IJCRR - 4(12), June, 2012
Pages: 55-78
Date of Publication: 22-Jun-2012
Print Article
Download XML Download PDF
IN-VITRO OPTIMIZATION AND BIOTECHNOLOGICAL ASSESSMENT OF MUTANTS, HYBRID AND WILD STRAINS OF TWO WHITE-ROT FUNGUS, LENTINUS SUBNUDUS AND L. EDODES CULTURED BY SUBMERGE FERMENTATION (SMF)
Author: Majolagbe, O.N, Oloke, J.K., Deka Boruah, H.P
Category: General Sciences
Abstract:Lentinus subnudus was picked in the wild during the rainy season in the month of June and then transferred aseptically onto Potato Dextrose Agar (PDA) slant by tissue culture protocol. The optimal culture conditions for biomass and exopolysaccharide production was investigated. Growth requirements
of the fungus were optimized for carbon, nitrogen, carbon-nitrogen ratio, seed culture volume and initial
pH. Temperature condition was maintained at 25oC at 100 rpm for 5 days. Each of the culture arameters investigated gave significant increament on biomass and exopolysaccharide production. For Coptimization,the highest biomass and exopolysaccharide was produced by starch (6.56gl-1, 1.84gl-1)
followed by glucose (2.01gl-1, 3.28gl-1) respectively. Tryptone and peptone N2-sources gave the highest
biomass and exopolysaccharides as 2.44gl-1 and 3.06gl-1 respectively. The optimal C:N ratio for biomass and exopolysaccharides was found to be 1:? (2.75gl-1) and 1:1/5 (5.26gl-1) respectively. Highest biomass and exopolysaccharides were also produced at pH 8.5 (1.90 gl-1) and pH 5.5 (2.00gl-1) respectively. There was no significant difference in the biomass and exopolysaccharides produced when different volume of the substrate was used. Mutants and hybrid strains of L. subnudus were produced by exposure to ultraviolet radiation and by crossbreeding with L. edodes respectively. Three (3) of the mutants that gave higher yield and better performance were selected for further biochemical analyses and then compared with the wild and hybrid varieties. Biochemical analyses such as total protein, total sugar, total phenol, total ascorbic acid content, reducing and non-reducing sugar, starch contents, anti-oxidant capacities of the wild, mutants and hybrid were performed. From our result, the mutants gave higher production and performance as compared to the wild and hybrid strains.
Keywords: optimization, biomass, exopolysaccharides, Lentinus subnudus, L. edodes
Full Text:
INTRODUCTION
Mushrooms are the fruit bodies of macroscopic, filamentous and epigeal fungi made up of hyphae which form interwoven web of tissue known as mycelium in the substrate upon which the fungus feeds; most often their mycelia are buried in the soil around the root of trees, beneath leaf litters, in the tissue of a tree trunk or in other nourishing substrate (Ramsbottom, 1989; Wilkinson and Buezaeki, 1982). Mushrooms belong to the class basidiomycetes in the order Agaricales whose fleshy fruit bodies and hymenia are borne on gills. They are ubiquitous group of fungi with many uses. They appear in traditional art work in form of drawings on textile material (Adenle, 1985).
Several authors like Fasidi and Kadiri, (1990a and 1993), Zoberi (1972 and 1973), Kadiri (1991), Oso (1975) and Nicholson, (1989) have shown in their various works that edible mushrooms are rich in ascorbic acid, amino acids, protein, minerals, glycogen, sugar and that protein is their most abundant nutrient. Also, mushrooms are sought for because of their toughness, meaty taste, desirable flavour and medicinal value. They are used extensively, as food item and for medicinal purposes (Oso, 1977., Chu et al., 2002., Akpaja, et al., 2003., Chiroro, 2004., Okhuoya, et al., 2005) and due to their high economic value and the enzymes they produce. Lentinus subnudus also known as L. squarrosulus is an highly-prized Nigeria whiterot mushroom, which can be picked in the wild during the rainy season between the month of April - August. It could be easily identified by its tough texture of matured sporophores, velvety stipe and funnel shaped whitish pileus (Jonathan, 2002) and belong to the group of the Basidiomycetes (Noda, 1998); family: Polyporaceae and Order: Polyporales. L. subnudus and other varieties of mushrooms are known to grow on a wide variety of substrates and habitats. Many species of Lentinus have been reported to live in nature and can be cultivated on special substrate and can be cultivated on pasteurized substances (Morias et al., 2000; Philippousis et al., 2001). Moreover, L subnudus have been successfully cultivated on cassava peels, rice straw, Andropogon straw, hard wood species such as Chlorophora excelsa, Spondias mombin, Terminalia ivorensis, and T. superba (Kadiri and Arzai, 2004; Adesina, et al., 2011). Optimization of industrial mushroom production depends on improving the culture process (Larraya, et al., 2003). A range of parameters including temperature, light, carbon dioxide concentration, humidity and pH have been shown to influence carpophore production Wessels, et al., 1987). Fruiting may also be stimulated by mechanical injury and chemical treatments (Hibbett, et al., 1994.). The effect of factors such as spawn grain, culture medium, oil type and rate on the culture of Psathyrella atroumbonata and L. squarrosulus has been reported (Nwanze, et al., 2004a, Nwanze, et al., 2005a; Nwanze, et al., 2005b). In our research, mutants and hybrid of L. subnudus were produced by exposure to ultraviolet radiation ( = 280nm) and crossbreeding with L. edodes respectively. Optimal culture conditions for the wild were investigated while biochemical analyses were performed for comparison of higher yield and performance among all the fungal strains.
MATERIALS AND METHODS
Organism and culture conditions
L. subnuduswt wild type (SWT) was collected from green vegetation environment growing on a decaying mango log of wood of six months. The fruiting body of the plugged fungus was washed with 90% ethanol, and then dissected longitudinally through the gill with a sharp and sterile razor blade. The mycelium was picked and transferred aseptically onto a freshly prepared Potato dextrose agar (PDA) slants. It was maintained on slants by sub-culturing in every one month interval. The ambient temperature for culture of the fungus is 25-28oC and it takes a minimum of 72 hours for optimum mycelia elongation. The fully grown fungus was maintained at 40C until when needed for use. Lentinula edodeswt wild type (EWT) was collected from Mushroom Research Centre, Himachal Pradesh Solan India. Screening of standard media for higher yield and performance Five fungal broths were used for culturing L. subnuduswt to screen for the best media with high yield performance and productivity in terms of biomass production, exopolysaccharide and enzymatic activities. Four out of the five different liquid media were used were supplied by Hi-Media Laboratory Private Ltd. Vadhani, Ind. Est. LBS Marg, Mumbai India while the other consist of a broth medium formulation according to Kwong et al., 2009. The ingredients formulation and culture conditions of each of the culture media are as stated below: Czapek Dox Broth (CDB): Sucrose (30g/l), Sodium nitrate (3.00g/l), Dipotassium phosphate (1.0g/l), Magnesium sulphate (0.5g/l), Potassium Chloride (0.5g/l), Ferrous Sulphate (0.01g/l); pH 7.3±0.2 at 25oC Mycological Broth (MB): Papaic digest of Soyabean meal (10.0g/l), Dextrose (40.0g/l), pH 7.0±0.2 at 25oC Potato Dextrose Broth (PDB): Potatoe infusion (200.00g/l), Dextrose (20.00g/l), pH, 5.1±0.2 at 25oC Yeast Mannitol Broth (YMB): Yeast Extract (1.00g/l), Mannitol (10.00g/l), Dipotassium phosphate (0.50g/l), Magnessium sulphate (0.20g/l), Sodium chloride (0.10g/l), Calcium carbonate, (1.00g/l), pH 6.8±0.2 at 25oC. Yeast Magnessium Potassium (YMK) broth: Glucose (20.0g/l), Yeast Extract (1.0g/l), MgSO4. 7H2O (1.0g/l), KH2PO4 (2.0g/l), pH 6.5±0.2 at 25oC Optimization of culture conditions for biomass, exopolysaccharide and enzymatic activities The flask culture experiments were performed in a 250 ml Erlenmeyer flask containing 100 ml of the seed culture medium. The medium was inoculated with 6mm agar plug of the fungus. The following optimal culture conditions were investigated for biomass, exopolysaccharide and enzyme activities. Carbon sources To find a suitable carbon source for biomass, exopolysaccharide and enzymatic activity of L. subnudus various carbon sources at a concentration of 10g/l were monitored for 5 days at 25oC, 100rpm on a rotary shaker incubator. The following carbon sources were used for the investigation: mannitol, starch, glucose, lactose, maltose, sucrose, fructose, mannose, galactose, arabinose, molasses and xylose. Control set-up was carbon source free in the seed culture medium.
