IJCRR - 3(9), September, 2011
Pages: 42-51
Print Article
Download XML Download PDF
AMELIORATION OF L-ASPARAGINASE PRODUCTION BY ASPERGILLUS ORYZAE USING PINE APPLE PEEL AS SUBSTRATE
Author: A.R. Soniyamby, S. Lalitha, B.V. Praveesh
Category: General Sciences
Abstract:The production of enzymes by bioprocesses is a good alternative to add value to agro industry residues. The aim of this work was to utilize different agro industrial byproducts as a substrate for L-asparaginase production and optimize the fermentation condition in solid state fermentation using Aspergillus oryzae. Production parameters were optimized as incubation period (96 hours), initial moisture content of 40% with inoculum size of 3%. Incubating temperature of 30 \?C and pH 6 was found to be the best for Lasparaginase production. Among different carbon source supplemented, glucose showed enhanced enzyme production (41.33 \?0.80). Addition of soy bean also increased the L-asparaginase production (45.51\? 1.18). There were no reports on production of L-asparaginase by using pine apple peel as substrate.
Keywords: L-asparaginase, Solid state fermentation, Aspergillus oryzae, pine apple peel.
Full Text:
INTRODUCTION
Therapeutic enzymes have a broad variety of specific uses: as oncolytics, thrombolytics or anticoagulants and as replacement for metabolic deficiencies. A major potential therapeutic application of enzymes is in the treatment of cancer. L-asparaginase has proved to be particularly promising for the treatment of acute lympbocytic leukemia. It action depends upon the fact that tumour cells are deficient in aspartate-ammonia ligase activity, which restricts their ability to synthesize the normally non-essential aminoacid, L-asparagine. Therefore, they are forced to extract it from body fluids. The action of asparaginase does not affect the functioning of normal cells. Which are able to synthesize enough for their own requirements, but reduces the free exogenous concentration, and so induce a state of fatal starvation in the susceptible tumor cells [1]. This fact suggested that the development of this enzyme as a potent antitumor or antileukemic drug. L-asparaginase is broadly distributed among the plants, animals and microorganisms. The microbes are better sources of L-asparaginase, because they can be cultured easily and the extraction and purification of L-asparaginase from them is also convenient, facilitating the large-scale production. Wide range of bacteria, fungi, yeast, actinomycetes and algae are very efficient producers of L-asparaginase [2]. It‘s presence in guinea pig serum was first reported by Clementi [3]. Large number of microorganisms that include Erwinia carotovora [4], Pseudomonas stutzeri [5], Pseudomonas aerugenosa [6] and E. coli [7, 8]. It has been observed that eukaryotic microorganisms like yeast and fungi have a potential for asparaginase production [9, 10]. For example, the mitosporic fungi genera such as Aspergillus, Penicillium, and Fusarium, are commonly reported in scientific literature to produce asparaginase [11- 13]. L-Asparaginase is produced throughout the world by submerged fermentation (SF). This technique has many disadvantages, such as the low concentration production, and consequent handling, reduction, and disposal of large volumes of water during the downstream processing. Therefore, the SF technique is a cost intensive, highly problematic, and poorly understood unit operation [14]. Solid-state fermentation is a very effective technique as the yield of the product is many times higher when compared to that in SF [15], and it also offers many other advantages [16]. This paper deals with the production of Lasparaginase from Aspergillus oryzae through solid state fermentation by using different agroindustrial by-products as a substrate. And also optimize the fermentation conditions to enhance the enzyme production.
MATERIALS AND METHODS
Microorganism
Aspergillus oryzae sp isolated from soil sample from Nagercoil, Tamilnadu, India was used for the production of L-asparaginase.
