fruit numbers include blossom drop, a situation whereby all cells and tissues at the distal and blossoms end of the plants stems fail to receive enough moisture to maintain their body, grow and develop, and so leads to cell breakdown , flower abortion and its subsequent drop (Berrie et al., 1990). These were observed on some treatment levels at varying degrees, and were highest especially under T4 four where the drought stress coupled with the higher night temperatures favoured flower failure and this could be responsible for lowest number of fruits obtained. The results of the study are in agreement with the results of the study by Pill and Lambeth (1980) who observed a reduction in the fruit number with decreasing soil water, explaining that lower soil moisture could result in pollen and stigma dehydration as well as unnecessary elongation of the flower?s style which could result in up to 50% reduction in fruit setting and final fruit yield. Fruit weight T1 produced the heaviest fruits weighing 0.052 Kg. This was followed by T2 weighing 0.034 Kg while T3 recorded a mean weight of 0.025 Kg and the lowest mean fruit weight was recorded by T4 which was 0.018 Kg. T 1 was not significantly different from T 2 but was significantly different form T 3 and T 4. However, T2, T3 and T4 were not significantly different from each other. Plants can make virtually everything they need from water and air with a few nutrients that the roots absorb from the soil. The plant uses sunlight to split water into hydrogen and oxygen. It discards the oxygen as a waste product. The plant uses the hydrogen to make sugar from carbon dioxide in the air. Plants use oxygen in the air to burn sugar and make energy to live. The sole purpose of the leaves is to harvest light and make sugar (Longstroth, 1996) When the rate of photosynthesis is reduced as a result of reduced amount of water, the sensitive phytochrome pigments (chlorophyll pigmentation) that intercepts light for the process is affected then plants subjected to drought stress should be expected to have small and light fruits weights (Pill and Lambeth, 1980)
Mean fruit size
The results in the third column of Table 3 indicate that no significant differences exist in the mean fruit sizes at the various treatments levels. Pill and Lambeth (1980) investigated the effect of water on plants and concluded that water stress is capable of restricting plants to achieve their full genetic potential. As noted by Longstroth (1996) the early period of fruit growth is very important in determining final fruit size. For about a month after bloom the fruit grows by cell division. Later, the fruit grows by cell enlargement. So, two factors influence fruit size, cell number and cell size. Bigger fruits have more cells, so the final fruit size is determined in the month after bloom. Lack of water reduces the growth of new shoots and leaves. This means that there is less sugar to be used for fruit growth hence smaller sizes. Their work support the findings made in this study. Plants that were given full water application yielded the largest fruits while the opposite was true for treatment four which received the least amount of water applied.
Conclusion
The most important finding in the experiment was the crop water requirement and crop coefficient values for the various growth stages of hot pepper which was determined using irrigation interval of two days. At 100% water application (full irrigation), crop coefficient for hot pepper was determined to be 0.43, 0.75, 1.37 and 0.94 for initial, development, mid-season and the late season stages respectively and the total amount of water applied for the 121 days was 526.7 mm. It is also important to note that reducing water application by 20% has no significant reduction on the yield of hot pepper but above this has adverse effect on the plant and yield as indicated by treatment four recording the lowest yield of 3.3. As a result, 20% reduction in water application could be recommended for deficit irrigation in hot pepper production.
References:
References
1. Agodzo SK, Huibers FP, Chenini F, van Lier JB, Duran A. (2003) Use of wastewater in irrigated agriculture. Country studies from Bolivia, Ghana and Tunisia. Volume
2: Ghana. Wageningen: WUR: 36pp 2. Alberte, R. S., Thornber, J. P. and Fiscus E.C. (1997) Water stress, effects on the content and organization of chlorophyll in the mesophyll and bundle sheath chloroplast. Plant Physiology 59:351
3. Anaç, M.S., Ali Ul, M., Tuzal, I.H., Anac, D., Okur B. & Hakerlerler, H.1999. Optimum irrigation schedules for cotton under deficit irrigation conditions. In: C. Kirda, P. Moutonnet, C. Hera, D.R Nielsen, (eds.) Crop Yield Response to Deficit Irrigation. p. 196-212. Dordrecht, The Netherlands, Kluwer Academic Publishers
4. Allen, R. and Smith, M. (1998) Crop evapotranspiration: guidelines for computing crop water requirements. FAO Irrigation and Drainage Paper No. 56. Rome, Italy.