Nitrogen Sources
To investigate the effect of different nitrogen sources on biomass, exopolysaccharide and enzymatic activity of the fungi, nitrogen compounds based on inorganic and organic nitrogen were added individually to N-source free medium at a concentration of 1g/l. Eight different N-sources were used as follows: Yeast extract, Malt extract, Beef extract, Peptone, Tryptone, NaNO3, NH4Cl, (NH4)2SO4. The experiment was set-up for 5 days at 25oC, 100rpm on a rotary shaker incubator. Nitrogen source-free medium was used as control. Carbon-Nitrogen ratio In order to investigate the effects of carbon-tonitrogen ratio (C/N) ratio on mycelial growth, exopolysaccharide and enzymatic activity of Lentinus subnuduswt, the concentration of the carbon and nitrogen in g/l were varied as follows: 1:0.1, 1:0.2, 1:0.25 1:0.33, 1:0.5. A carbon and nitrogen free media (0:0) was used as control. Initial pH To determine the optimal pH for mycelial growth, exopolysaccharide and enzymatic activity the fungus was cultivated under the initial pH ranges of 2.0 - 10.0. Experiment was carried out in 250ml Erlenmeyer flasks containing 100ml of seed culture medium on a rotary shaker incubator. The initial pH was controlled using 2 N NaOH and 2 N HCl. Volume of seed culture The effect of different volume of seed culture was investigated by culturing the fungus in 50 ml, 100 ml, 150 ml, 200 ml, 250 ml and 300 ml.
The 50 ml, 100 ml, 150 ml were carried out in 250 ml flask while the 250 ml and 300 ml experiment was performed in 500 ml flask. Harvest and Quantification of Biomass and Exopolysaccharides from culture broth (a) Biomass: The mycelia biomass was harvested from the seed culture on the 5th day of cultivation by filtering with 0.45 diameter Whatman Filter paper and the wet weight determined. The wet biomass was dried at 60oC until a constant weight was obtained (Kwon et al., 2009). (b) Exopolysaccharide (eps): Exopolysaccharides was derived from the liquid culture broth by 99% (v/v) acetone precipitation method, stirred vigorously and left overnight at 4 oC in a refrigerator. The precipitated crude eps was recovered by centrifugation at 10,000 rpm for 10 mins and then filtered with 0.45µm Whatman filter paper. The precipitated eps was oven dry to a constant weight and the weight of the polymer estimated. Production of Mutants Lentinus subnudusmt mutant types (SMT) were produced by exposing an actively growing culture (5days old) of the fungus on Potato Dextrose Agar (PDA) plate to an ultravioletradiation ( = 280nm) at different time durations of 15 minutes interval for 4 hrs. Mycelia plugs obtained from the culture were transferred onto the centre of a fresh PDA plates, incubated at 25oC. Production of Hybrid by crossbreeding L. subnudusht hybrid type (SHT) was produced by cross-breeding L. subnuduswt (SWT) and L. edodeswt (EWT) as follows: EWT was cultured on PDA plate and incubated at 25oC. On the fourth day after inoculation, SWT was inoculated onto the same plate at a distance of 8mm apart with the culture conditions remaining the same. The junction where the two fungal strains intersect was picked with a sterilized cutter and then inoculated onto a freshly prepared PDA media. The new fungal strain obtained was used as the hybrid type. Comparison and screening of mutants for higher yield performance in biomass, exopolysaccharide and protein contents A total of eighteen (18) fungal strains were cultured in the optimized culture conditions as follows Glucose 10g/l, Yeast Extract 10g/l, K2HPO4 0.5 g/l, CaCO3 1.0 g/l, MgSO4.7H2O 0.2g/l and NaCl 0.1g/l. The initial pH of the medium was adjusted to 5.5 using 2N NaOH and 2N HCl. Equal size of each fungal agar plugs kept on PDA slants were inoculated into 100 ml substrate volume, cultured for 5 days at 25oC. The wet and dry weight of the biomass from each culture flask was determined as earlier discussed above. The exoploysacharides was obtained by acetone precipitation method as already mentioned. The EPS was lyophilized and the dry weight estimated. The extracellular protein was determined according to Lowry et al., (1951). SMT060, SMT-120 and SMT-135 were selected out of the eighteen mutants based on biomass, exopolysaccharides and protein production levels. Biochemical analysis i. Total Protein estimation Total protein was estimated by FolinCiocalteau's method as modified by Lowry et al., (1951) using bovine serum albumin as standard. The blue colour developed was measured at 660 nm against the blank and standard solution of protein was also run simultaneously. ii. Enzyme Activity assay Two extracellular lignocellulotic enzymes (Pyranose 2-oxidase and Laccase) associated with white-rot fungus were extracted and assayed accordingly as follows: a. Determination of Pyranose 2-oxidase Activity assay Pyranose 2- oxidase (P2OX) activity was determined using the chromogen ABTS [(2,2- azinobis(3-ethylbenzthiazolinesulfonic acid)] (ε420 = 3.6 x 104 mM-1 cm-1 ) according to the modified method of Danneel et al., 1993. The standard 1 ml assay mixture contains 10mM ABTS, 2U of Horseradish Peroxidase Type II Sigma, 100 μmol D-glucose, in 10 mM sodium phosphate buffer (pH 6.5), 100µl of the diluted enzyme was used. The mixture was incubated at 30oC in Water-bath B-480 (manufactured by Buchi, Switzerland) for 3mins. Oxidation of ABTS was monitored by spectrophotometer at 420nm. b. Determination of Laccase Activity assay: Laccase activity assay was determined according to the modified method of Shin and Lee, 2000; Saranyu and Rakrudee, 2007 as follows: The routine assay for laccase was based on the oxidation of [(2,2-azinobis(3- ethylbenzthiazoline-6-sulfonate) diammonium salt] (ABTS) (Sigma) (ε420 = 3.6 x 104 mM1 cm-1 ). 50µl of the enzyme was incubated in 940µl of 0.1 M Sodium acetate buffer (pH 4.5) containing 10µl of 10mM ABTS incubated at 30oC in Water-bath B-480 for 10mins. The reaction mixture was stopped by adding 50µl of 50% (w/v) Trichloroacetic acid (TCA). Oxidation of ABTS was monitored by spectrophotometer at 420nm. iii. Estimation of total sugar content of the exopolysaccharides The total sugar contents (TSC) of the exopolysaccharides produced from the submerge cultures of wild, mutants and hybrid fungal strains were estimated using Anthrone method with glucose as standard, according to Sadasivam and Manickam 1992. Briefly, 100mg of the sample was weighed into a boiling tube, hydrolyzed in boiling water for 3h with 5ml of 2.5 N HCl. The sample was cooled to room temperature and then neutralized with solid sodium carbonate until effervescence ceases. The volume was made up to 100ml and then centrifuged. The supernatant was collected and 0.5 and 1 ml aliquots taken for analysis. The volume was made to 1 ml in the tubes by adding distilled water. 4 ml of anthrone reagent was added to the tubes heated for 8 minutes in a boiling water bath. The sample tubes were cooled rapidly and the green to dark green colour read at 630 nm. iv. Estimation of starch content by Anthrone method 0.1g of the sample was weighed and homogenized in hot 80% ethanol to remove the sugars. This was centrifuged and the residue was retained. The residue was washed repeatedly with hot 80% ethanol till the washings did not give colour with anthrone reagent. The residue was dried well over a water bath. 5.0 ml of water and 6.5 ml of 52% Perchloric acid was added to the residue. This was centrifuged and the supernatant saved. The extraction process was repeated using fresh Perchloric acid and then centrifuged. The supernatant was pooled and made up to 100 ml with distilled water.1 ml aliquot of the sample was used for analysis.. 4 ml of anthrone reagent was added, heated for 8 minutes in a boiling water bath. The tubes were cooled rapidly and the intensity of green to dark green colour read at 630 nm. v. Estimation of reducing and non-reducing sugars by DNS method Reducing and non-reducing sugar components of the exopolysaccharides were determined using Dinitrosalicyclic acid (DNS) reagent as follows: 100mg of each sample was weighed and the sugars were extracted with 5ml of hot 80% ethanol twice. The supernatant was collected and evaporated on water bath. 10ml of water was added to dissolve the sugars. 