Screening substrate for L-asparaginase production by Solid state fermentation
The basal fermentation medium contained: 0.2 % (w/v) glucose, 1% (w/v) L-asparagine, 0.152 % (w/v) K2PO4, 0.052 % (w/v) KCl, 0.052 % (w/v) MgSO4. 7H2O. 0.003 % (w/v), pH 6.2. The different substrate (collected agro industrial waste, 10 g) was washed and dried was added to flask, moistened (15%) with fermentation medium and sterilized at 121 ºC for 20 min. After cooling the flasks were inoculated 1 ml of Aspergillus oryzae spore suspension (106 spores/mL) and incubated at 30 °C for 4 days. Enzyme extraction was performed by adding 100 ml of 0.1 M phosphate buffer to solid mouldy in a rotatory shaker (100 rev/ min) for 60 min. the extracts were squeezed through a cloth and clarified by centrifugation at 10,000×g for 15 min. the supernatants were assayed for Lasparaginase activity. The agro industrial waste used as substrate include: Ground nut oilcake(GNOC), sugarcane bagasse (SB), cotton seed oil cake (CSOC), orange peel (OP), corn flour (CF), pine apple peel (PP), maize stover (MS), lemon peel (LP), corn cob (CC) and tamarind shell (TS).
L-asparaginase assay
L-asparaginase activity was measured following method of Imada et al. [13]. This method utilizes the determination of ammonia liberated from Lasparagine in the enzyme by the Nessler‘s reaction. Reaction was started by adding 0.5 ml supernatant into 0.5 ml 0.04 M L-asparagine and 0.5 ml 0.05 M tris (hydroxymethyl) aminomethane (tris HCl) buffer, pH 7.2 and incubated at 37 ºC for 30 min. the reaction was stopped by the addition of 0.5 ml 1.5 M trichloro acetic acid (TCA). The ammonia released in the supernatant was determined calorimetrically by adding 0.2 ml Nessler‘s reagent into tubes containing 0.1 ml supernatant and 3.75 ml distilled water and incubated at room temperature for 10 min, and absorbance of the supernatant was read using a UV visible spectrophotometer at wavelength of 450 nm. One unit of asparaginase is the amount of enzyme which catalyzed the formation of 1 µmol of ammonia per min at 37 ºC. The reaction mixture from each strain was assayed in triplicate.
Optimization of the culture condition for Lasparaginase production
Various process parameters that enhance the yield of L-asparaginase by Aspergillus oryzae under solid state fermentation were investigated by taking one factor at a time. The impact of incubation time (0-168 h), initial moisture content of the substrate (20-80 % v/w), inoculum concentration (1-6ml) and incubation temperature (25-50°C) pH (3-8) was studied. Moreover, the effect of incorporation of additional carbon sources (maltose, glucose, fructose, soluble starch, lactose, and sucrose at 1%w/v), nitrogen sources (peptone, yeast extract, beef extract, soy bean, sodium nitrate and ammonium sulphate at 1% w/v) were also studied. All the experiments were conducted in triplicate and the mean values are considered.
Statistical analysis
All the experiments were carried out in triplicate, the values presented in the graphs are those of the mean of three independent experiments and the error bars indicate standard deviation.
RESULTS AND DISCUSSION
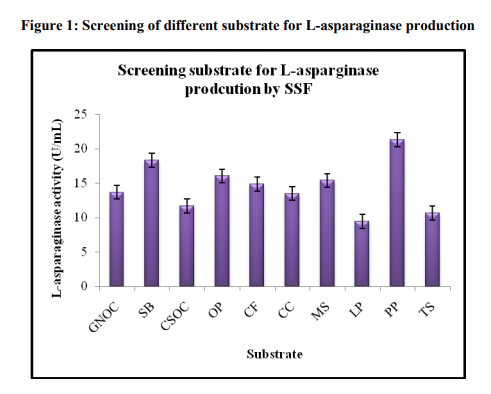
Screening of potential substrate for Lasparaginase production
The selection of an ideal agro-industrial residue for enzyme production in a SSF process depends upon several factors, mainly related with cost and availability of the substrate material, and thus may involve screening of several agroindustrial residues [17]. Among all the substrate, the maximum Lasparaginase production (21.29± 0.90) was observed with pine apple peel (Figure 1). Agroindustrial residues are generally considered the best substrates for the process of enzyme production. Several naturally occurring agricultural byproducts such as wheat bran, coconut oil cake, ground nut oil cake, rice bran, wheat and paddy straw, sugar beet pulp, fruit pulps and peel, corn cobs, saw dust, maize bran, rice husk, soy hull, sago hampas, grape marc, coconut coir pith, banana waste, tea waste, cassava waste, aspen pulp, sweet sorghum pulp, and starch could be used in one or the other industrial bioprocess for the production of value added products through SSF [18, 19].