5. Alvino, A., Centritto, M. and Lerenzi, F. (1994) Photosynthesis response of sunlight and shade pepper leaves at different positions in the canopy under two water regimes. 21:377-391.
6. AVRDC (2006) Report. Asian Vegetable Research and Development Centre, Shanhua, Tainan, Taiwan. (ROC), pp. 298-301
7. Berrie, G. K., Berie, A. and Eze, J. M. (1990). Berrie, G.K (1990) Tropical plant science.2nd ed. Longman group UK
8. Bray E.A. (1997). Plant responses to water deficit. Trends Plant. Sci. 2: 48–54 9
. Broeshart, H., Haunold, E. and Fried, M. (1965). The effect of water conditions and oxidation status of rice soils on the availability of soil and fertilizer phosphate. Plant and Soil 23, no. 3. Pp305-313
10. Cline, R. A. and Erickson, A. E. (1959). The effect of oxygen diffusion rate and applied fertilizer on the growth, yield and chemical composition of peas. Soil Sci. Soc. Amer. Proc 23:331-335.
11. Cobbley, L.S. and Steele, W.M. (1979). An introduction to Botany of tropical crops.2nd ed. Longman, London. pp. 245-248
12. Doorenbos, J. and Pruitt, W.O. (1979). The mechanism of regulation of „Bartlett? pear fruit and vegetative growth by irrigation withholding and regulated deficit irrigation. Journal of American Society of Horticultural Science 111:904.
13. During, H. (1986). ABA and water stress in grapevines. Acta Horticulture 179:413
14. Fereres, E., F. Orgaz, and F.J. Villalobos (1993). “Water Use Efficiency in Sustainable Agricultural Systems.” International Crop Science (1993): 83-89
15. Falcetti, M., Stringari, G., Bogoni, M. & Scienza, A. 1995. Relationships among pedo-climatic conditions, plant available water and nutritional status of grapevines. Acta Horticulturae 383: 289-297
16. FAO ( 2003). Agriculture, food and water. Rome.
17. FAOSTAT. (2001). FAO Rome
18. Fisher, K.H., Cline, R.A., and Bradt, O.A. (1985). The effects of trickle irrigation and training systems on the performance of concord grapes. Drip/Trickle Irrigation in Action 1:220-230.
19. Trout, T.J. 2009. Use Of Crop Canopy Size To Estimate Water Requirements Of Vegetable Crops. Natural Resources Research Update (NRRU). Update # 241617
20. Grimes, D.W., and L.E. Williams. 1990. Irrigation effects on plant. water relations and ... Seedless grapevines. Crop Sci. 30:255-260.
21. Iwena, O.A., 2002. Essential Agricultural Science for Senior Secondary Schools. Tonad Publishers Ltd. Ikeja, pp: 55-63.
22. Levetin, E. and Mcmahon, K. (1999) Plant and society. 2 ed. Mc GrawHill Boston WCB pp. 279-281
23. Longstroth, H. (1996) Effect of soil moisture and nitrogen on plant growth, mineral composition and productivity of bell pepper (Capsicum frutescens). American Journal of Agronomy and Horticulture 21:13-17
24. Pill, W.G. and Lambeth, V.N. ( 1980). Effects of soil water regime and ... yield, water relations, and elemental composition of tomato. J. Am. Soc. Hort. Sci. 105, pp. 730– 734.
25. Shapiro, R. E., Taylor, G. S., and Volk G. W., (1956). Soil oxygen contents and ion uptake by corn. Soil Sci. Soc. Amer. Proc 20:193-197.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License