1ml aliquot of each of the sample was taken for analysis and the volume made up to 3 ml in testtube. Each sample was prepared in triplicate. 3ml of DNS reagent was added and mixed together. The solution was heated for 5mins in a boiling water bath. After the colour has developed, 1ml of 40% Rochelle salt solution was added when the contents in the tube were still warm. The tubes were cooled under running tap and the absorbance was measured at 510nm. The amount of reducing sugar in each of the sample was calculated using glucose as standard. vi. Determination of total phenols in the exopolysaccharides Total soluble phenols were estimated using Folin-Ciocalteu reagent (FCR) with catechol as standard (Malick and Sigh 1980). The blue coloured complex was taken at 650nm against a reagent blank. vii. Estimation of Ascorbic acid using 2, 6- dichlorophenol indole phenol titration method 5ml of the working standard was taken with a pipette into a 100ml conical flask. 10ml of 4% oxalic acid was added and this was titrated against the dye. 1g of each of the sample was extracted in 4% oxalic acid and the volume made up to 100ml. This was then centrifuged at 10,000 rpm for 15 mins. 5 ml of the supernatant was taken and 10ml of 4% oxalic acid added. This was titrated against the dye. The end point was the appearance of pink colour which persisted for a few minutes. The amount of the dye consumed is equivalent to the amount of the ascorbic acid. viii. Determination of antioxidant activities of the exopolysaccharides. Methanol and dichloromethane extractions were used for determining hydrophilic and lipophylic antioxidant activities of the exoploysaccharides (Arnao et al., 2001). Sample Extraction (a) Methanol extraction 1g of the eps was mixed with 10ml methanol and then homogenized using vortex. The homogenate were kept at 4oC for 12 hours and then centrifuged at 10,000 rpm for 20 mins. The supernatants were recovered and stored at -20oC until analysis. (b) Dichloromethane extraction The pellet obtained from the methanol extract was re-dissolved with 10ml dichloromethane, and homogenized for anti-oxidant activity. The homogeneate was centrifuged at 10,000 rpm for 20 mins. The supernatants were recovered and stored at -8 oC until further analysis. Briefly, 150µl of each of the extract for each sample was mixed with 2850 µl of the ABTS. It was kept for 2 hours in a dark condition. Absorbance was taken at 734nm using spectrophotometer. The standard curve was obtained using Trolox and results were expressed in µM Trolox Equivalent (TE)/g fresh weight. Statistics Analysis of the data was performed by one-way analysis of variance (ANOVA), One-Sample ttest and Karl Pearson Correlation coefficient was determined. Differences at P < 0.05 were considered to be significant. Analysis was done using Ms Excel Sheet 2007, Origin Lab 8 and Graph Pad Prism 5 statistical softwares. Each experiment was repeated three times with three replicates each. RESULTS AND DISCUSSION Submerge culture of L. subnuduswt (SWT) in different standard media Our first attempt was to culture SWT in five different media by sub-merge fermentation for biomass, exopolysaccharide, protein production and enzyme assay. Among all the five media used, YMKB gave the highest biomass (60±6.39mgl-1 ) and exopolysaccharide (960±10.83 mgl-1 ) production and least in CDB which yielded 19.50±4.58 mgl-1 and 100±20.53 mgl-1 biomass and exopolysaccharide respectively, although higher exopolysaccharide was also recorded by MB as shown in Table 1. Total protein estimated was highest in YMB (1.734±0.93 mgml-1 ) and least in PDB (0.186±0.05 mgml-1 ). There were significant differences (P < 0.05) between the biomass, exopolysaccharide and protein produced among the five broth media used. Previous researchers already reported that submerge culture obviously give rise to potential advantages of higher mycelial production in a compact space and shorter time without significant problem of contamination (Bae et al., 2000; Park et al., 2001; Sinha et al., 2001). Enzyme assay done showed highest laccase and Pyranose 2-oxidase activity in CDB and YMKB as 13.345 Uml1min-1 and 12.87 Uml-1min-1 respectively (Table 1). Effect of different carbon sources In order to determine the suitable carbon source for higher biomass and exopolysaccharide production, SWT was cultured in broth media, each containing various carbon sources. Each of the carbon sources was added to the synthetic media at a concentration of 1% (w/v) and then cultured for five days. The fungus showed a good growth response to most of the carbon sources as compared to the control experiment in which no carbon source was added. As shown in Fig. 1, starch gave the highest biomass production (6.56gl-1 ) followed by dextrose (2.01gl-1 ) among all the twelve carbon sources investigated. This is contrary to previous report on the optimization for mycelial biomass and exopolysaccharide production in submerge cultures of Cordyceps militaris (Jeong, et al., 2009). For exopolysaccharide production, the highest yield was recorded in molasses (3.96 gl1 ), followed by maltose (3.68 gl-1 ) as carbon sources. Although, glucose has already been reported by some previous investigators as a good carbon source for biomass and exopolysaccharides production in submerge cultures (Xu et al., 2003; Nour El-Dein et al., 2004; En-Shyh and Shu-Chiao, 2006), our result shows that L. subnudus was able to utilize both monosaccharides and disaccharides sugar compounds for biomass and exopolysaccharides production. This result is in accordance with nutritional requirement for biomass and exopolysaccharide production in several mushrooms (Sudhakaran and Shewale 1988; Fang and Zhong, 2002b; Ji-Hoon et al., 2004; Jeong, et al., 2009). Lactose and arabinose gave the lowest biomass and eps yield as 0.15 gl-1 and 0.16 gl-1 respectively. This result is in consonance with that of previously reported investigators where arabinose gave lowest biomass yield (Madunagu 1988, Jonathan and Fasidi, 2001), although contrary to report by Gbolagade et al (2006). In order to further understand the relationship between biomass and exopolysaccharide produced as a result of various carbon sources used, Pearson coefficient of correlation (r) was worked out. The outcome shows that there is no correlation between biomass and exopolysaccharide (r = - 0.087), suggesting that, both products are independent of each other although there was significant difference among the values obtained (P < 0.05). We also observed in our results that mushrooms may show different growth responses to carbohydrate utilization as compared to results already reported by other researchers. Effects of different nitrogen sources To investigate the effect of different nitrogen sources on biomass and exopolysaccharides production in submerge culture of L. subnudus, eight nitrogen sources were used consisting of five organic and three inorganic nitrogen sources. The basal medium was supplemented with the nitrogen sources at a concentration of 0.1% (w/v). A nitrogen limited control was also set-up. Among the nitrogen sources tested, tryptone gave the highest biomass production (2.44gl-1 ), followed by ammonium sulphate (1.36gl-1 ) and ammonium chloride (1.30gl-1 ) as shown in Fig. 2. Moderate biomass yield were recorded in beef and yeast extract, peptone and sodium nitrate with the lowest in meat extract (0.045gl-1 ). For eps production, peptone gave the highest yield (3.06gl-1 ), followed by ammonium chloride (2.66gl-1 ), with moderate yields in beef, meat and yeast extract. Reduced production of exopolysaccharide was recorded for both sodium nitrate and ammonium sulphate, whereas, tryptone gave the lowest yield (0.62gl-1 ). This means that a nitrogen source used to improve biomass production may not necessarily support a high yield of exopolysaccharides in L. subnudus. Our result reveals that organic nitrogen sources were favourable in biomass and exopolysaccharide production in sub-merge culture of L. subnudus as compared to inorganic sources. The result of which is in agreement with previous report on biomass and exopolysaccharide production in submerge fermentation of Sarcodon aspratus (Ji-Hoon, 2004), and L. tuberregium (Manjunathan and Kaviyarasan, 2011). Pearson correlation coefficient (r = 