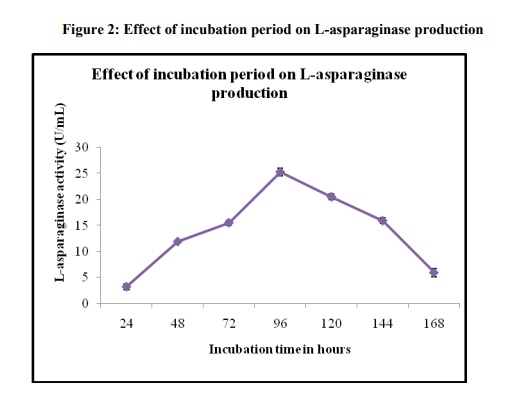
Effect of Incubation period on Lasparaginase production
The results on the optimum incubation period require for maximum L-asparaginase production showed in Figure 2 that the enzyme production started after 24 h of incubation and progressively increased with time, the maximum production of 25.28±0.70 was observed after 96 h incubation. Thereafter, the enzyme production started decreasing. Decreased enzyme yield on prolonged incubation could also be due to inhibition and denaturation of the enzyme [20].
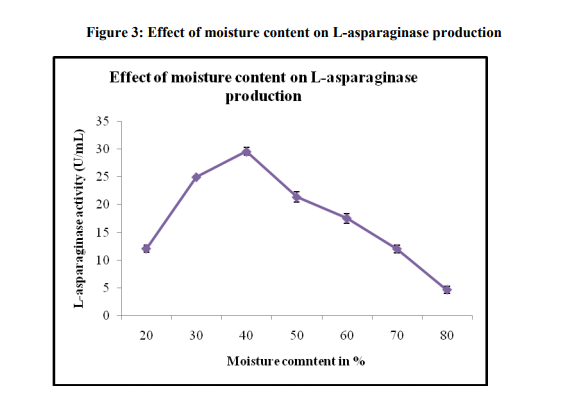
Effect of moisture content
The effect of moisture content on Lasparaginase production using pine apple peel as a substrate was examined (Figure 3). Maximum production of 29.54±0.68 was observed at 40 % of initial moisture content. Water is present in very limited amount in the SSF system and thus an optimum content is important as it determines the productivity of a SSF process [16]. Moisture content in SSF system can vary due to evaporation of the existing water through metabolic heat evolution, water consumption and liberation through fungal metabolism and also due to environmental factors. The moisture content in the substrate also depends on the type of microorganisms and the substrate used in the SSF. At the same time, the amount of moisture content also varied depending on the water binding characteristics of the substrates [21]. The optimum L-asparaginase was observed at 40 % which indicated that the water binding capacity of pine apple peel was high.
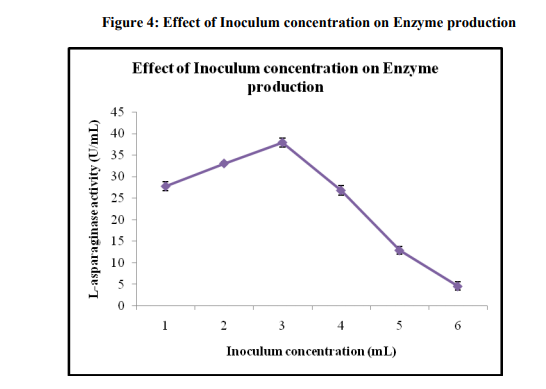
Effect of inoculum concentration
Maximum enzyme production (37.93 ± 1.02) was found at 3 ml (106 spores/mL) of inoculum size (Figure 4). Lower inoculum size required longer time for the cells to multiply to sufficient number to utilize the substrate and produce enzyme. An increase in the number of spores in inoculum would ensure a rapid proliferation and biomass synthesis. After a certain limit, enzyme production could decrease because of depletion of nutrients due to the enhanced biomass, which would result in a decrease in metabolic activity [22]. A balance between the proliferating biomass and available nutrient would yield an optimum at which the enzyme synthesis would be maximum [23].