0.0084) shows that there was a low degree of correlation between biomass and exopolysaccharide produced when different Nitrogen source were used whereas on the other way round, there was a significant difference between the means of the values obtained (p<0.05). Effect of carbon-nitrogen ratio In order to determine the effects of carbonnitrogen ration (C/N) on biomass and exopolysaccharide production, of L. subnudus, the concentration of dextrose and yeast extract in the basal medium was varied. Concentration of 1% (w/v) of each of dextrose and yeast extract in the basal medium was used for ratio 1:1. Other ratios were varied accordingly as follows: 1: 0.1, 1: 0.2, 1: 0.25 1: 0.33, 1: 0.5. Highest increase in biomass was recorded in C/N ratio 1: 0.3 (2.75 gl-1 ) and least in 1: 0.1 (0.33 gl-1 ) as compared to the control without carbon and nitrogen sources (Fig. 3). The trend in the result shows that there was increase in biomass production as the C/N ratio increases up to a point after which further increase in nitrogen brought a decrease in biomass production. This result is the same as that reported in L. tubberregium (Manjunathan and Kaviyarasan, 2011). Highest exopolysaccharide was obtained in C/N ratio 1:0.2 (5.26g-1 ), and least in 1:0.5 (0.76gl-1 ). There was low correlation between the biomass and exopolysaccharide (r = 0.026) although significant effect was observed at P < 0.05 Effect of initial pH Among the environmental factors, culture pH has been known to significantly influence cellular morphology and metabolites biosynthesis (Heald and Kristiansen 1985; Salleh et al., 2008). To determine the optimal pH for biomass and exopolysaccharides production, L. subnudus was cultivated under different initial pH (2-10) in shake flask culture. Figure 4 shows that the effects of initial pH on biomass and exopolysaccharide production were variable. In these experiments, high biomass was obtained at pH 6.5 (1.79gl-1 ) and highest at pH 8.5 (1.9 gl-1 ). For eps production, high amount was recorded at pH 6.5 (1.96gl-1 ) and maximum at pH 5.5 (2.00 gl-1 ). This shows that L. subnudus can grow optimally either at subacidic and sub-basic culture medium. This result is in alignment with result already reported for biomass production in submerge cultures of C. militaris (Jeong et al., 2009). Jonathan (2002) already suggested that good growth of mushrooms could be obtained at moderately or slightly acidic pH. At lower and higher value of pH, biomass and eps obtained declined- the result of which suggest that strongly acidic and highly alkaline culture conditions were unfavourable to biomass and eps production in submerge cultures of L. subnudus. On the other hand, some other workers have recorded optimum pH 5.5 for other exopolysaccharide synthesizing fungi such as Pleurotus pulmonarius (Nour El-Dein et al., 2004). Further more, it has also been reported that many kinds of ascomycetes and basidiomycetes exhibit more acidic pH optima during submerge culturing
(Yang and Liau, 1998a,b; Lee et al., 1999; Yang et al., 2000a,b; Park et al., 2001). All of these results lead to the conclusion that there is a relationship between the degree of exopolysaccharide production, biomass and the initial pH of the culture medium. (En-Shyh Lin and Shu-Chiao Sung, 2006). There is a moderate correlation (r = 0.583) between biomass and exopolysaccharide produced as the pH of the culture medium changes. However, low correlation exists between pH and biomass (r = 0.098); pH and exopolysaccharide (r = 0.346) respectively. Effect of seed culture volume Fig. 5 shows the effect of seed culture volume on biomass and eps production. Equal size of L. subnudus agar plugs were inoculated into each of the different seed culture volume, while other culture conditions remain constant. As indicated in Fig. 5, we obtained variability in the quantitative estimate of biomass and eps produced with the highest biomass and eps obtained in the 100ml (0.095g) and 300ml (0.22g) seed culture volume respectively, although, the values were not significantly different as compared to other seed culture volume used. This suggests that, the mineral composition, and cultural conditions has a greater influence than the seed culture volume in biomass and exopolysaccharide production by L. subnudus. This is further established as there is was no correlation (r = -0.083) between biomass and exopolysccharide obtained. Generation of new hybrid by cross-breeding A new hybrid of L subnudus tagged as ?subnudus hybrid type? (SHT) was produced by cross-breeding the wild strain of the fungus (SWT) with L. edodes wild strain (EWT) which has been reported as a very important mushroom of desirable characteristics. This is the first time we are reporting the generation of a hybrid from the two wilds in the same genus. Further biochemical analysis were done using the new strain and then compared with the wild and selected mutants. Culture of wild, mutants and hybrid of L. subnudus in fungal broth Fifteen (15) mutants and one hybrid were produced as earlier discussed. Out of the fifteen mutants, the best three (SMT-060, SMT-120 and SMT-135) with good physiological and biochemical characteristics were selected as shown in Plate 1 (a-c) and then compared with L. subnuduswt (SWT), L. edodeswt (EWT) and their hybrid (SHT). We cultured the five strains in the optimized medium to investigate if there would be increase or improvements in their biomass and exopolysaccharide production in submerge fermentation. It was observed that SMT-120 gave the highest biomass (2.830gl-1 ) and exopolysaccharide (17.71 gl-1 ) among the fugal strains with the least production obtained in SWT. Extracellular proteins extracted and quantified from submerge culture of each of the fungal strains by Lowry‘s method showed that total protein estimated was highest in SMT-060 (5.187 gl-1 ) and least in SWT ( 0.734 gl-1 ) as indicated in table 2. There was significant difference (P < 0.05) in biomass, exopolysaccharide and protein produced among all the fungal strains Enzyme extraction and purification The culture filtrate of each of the wild, mutants and hybrid strain were evaluated for the production of two lignolytic enzymes namely; pyranose 2 – oxidase and laccase. The crude enzymes were fractionated with (NH4)2SO4 and enzyme assay done accordingly. Enzyme assay and activity Pyranose 2-oxidase: Pyranose 2 oxidase (P2OX) has already been reported to have roles in ligninolysis (Daniel et al., 1994; Volc et al., 1996), production of peroxide for peroxidases, and synthesis of the antibiotic cortalcerone (Baute and Baute, 1984). It is also of interest for numerous biotechnological applications, analytical methods, and synthesis of novel chemicals (Giffhorn, 2000; Theodorus et al., 2004). P2OX activity was carried out among all the five fungal strains. EWT gave the highest activity (10.14 Uml-1min-1 ) and least in SMT135 (0.821 Uml-1min-1 ), although activity was relatively higher in SMT-120 and SMT-060 (Table 2). Moreover, several wood decaying fungi in addition to L. subnudus and L. edodes have been reported to also produce P2OX (Giffhorn, 2000), including Oudemansiella mucida, Trametes versicolor (Daniel et al., 1994), Polyporus obtusus (Ruelius et al., 1968), Phlebiopsis gigantea (Scha¨fer et al., 1996), and Trametes multicolor (Volc et al., 1999). Some POX activity has also been observed in culture fluid, but this appears to be related to cell lysis (Daniel, et al., 1992). P2OX can also be extracted from decayed birch (Daniel, et al., 1994, Daniel, et al., 1992), and an extracellular activity is observed when the fungus is grown on the natural lignocellulosic substrate such as wheat straw (Vyas et al., 1994) Laccase: Although there are many reports dealing with extracellular Laccases produced by white-rot basidiomycetes and ascomycetous fungi (Leonowicz et al., 2001; Claus, 2004), in our research, attempt was made to also produce extracellular laccase from the submerge cultures of wild, mutants and hybrids of L. subnudus and compare their activity with one another. Besides, intracellular Laccase from fully browned gills of the fruiting body of L. edodes (Masaru et al., 2003), L. Panus (trigrinus) and other white-rot fungus has also been reported (Burke and Cairney, 2002; Schlosser et al., 1997; RoyArcand and Archibald, 1991). Laccases have received much attention from researchers in last decades due to their ability to oxidise both phenolic and non-phenolic lignin related compounds as well as highly recalcitrant environmental pollutants, which makes them very useful for their application to several biotechnological processes (Susana and José, 2006). Such applications include the detoxification of industrial effluents, mostly from the paper and pulp (Kuhad et al., 1997), textile and petrochemical industries (Abadulla et al., 2000; Blánquez et al., 2004; Hou et al., 2004; Rodríguez Couto et al., 2004a, 2005), medical diagnostics and as a bioremediation agent to clean up herbicides, pesticides and certain explosives in soil ((Durán and Esposito 2000; Pointing, 2001). Among the five fungal strains tested for laccase activity, EWT showed the highest enzyme activity (26.60 Uml-1min-1 ) and least activity was expressed in SMT-060 (2.16 Uml-1min-1 ) as shown in table 2. Total sugar, total phenol and total ascorbic acid of the exopolysaccharide The total sugar contents (TSC) of the exopolysaccharides produced from the submerge cultures of wild, mutants and hybrid fungal strains were estimated using Anthrone method with glucose as standard (Sadasivam and Manickam, 1992) while total phenol was estimated using Folin-Ciocalteu reagent (FCR) using catechol as standard as already explained. Ascorbic acid (which is a sugar acid and an antioxidant agent) in the exopolysaccharides was quantified using 2, 6-dichlorophenol indolephenol dye method (Sadasivam and Theymoli, 1987; Heinoen et al., 1989). Analysis of the total sugar, total phenol and total ascorbic acid contents among the five fungal strains tested is shown in table 3. EWT showed the highest total sugar content and least in SMT-060. Total phenol was highest in SMT-135 and least in SHT while total ascorbic acid was highest in SMT-060 and least in EWT, although the values are not significantly different from one another. The presence of phenol and ascorbic acid is an indication that these fungus are promising source of antioxidant which are of natural origin as already also implicated in medicinal mushroom such as Inonotus obliquus (In et al., 2007) and Kale plants (Imtiyaz et al., 2005). Phenolic compounds are known as powerful chain breaking antioxidants (Shahidi and Wanasundara, 1992). Phenols are very important plant constituents because of their scavenging ability due to their hydroxyl groups (Hatano et al., 1989). The phenolic compounds may contribute directly to antioxidative action (Duh et al., 1999). It is suggested that polyphenolic compounds have inhibitory effects on mutagenesis and carcinogenesis in humans, when ingested up to 1g daily from a diet rich in fruits and vegetables (Tanaka et al., 1998). The Folin-Ciocalteau assay has been utilized to measure polyphenols for a number of different plants, including mushrooms. A correlation has also been shown between higher antioxidant activity and larger amount of total phenolics in extracts from various commercial mushrooms (Cheung and Lee, 2000). Analysis of our results shows that there was no significant difference in the phenol produced among all the strains (P < 0.05), although there were significant differences in sugar, ascorbic acid and total protein (P < 0.05) among all the fugal strains as indicated in table 3. Determination of reducing sugar, nonreducing sugar, amount of starch of the exopolysaccharide Sugar were assayed using 3, 5-dinitrosalicyclic acid (DNS) which assays sugars by their reducing properties (Sadasivam and Manickam, 1992). The content of non-reducing sugars was obtained by subtracting the reducing sugars from total carbohydrate contents, while amount of starch of the exopolysaccharides were determined by the Anthrone method (Hodge and Hofreiter 1962; Thayumanavan and Sadasivam, 1984; Sadasivam and Manickam, 1992). Table 4 shows the percentage composition of the reducing, non-reducing and amount of starch in the exopolysccharides of the fungal strains. EWT gave the highest composition of the reducing and non-reducing sugars as 14.93 % and 50.40 % respectively although SMT-135 also yielded a very good composition of these sugars. Percentage starch composition was highest in SMT-060, although there was no significant difference in the percentage starch composition among the fungal strain (P < 0.05) Antioxidant activity determinations in methanolic and dichloromethane extract. Antioxidant activities in methanolic and dichloromethane extract was measured using 2,2-Azinobis-(3-ethyl) benzothiazolline)-6 sulphonic acid diammonium salt ABTS as earlier mentioned. Antioxidant activity was highest in dichlorometane extract especially in EWT and reduced in methanolic extract of SMT-060 although there was no significant differences (P< 0.05) among the values obtained as shown in table 5). This result is similar to previous report by researchers who reported potent antioxidant activity in Inonotus obliquus (In et al., 2007) and Pleurotus pulmonarius (Badole et al., 2008). There is considerable evidence that these antioxidants could help to prevent various diseases including myocardial and cerebral ischemia, diabetes, rheumatoid arthritis, inflammation, and cancer-initiation as well as in aging process (Coyle 1993; Margaill et al., 2005) because they have the capacity to quench free radicals. These natural antioxidants have advantage over some synthetic antioxidants such as butylated hydroxyanisola (BHA) and butylated hydroxytoluene (BHT), which have been reported to exerts toxicological effects as compared to natural antioxidants (Saito et al., 2003; Stefanidou et al., 2003). CONCLUSION All the fungal strains gave remarkable potentials in enzymes synthesis, anti-oxidants, sugar, phenol, ascorbic acid, exopolysaccharides and proteins. Therefore, the exploitation of wild, mutants and hybrid strains of L. subnudus using biotechnological tools and protocols is thereby suggested for large scale production. ACKNOWLEDGEMENTS The authors are grateful to Council of Scientific and Industrial Research (CSIR), New Delhi, India, Third World Academy of Science (TWAS), Italy for providing fund with necessary facilities. They are also thankful to Dr. P.G RaoDirector, North-East Institute of Science and Technology (NEIST), Jorhat, Assam India for his encouragement and provision of conducive research environment to carry out this work. The authors also acknowledged the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
References:
1. Abadulla E, Tzanov T, Costa, S, Robra KH, Cavaco-Paulo A, Gübitz G (2000) Decolourization and detoxification of textile dyes with a laccase from Trametes hirsuta. Appl Environ Microbiol. 66, 3357–62.
2. Adenle VO (1985) The most popular mushroom in South Western Nigeria Mushroom Newsl. Tropics 5: 20-21, in Fasidi and Kadiri, (1993). Use of agricultural waste for the cultivation of Lentinus subnudus (Polyporales: Polyporaceae) in Nigeria. Rev. Biol. Trop. 41 (3): 411-415.
3. Adesina FC, Fasidi IO, Adenipekun OC (2011) Cultivation and fruit body production of Lentinus squarrosulus Mont. (Singer) on bark and leaves of fruit trees supplemented with agricultural waste. Afr. J. Biotechnol. 10 (22), 4608-4611
4. Akpaja EO, Isikhuemhen OS, Okhuoya JA (2003) Ethnomycology and usage of edible and medicinal mushrooms among the Igbo people of Nigeria. Int. J. Med. Mushrooms, 5, 131 – 319.
5. Arnao MB, Cano A, Acosta M (2001) The hydrophilic and lipophilic contribution of total antioxidant activity. Food. Chemistry, 73, 239-244.
6. Badole SL, Thakurdesai PA, Bodhankar SL (2008) Antioxidant activity of aqueous extract of pleurotus pulmonarius (fries) quel-champ. Pharmacologyonline 2, 27-41.
7. Bae JT, Sinha J, Park JP, Song CH, Yun JW, (2000) Optimization of submerged culture conditions for exo-biopolymer production by Paecilomyces japonica. J. Microbiol. Biotechnol. 10, 482–487.
8. Baute MA, Baute R (1984) Occurrence among macrofungi of the bioconversion of glucosone to cortalcerone. Phytochemistry 2, 271–274.
9. Blánquez P, Casas N, Font X, Gabarrell M, Sarrá M, Caminal, G (2004) Mechanism of textile metal dye biotransformation by Trametes versicolor. Water Res. 38, 2166– 72.