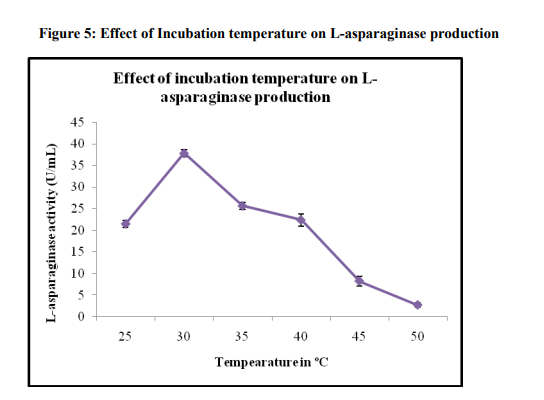
Effect of Incubation temperature
The effect of fermentation temperature in the range of 25-50 ºC was examined on the production of L-asparaginase. Temperature plays an important role in SSF processes as it significantly affects the germination of spores. However, as the spore germinates, the optimum temperature for mycelia propagation may change [24]. The temperature shift is made more complex considering SSF generated substantial amount of heat throughout the process which will affect the water content in the SSF system. As in the case of Aspergillus oryzae, the optimum temperature was found to be at 30 ºC with the maximum L-asparaginase activity of 37.84±0.78U/mL (Figure 5). Similar result was also reported by Mishra [25] for the production of L-asparaginase at 30 ºC by Aspergillus niger. The significance of incubation temperature in development of a biological process is such that it could determine the effects of protein denaturation, enzyme inhibition, cell viability and death [26]. Incubation at lower temperature resulted in longer time to the maximum enzyme activity. Incubation at higher temperature affected the fungus harmfully, which reflected on the enzyme synthesis. Since enzyme is a secondary metabolite produced during exponential growth phase, the incubation at high temperature could lead to poor growth and thus a reduction in enzyme yield [27].
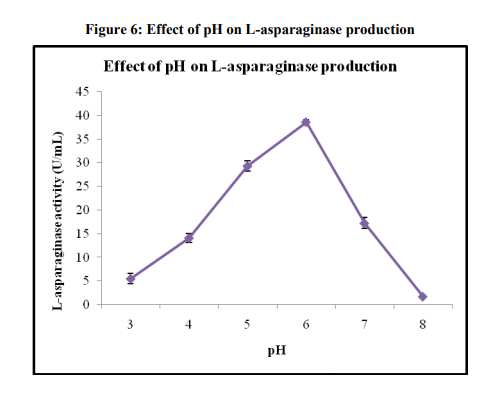
Effect of pH on enzyme production
The L-asparaginase activity at various initial pH (3-8) of the fermentation media is given in Figure 6. It was observed from Figure 6 that the maximum enzyme activity of 38.58±0.52 U/mL was obtained at the pH 6. As the initial pH was increased from 3 to 6 the L-asparaginase activity was found to increase and it was found to maximum for initial pH of 6. The activity was found to decrease for further increase in initial pH beyond 6. The optimum initial pH 6 was chosen for further studies. Similar result was reported in the literature of Baskar and Renganathan [28]. They got maximum Lasparaginase production at pH 6.