10. Burke RM, Cairney JWG (2002) Laccases and other polyphenol oxidases in ecto- and ericoid mycorrhizal fungi. Mycorrhiza 12, 105–116.
11. Cheung PC, Lee MY (2000) Fractionation and characterization of mushroom dietary fiber
12. (non-starch polysaccharides) as potential nutraceuticals from sclerotia of Pleurotus tuberregium (Fries) singer. J. Agric Food Chem, 48, 3148-51.
13. Chiroro CK (2004) Poverty alleviation by mushroom growing in Zimbabwe. Mushroom growing for a living worldwide. Heineart Inc. Seoul, Korea, P. 298.
14. Chu KK, Ho SS Chow AH (2002) Coriolus versicolor: a medicinal mushroom with promising immunotherapeutic values. J. Clin. Pharmacol, 42, 976 – 984.
15. Claus H (2004). Laccases: structure, reactions, distribution. Micron, 35, 93-96. 16. Coyle J T (1993) Puttfarcken, P. Science. 262, 689.
17. Daneel HJ, Rossner E, Zeec A, Giforn F (1993) Purification and characterization of a pyranose oxidase from the basidomycete Peniophora gigantea and chemical analyses of its reaction products, Eur. J. Biochem. 214, 795-802.
18. Daniel G Volc J, Kubatova E, Nilsson T (1992) Ultrastructural and immunocytochemical studies on the H2O2- producing enzyme pyranose oxidase in Phanerochaete chrysosporium grown under liquid culture conditions. Appl. Environ. Microbiol. 58, 3667–3676.
19. Daniel G, Volc J, Kubatova E (1994) Pyranose oxidase, a major source of H2O2 during wood degradation by Phanerochaete chrysosporium, Trametes versicolor, and Oudemansiella mucida. Appl. Environ. Microbiol. 60, 2524–2532.
20. Duh PD, Tu YY, Yen GC (1999) Antioxidant activity of water extract of Harng Jyur (Chrysanthemum morifolium Ramat). Lebensm-Wiss. Technol.; 32: 269- 277.
21. Durán N, Esposito E (2000) Potential applications of oxidative enzymes and phenoloxidase-like compounds in wastewater and soil treatment: a review. Appl Catal B Environ; 28, 83–99.
22. En-Shyh L, Shu-Chiao S (2006) Cultivating conditions influence exopolysaccharide production by the edible Basidiomycete Antrodia cinnamomea in submerged culture. International Journal of Food Microbiology 108, 182-187.
23. Fang QH, Zhong JJ (2002b) Submerged fermentation of higher fungus Ganoderma lucidum for production of valuable bioactive metabolites– ganoderic acid and polysaccharide. Biochemical Engineering Journal 10, 61–65.
24. Fasidi IO., Kadiri M (1990a) Changes in nutrient contents of two Nigerian mushrooms, Termitomyces robustus (Beeli Heim) and Lentinus subnudus Berk during sporophore development. Die. Nahrung 34: 416-420.
25. Fasidi IO, Kadiri M (1993) Use of agricultural waste for the cultivation Lentinus subnudus (Polyporales: Polyporaceae) in Nigeria. Rev. Biol. Trop., 41(3): 411-415.
26. Gbolagade JS, Fasidi IO Ajayi EJ Sobowale AA (2006) Effect of physicochemical factors and semi-synthetic media on vegetative growth of Lentinus subnudus (Berk.), an edible mushroom from Nigeria. Food Chemistry 99, 742–747.
27. Giffhorn F (2000) Fungal pyranose oxidases: occurrence, properties and biotechnical applications in carbohydrate chemistry. Appl. Microbiol. Biotechnol. 54, 727–740
28. Hatano T, Ogawa N, Kira R, Yasuhara T, Okuda T (1989) Tannins of cornaceous plants. In Cornusiins A, B and C, dimeric monomeric and trimeric hydrolyzable tannins from Cornus officinalis, and orientation of valoneoyl group in related tannins. Chem Pharm Bull (Tokyo) 37, 2083-90.
29. Heald PJ, Kristiansen B (1985) Synthesis of polysaccharide by yeast-like forms of Aureobasidium pullulans Biotechnol. Bioeng. 27, 1516-1519.
30. Heinoen MI, Linkola EK, Varo PT, Koivistoinen PE (1989) J. Agric Food Chem. 37, 655.
31. Hibbett DS, Tsuneda A Murakami S (1994) The secotioid form of Lentinus tigrinus: Genetics and development of a fungal morphological innovation. Am. J. Bot. 81, 466-478.
32. Hodge JE, Hofreiter BT (1962) In: Methods in Carbohydrate Chemistry (eds. Whistler, R.L and BeMiller, J.N.), academic Press, New York.
33. Hou H, Zhou J, Wang J, Du C Yan B (2004) Enhancement of laccase production by Pleurotus ostreatus and its use for the decolourization of anthraquinone dye. Process Biochem 39, 1415–9
34. Imtiyaz M, Beigh GM, Tanveer AS, Amjad H, Athar AK, Charanjit K (2005) Antioxidant activity and Total phenolic content of Kale Genotypes Grown in Kashmir Valley. J. Plant Biochemistry and Biotechnology 14, 215-217
35. In KL, Young SK, Yoon WJ, Jin YJ Bong SY (2007) New anti-oxidant polyphenols from the medicinal mushrooms Inonotus obliquus Bio-organic and Med. Chem. Lettr. 17, 6678-6681.
36. Jeong S, Jong K, Lee S, Won C, Keun S, Lee E, and Eock KH (2009) Optimization of Culture Conditions and Medium Components for the Production of Mycelial Biomass and Exo-polysaccharides with Cordyceps militaris in Liquid Culture Biotechnology and Bioprocess Engineering, 14, 756-762.
37. Ji HJ, Jong ML, Hyun OK, Sang WK, Hye JH, Jang WC, Jong WY (2004) Optimization of submerged culture conditions for exopolysaccharide production in Sarcodon aspratus (Berk) S.lto TG-3. World Journal of Microbiology and Biotechnology 20, 767–773.
38. Jonathan SG, Fasidi IO (2001) Effect of carbon, nitrogen and mineral sources in growth of Psythyrella atroumbonata (Pegler), a Nigerian edible mushroom. Food Chemistry, 50, 397–401.
39. Jonathan SG (2002). Vegetative growth requirement and antimicrobial activities of some higher fungi in Nigeria. Ph.D thesis, University of Ibadan, Ibadan, Nigeria.
40. Kadiri M (1991) The effects of chemical soaking of substrate raw materials on the mycelia growth and fructification of Lentinus subnudus Berk. Mushroom J. Tropics, 11: 53-58.
41. Kadiri M, Arzai AH (2004) Cultivation of Lentinus subnudus Berk (Polyporales) on woodlogs. Bioresource Technology. 94, 65- 67
42. Kuhad RC, Singh A, Eriksson KEL (1997) Micro-organisms and enzymes involved in the degradation of plant fiber cell wall. In: Eriksson KEL, editor. Biotechnology in the Pulp and paper industry. Advances in biochemical engineering biotechnology. Berlin:Springer Verlag; Chapter 2.
43. Larraya LM, Alfonso M Pisabarro AG Ramirez L (2003) Mapping of genomic regions (quantitative trait loci) controlling production and quality in industrial cultures of the edible basidiomycete Pleurotus ostreatus. Appl. Environ. Microbiol. 69. 3617-3625.
44. Lee KM, Lee SY, Lee HY (1999). Bistage control of pH for improving exopolysaccharide production from mycelia of Ganoderma lucidum in an air-lift fermentor. J. Biosci. Bioeng. 99, 646–650.
45. Leonowicz A, Cho NS, Luterek J, Wilkolazka A, Wojtas-Wasilewska M, Matuszeska A, Hofrichter M, Wesenberg D Rogalski J (2001) Fungal laccase: properties and activity on lignin. J. Basic Microbiol 41, 185–227.
46. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with Folin phenol reagent. Journal of biological chemistry. 193: 265-275.
47. Madunagu BE (1988) Collection and studies on cultivation of Pleurotus squarrrosulus (Mont.). Singer. Nigerian Journal of Science, 22, 51–55.
48. Malick CP, Sigh MB (1980) In Plant Enzymology and Histo-enzymology, Kalyani Publications, New Delhi, p. 286.
49. Margaill I, Plotkine K, Lerouet D (2005) Free Radical Biol. Med., 39, 429.
50. Masaru N, Maki K, Hisayuki W, Machiko O, Kumiko S, Toshikazu T, Katsuhiro K Toshitsugu S (2003) Important roles of fungal intracellular laccase for melanin synthesis: purification and characterization of an intracellular laccase from Lentinula edodes fruit bodies Microbiology 149, 2455–2462.
51. Morias HI, Ramos AC, Matou N, Santous EJ (2000) Note: production of shiitake mushroom (Lentinus edodes on lignocellulosic residues. Food. Sci. Technol. Int. 6. 123-128.
52. Nicholson RA (1989) Common mushrooms found in Akwa Ibom state. Nigerian Field. 54: 9-32.
53. Noda S (1998) A preparation for kidney treatment possessing anti-inflammatory activity, obtained from Basidiomycetes, e,g Lentinus, Pleurotus, Flammulina, and Tricholoma J P Patent 8511888.
54. Nour-El-Dein MM, El-Fallal AA, El-Shahat AT, Hereher FE (2004) Exopolysaccharides production by Pleurotus pulmonarius: factors affecting formation and their structures. Pak. J. Biol. Sci. 7, 1078–1084.
55. Nwanze PI, Khan AU, Ameh JB, Umoh VJ (2004a) The effect of various grains, culture media, oil type and rate on stipe lengths and diameters, wet and dry weights and pileus diameters of Psathyrella atroumbonata. ROAN 1and2 85-97.
56. Nwanze P I, Khan AU, Ameh JB Umoh VJ (2005a) The effect of the interaction of various spawn grains with different culture media on carpophore production of Lentinus squarrosulus (Mont.) Singer. Afr. J, Biotechnol. 4, 615-619.
57. Nwanze PI, Khan AU, Ameh JB Umoh VJ (2005b) The effect of the interaction of various spawn grains with different culture media on carpophore production of Lentinus squarrosulus (Mont.) Singer. Afr. J, Biotechnol. 4, 472-477.
58. Okhuoya JA, Akpaja EO, Abbot O (2005) Cultivation of Lentinus squarrosulus (Mont) Singer on sawdust of selected tropical tree species. Int. Journal of Mushroom Scienc, 2, 41– 46.
59. Oguri S, Ando A, Nagata Y (1996) A novel developmental stage specific lectin of the Basidiomycetes Pleurotus cornucopiae. J. Bacteriol., 178, 5692.
60. Optletal l, Jahodar L, Chabot V, Zdansky P, Lukeas J, Bratova M, Solichova D, Blunden G,
61. Oso BA (1975) Mushroom and the Yoruba people of Nigeria. Mycologia 67: 311-319.
62. Oso BA (1977a) Pleurotus tuber-regium from Nigeria. Mycologia 69:271-279.
63. Oso BA (1977b) Mushroom in Yoruba mythology and medicinal practises. Econ. Bot. 31: 367-371.
64. Park JP, Kim SW, Hwang HJ, Yun JW (2001) Optimization of submerged culture conditions for the mycelial growth and exobiopolymer production by Cordyceps militaris. Lett. Appl. Microbiol. 33, 76–81.
65. Philippousis A, Zervakis G, Diamantopoulou P (2001) Bioconversion of agricultural lignocellulosic wastes through cultivation of the edible mushrooms Agrocybe aegerite, Volvariella volvoceae and Pleurotus spp. World J. Microbiol. Biotechnol. 7 (24) 191-200.
66. Pointing SB (2001) Feasibility of bioremediation by white-rot fungi. Appl. Microbiol Biotechnol; 57:20–33.
67. Ramsbottom J (1989) Mushrooms and Toadstools. London: Bloomsbury Books.
68. Rodríguez Couto S, Hofer D, Sanromán MA, Gübitz GM (2004a) Production of laccase by Trametes hirsuta grown in an immersion bioreactor. Application to decolourisation of dyes from a leather factory. Eng Life Sci; 4, 233–8.
69. Rodríguez CS, Sanromá MA, Gübitz GM (2005) Influence of redox mediators and metal ions on synthetic acid dye decolourization by crude laccase from Trametes hirsuta. Chemosphere; 58, 417– 22.
70. Roy-Arcand L, Archibald FS, (1991) Direct dechlorination of chlorophenolic compounds by laccases from Trametes (Coriolus) versicolor. Enzyme Microb. Technol 13, 194–203.
71. Ruelius HW, Kerwin, RM, Janssen FW (1968) Carbohydrate oxidase, a novel enzyme from Polyporus obtusus. I. Isolation and purification. Biochem. Biophys. Acta 167, 493–500.
72. Sadasivam S, Theymoli B (1987) In: Practical Manual Biochemistry, Tamil Nadu Agricultural University, Coimbatore p 14.
73. Sadasivam S, Manickam A (1992) In: Biochemical methods for Agricultural sciences. Wiley Eastern Limited , New Delhi, pp. 6-7; 11-12
74. Saito M, Sakagami H, Fujisawa S (2003) Anticancer Res., 23, 4693.
75. Salleh M, Tsuey M, Ariff LS (2008) The profile of enzymes relevant to solvent production during direct fermentation of sago starch by Clostridium saccharobutylicum P262 utilizing different pH control strategies. Biotechnol. Bioprocess Eng. 1: 33-39.
76. Saranyu K, Rakrudee S (2007) Laccase from spent Mushroom Compost Lentinus polychrous Lev. and its potential for Remazol Blue R Decolourization. Biotechnol. 6 (3), 408-413.
77. Scha¨fer A, Bieg S, Huwig A, Kohring GW, Giffhorn F (1996) Purification by immunoaffinity chromatography, characterization, and structural analysis of a thermostable pyranose oxidase from the white rot fungus Phlebiopsis gigantea. Appl. Environ. Microbiol. 62, 2586–2592.
78. Schlosser D, Grey R, Fritsche W (1997) Patterns of ligninolytic enzymes in Trametes versicolor. Distribution of extra- and intracellular enzyme activities during cultivation on glucose, wheat straw and beech wood. Appl Microbiol Biotechnol 47, 412–418.
79. Shin KS Lee YJ (2000) Purification and characterization of a new member of the laccase family from the white-rot basidiomycete Coriolus hirutus Arch. Biochem. Biophys., 384, 109-115.
80. Shahidi F, Wanasundara PK (1992) Phenolic antioxidants. Crit Rev Food Sci Nutr. 32:67-103.
81. Stefanidou M, Alevisopoulos G, Chatziioannou A, Kouteslinis A (2003) Vet. Hum. Toxicol. 45, 103
82. Susana RC, José LTH (2006) Industrial and biotechnological applications of laccases: A review. Biotechnology Advances 24, 500– 513.
83. Sudhakaran VK, Shewale JG (1988) Exopolysaccharide production by Nigrospora oryzae var. glucanicum. Enzyme and Microbial Technology 10, 547–551
84. Tabata K, Itoh W, Kojima T, Kawabate S, Misaki K (1981) Ultrasonic degradation of schizophyllan, an antitumor polysaccharide produced by Schizophyllum commune Fries. Carbohydr. Res. 89, 121–135.
85. Tanaka M, Kuei CW, Nagashima Y (1998) Application of antioxidative maillrad reaction products from histidine and glucose to sardine products. Nippon Suisan Gakkaishi 47, 1409-1414.
86. Thayumanavan B, Sadasivam S (1984) Qual. Plant foods Hum.Nutr. 34, 253.
87. Theodorus HK, Michael DM, Daniel C, Jill G, Philip JK (2004) Isolation and Purification of Pyranose 2-Oxidase from Phanerochaete chrysosporium and Characterization of Gene Structure and Regulation. Applied and Environmental Microbiology 70, 5794–5800,
88. Vyas BRM, Volc J, Sasek V (1994) Ligninolytic enzymes of selected white rot fungi cultivated on wheat straw. Folia Microbiol. 39, 235–240.