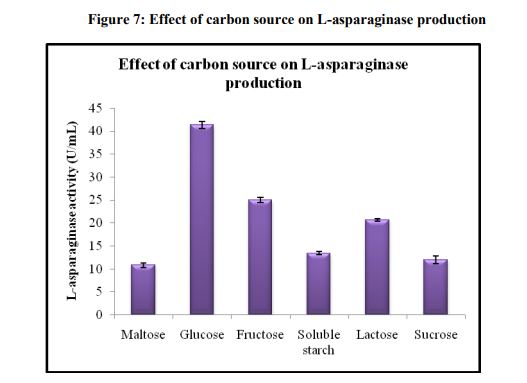
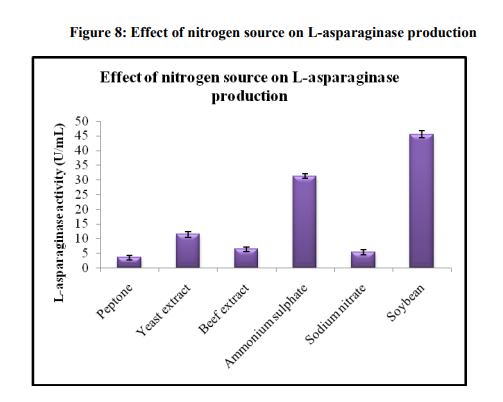
Effect of carbon and nitrogen source
L-asparaginase production depends on the availability of both carbon and nitrogen sources in the medium. Both have been shown to have regulatory effects on enzyme synthesis. Different carbon sources were used as additional carbon sources for the enhancement of asparaginase activity by Aspergillus oryzae sp (Figure 7). Additional carbon source at the level of 1 % was added to the pine apple substrate. Maximum production of asparaginase (41.33±0.80) was observed when glucose was used as carbon source. Glucose was optimized as the best carbon source for asparaginase activity. Baskar and Renganathan [28] reported maximum L-asparaginase production using glucose as best carbon source. As indicated in Figure 8, the maximum enzyme activity of 45.51±1.18U/mL was obtained when soy bean was used as nitrogen source. However, the asparaginase production was appeared to suppress in the presence of beef extract, peptone and sodium nitrate. With all the above optimized factors, the production of L-asparaginase with SSF was 45.51±1.18 U/mL. The optimum factors in the fermentation medium include pine apple as substrate, 40% moisture content, temperature 30 ºC and 96 hrs of incubation period, glucose and soy bean as carbon and nitrogen source respectively.
CONCLUSION
These studies showed that pine apple peel could be a good substrate for L-asparaginase synthesis by Aspergillus oryzae. Incubation period, initial moisture level of the medium, incubation temperature and inoculum size influenced the Lasparaginase production greatly. The optimum incubation period, initial moisture level, incubation temperature and inoculum size were 96 hours, 40%, 30 °C respectively and 3ml (106 spores/mL) of inoculum size. The supplement of glucose and soy bean favored the enzyme formation markedly.
ACKNOWLEDGEMENT
The authors are thankful to Chancellor, Advisor, Vice Chancellor and Registrar of Karpagam University for providing facilities and encouragement. We also extend our thanks to Co-ordinator and lab in charge of Karpagam University Instrument Facility Lab (KUIF).
References:
1. Sabu A. Source, properties and applications of microbial therapeutic enzymes. Indian Journal of Biotechnology 2003; 2:334-341.
2. Savitri NA, Wamik A. Microbial Lasparaginase: A potent antitumour enzyme. Indian Journal of Biotechnology 2003; 2: 184-194.
3. Clementi A. Arch. Intern. Physiol 1922; 19:369.
4. Cammack KA, Marlborangh DI, Miller DS. Biochem. J, 1972; 126-361.
5. Mannan S, Sinha A, Sadhukhan R, Chakrabarty SL. Purification, characterization and antitumor activity of Lasparaginase isolated from Pseudomonas stutzeri MB-405. Curr Microbiol 1995; 30: 291-298.
6. Abdel-Fatteh Y, Olama ZA. L-asparaginase produced by Pseudomonas aeruginosa in solid state culture: evaluation and optimization of culture conditions using factorial designs. Process Biochem 2002; 38: 115-122.
7. Qin M, Zhao F. L-asparaginase release from Escherichia coli cell‘s with aqueous twophase micelles, systems. Appl. Biochem. Biotahnol 2003; 110 (1): 11-21
8. Ghasemi Y, Ebrahiminezhad A, Amini S R, Zarrini G, Ghoshoon MB, Raee MJ, Morowvat MH, Kafilzadeh F , Kazemi A. An Optimized Medium for Screening of LAsparaginase production by Escherichia coli. American Journal of Biochemistry and Biotechnology 2008; 4 (4): 422-424.