89. Volc J, Kuba´tova´ E, Daniel G, Prikrylova´ V (1996) Only C-2 specific glucose oxidase activity is expressed in ligninolytic cultures of the white rot fungus Phanerochaete chrysosporium. Arch. Microbiol. 165, 421– 424.
90. Wessels JGH, Mulder GH, Springer J (1987) Expression of dikaryon-specific and nonspecific mRNAs of Schizophyllum commune in relation to environmental conditions and fruiting. J. Gen. Microbiol. 133, 2557-2561
91. Wilkinson J, Buezaeki S (1982) Mushrooms and Toadstools. Glasgow. Harper Collins.
92. Xu CP, Kim SW, Hwang HJ, Choi JW, Yun JW (2003) Optimization of submerged culture conditions for mycelial growth and exo-polymer production by Paecilomyces tenuipes C240. Process Biochem. 38, 1025– 1030.
93. Yang FC, Liau CB (1998a) The influence of environmental conditions on polysaccharide formation by Ganoderma lucidum in submerged cultures. Process Biochem. 33, 547–553.
94. Yang BK, Ha JY, Jeong SC, Das S, Yun JW, Lee YS, Choi JW, Song CH (2000a) Production of exo-polymers by submerged mycelial culture of Cordyceps militaris and its hypolipidemic effect. J. Microbiol. Biotechnol. 10, 784–788.
95. Yang FC, Ke F, Kuo SS (2000b) Effect of fatty acids on the mycelial growth and polysaccharides formation by Ganoderma lucidum in shakeflask cultures. Enzyme Microb. Technol. 27, 295–301.
96. Zoberi MH (1972) Tropical Macro fungi. London. Macmillan Press Limited.
97. Zoberi MH (1973) Some edible Mushrooms from Nigeria. Nigerian Field. 38: 81-90
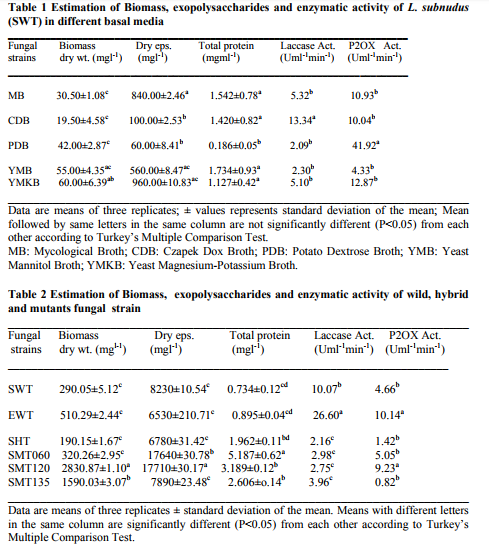
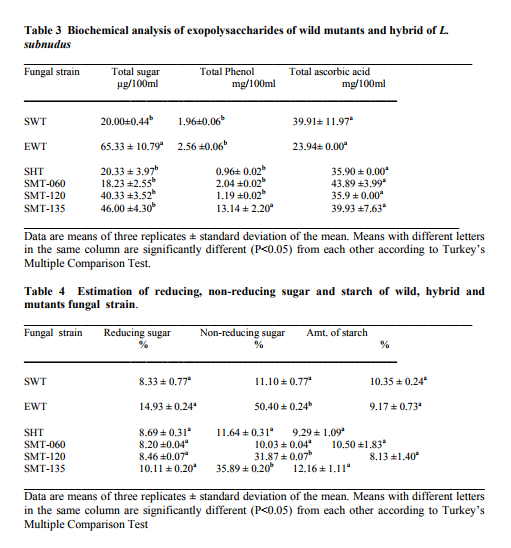
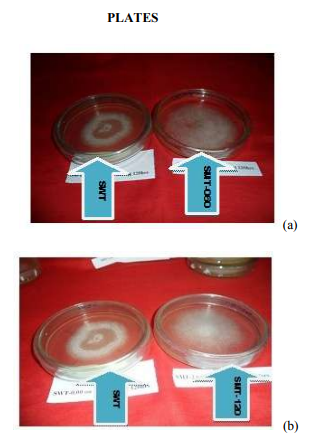

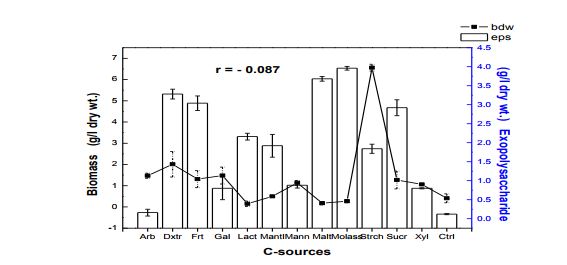
Fig. 1 Effect of different carbon sources on biomass and exopolysaccharide production in submerge culture of L. subnuduswt Data are means of three replicates; Error bar represents standard error of means of observed values; r = Pearson correlation coefficient Arb: arabinose; Dxtr: dextrose; Frt: fructose; Gal: galactose; Lact: lactose; Mantl: mannitol; Mann: mannose; Mol: molasses; Strch: starch; Sucr: sucrose; Xyl: xylose; Ctrl: control.
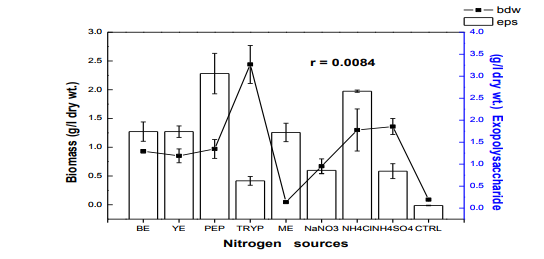
Fig. 2 Effect of different nitrogen sources on biomass and exopolysaccharide production in submerge culture of L. subnuduswt Data are means of three replicates; Error bar represents standard error of means of observed values; r = Pearson correlation coefficient BE: beef extract; YE: yeast extract; PEP: peptone; TRYP: tryptone; ME: meat extract; CTRL: control.
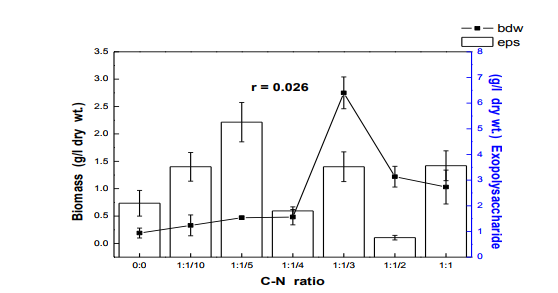
Fig. 3 Effects of carbon-nitrogen ratio on biomass and exopolysaccharides production in submerge culture of L. subnuduswt Data are means of three replicates; Error bar represents standard error of means of observed values; r = Pearson correlation coefficient
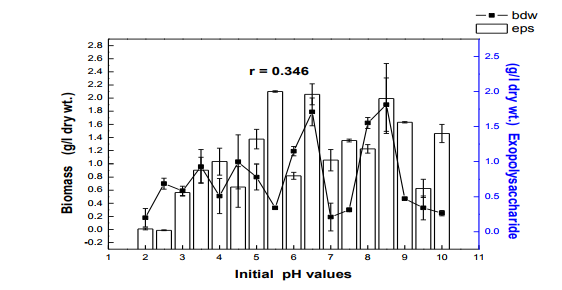
Fig. 4 Effect of initial pH on biomass and exopolysaccharide production in submerge culture of L. subnuduswt Data are means of three replicates; Error bar represents standard error of means of observed values; r = Pearson correlation coefficient

Fig. 5 Effect of seed culture volume on biomass and exopolysaccharide production in submerge culture of L. subnuduswt Data are means of three replicates; Error bar represents standard error of means of observed values; r = Pearson correlation coefficient
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License