9. Wade HE, Robinson HK, Philips BW. Asparaginase and Glutaminase a ctivities of bacteria. J. Gen. Microbiol 1971; 69: 299- 312.
10. Pinheiro IO, Araujo JM, Ximenes ECPA, Pinto JCS, Alves TLM. Production of Lasparaginase by Zymomonas mobilis strain CP4. Biometerial and Diagnostic BD06 2001; 243-244.
11. De-Angeli LC, Pucchiari F, Russi S, Tonolo A, Zurita VE, Cialanti E, Perin A. Nature 1970; 225-550.
12. Arima K, Igarasi S, Nakahama K, Isonom M. Production of extracellular Lasparaginases from microorganisms. Agricul. Biol.Chem 1972; 36: 356-361.
13. Imada A, Igarasi S, Nakahama K, Isono M. L-asparaginase and glutaminase activities of Microorganisms. Journal of General Microbiology 1973; 76:85-99.
14. Datar R. Economic of primary separation steps in relation to fermentation and genetics engineering. Process Biochem; 1986; 21: 19-26.
15. Arima K. Microbial enzyme production in Global Impact of Applied Microbiology, M.P. Starr (eds.), John Willey, New York, USA. 1964; pp. 279-299.
16. Lonsane BK, Ghildyal NP, Budiatman S, Ramakrishnan S V. Engineering aspects of solid-state fermentation. Enzyme Microb. Technol . John Willey, New York, USA. 1985; 7: 228-256.
17. Pandey A, Soccol CR, Nigam P, Brand D, Mohan R, Roussos S. Biotechnological potential of coffee pulp and coffee husk for bioprocesses. Biochem Eng. J. 2000; 6: 153- 162.
18. Pandey A, Soccol CR, Nigam P, Soccol VT. Biotechnological potential of agroindustrial residues. I: sugar cane bagasse. Bioresource Techno 2000; 74: 69-80.
19. Paranthaman R Vidyalakshmi R, Murugesh S, Singaravadivel K. Optimisation of Fermentation Conditions for Production of Tannase Enzyme by Aspergillus oryzae Using Sugarcane Baggasse and Rice Straw. Global Journal of Biotechnology and Biochemistry 2008; 3 (2): 105-110.
20. Gautam P, Sab, A, Pandey A, Szakacs G, Soccol CR. Microbial production of extracellular phytase using polystyrene as inert solid support. Bioresour. Technol 2002; 83: 229-233.
21. Pau HS, Omar IC. Selction and optimization of lipase production from Aspergillus flavus USMA10 via Solid state fermentation (SSF) on rice husks and wood dusts as substrate. Pak. J. Biolo. Sci 2004; 7: 1249-1256.
22. Kashyap P, Sabu A, Pandey A, Szakacs G. Extra-cellular Lglutaminase production by Zygosaccharomyces rouxii under solidstate fermentation. Process Biochem 2002; 38: 307–312.
23. Ramachandran S, Patel AK, Nampoothiri KM, Francis F, Nagy V, Szakacs G, Pandey A. Coconut oil cake–a potential raw material for the production of α-amylase. Bioresource Technol 2004; 93: 169–174
. 24. Raimbault M, Alzard D. Culture method to studyfungal growth in solid state fermentation. Eur. J. Appl. Microbiol. Biotechnol 1980; 9: 199-209.
25. Mishra A. Production of L-Asparaginase, an Anticancer Agent, From Aspergillus niger Using Agricultural Waste in Solid State Fermentation. Applied biochemistry and Biotechnology 2006;135:33-42.
26. Pandey A, Soccol, CR, Rodriguez-Leon JA, Nigam P. Solid state fermentation in biotechnology. New Delhi: Asia Tech Inc 2001; pp.237.
27. Sabu A, Sarita S, Pandey A, Bogar B, Szakacs G, Soccol CR. Solid-State Fermentation for Production of Phytase by Rhizopus oligosporus. Appl. Biochem. Biotechnol 2002; 102-103: 251-260.
28. Baskar G, Renganathan S. Optimization of Media Components and Operating Conditions for Exogenous Production of Fungal L-asparaginase. Chiang Mai J. Sci 2011; 38(2): 270-279.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License