IJCRR - 4(19), October, 2012
Pages: 15-24
Date of Publication: 15-Oct-2012
Print Article
Download XML Download PDF
EFFECT OF PROBIOTIC BACTERIA AS A BIOCONTROL AGENT AGAINST DISEASE CAUSING PATHOGEN IN CATLA CATLA (HAMILTON, 1822)
Author: R. Parthasarathy, V. Ramasubramanian, D. Ravi
Category: General Sciences
Abstract:The primary aim of the current study was to analyze and to find out the effectiveness of probiotic bacteria (Lactobacillus plantarum andBacillus megaterium) on growth performance and immuno response of Catla catla. Growth parameters like final weight, weight gain, specific growth rate, survival rate, feed intake and protein efficiency ratio were increased among Catla catla were which fed with a diet containing L.plantarum. The combined effect of selected probiotic bacteria is shows significant level of the protein content when compared with control as well as single probiotic bacteria feed. L.plantarum gave larger inhibition zone (4 cm) than B.megaterium (1.8 cm). From the present study L.plantarum had a probiotic effect in vitro and in vivo against Aeromonas hydrophila, while B.megaterium had a probiotic effect in vitro and it small extinct in the in vivo. The Red blood cell levels were gradually decreased in all treatments (T1, T2 andT3). But in the all controls (C1, C2 andC3) the RBC values were gradually increased. White blood cell values are decreased in T1, T2 andT3 treatments and controls C1, C2 andC3 there is no much differences/changes. The plasma total proteins showed significantly decreased in fish fed with diet containing L.plantarum and mixture of L.plantarum andB.megaterium. The above results show that the probiotic bacteria can eliminate the pathogenic bacteria which cause disease in fish and other aquaculture organism.
Keywords: Probiotics, Lactobacillus plantarum, Bacillus megaterium, Aeromonas hydrophila, hemorrhagic septicemia.
Full Text:
INTRODUCTION
Disease outbreaks are being increasingly recognized as significant constraints on aquaculture fields by affecting the production and trade in many countries. Among those diseases, bacterial infections are considered as the major cause of mortality in fish hatcheries and farms (Grisez and Ollevier, 1995). The selective pressure exerted on the microbial world and encourages the natural emergence of bacterial resistance. i.e. such chemotherapeutic treatment may cause the development of resistant bacteria (Aoki et al.,1985). Also the yield residues in fish and introduce potential hazard to public health and to the environment. A new approach alternative method, that is gaining acceptance within the aquaculture industry, is by use of probiotic bacteria to control potential pathogens (Gomez-Gil et al., 2000). In recent years, development of the probiotic bacterial treatment in aquaculture to improve disease resistance, water quality and growth of farmed fish (Verschuere et al., 2000). Probiotics helps to protect the host against invasion or colonization of foreign pathogen like bacteria, viruses and fungi by re – colonizing the gut with normal gut micro flora. Probiotics are critical to enhance resistance to infection and boosting immune status of the host. Probiotics are microbial cell preparations or components of microbial cells that have a beneficial effect on the health and well – being of the host (Salminen et al., 1999). Quite huge number of probiotic bacteria were been use to control many kind of diseases and also it is used to overcome any kind metabolic disorders as well as growth promoters (Swain et al., 2006). Based on the availability of many probiotic bacteria the present study reveals only for Lactobacillus plantarum and Bacillus megaterium, out of chosen probiotics (Lactobacillus acidophilus, Lactobacillus lactis, Streptococcus thermophilus, Bacillus subtillus). Aeromonas hydrophila is an opportunistic pathogen. Ventura and Grizzle (1987) and Eissa et al (1994) shows that A.hydrophila infected internal organs through the digestive tract or through uninjured skin under conditions of crowding (13.1 g of fish / L) and high temperature (24?C) (Cipriano, 2001). The author chosen the topic based on the above said importance and significant of the probiotic bacteria in controlling the disease out breaks in aquaculture farms. Therefore, the present study reveals that how to evaluate the role few of probiotic bacteria (Lactobacillus plantarum, Bacillus megaterium) as a biocontrol agent against common fish pathogen Aeromonas hydrophila, in the fish Catla catla (Hamilton, 1822) and the effects on normal micro flora and some physiological, biochemical, and immunological parameters.
MATERIALS AND METHODS
Healthy fishes (Catla catla, Hamilton, 1822) weighed between 5 – 6.5 g were obtained from Tamilnadu fisheries development corporation, Azliyar, Coimbatore District, Tamilnadu, India. And they were allowed to acclimatize the laboratory condition for 2 weeks and then used for experimental studies. Experiments were carried out in culture tubs with 30 L capacity filled with fresh, clean and unchlorinated ground water, and change of water was done once in 2 days intervals. The fishes were fed with feed composition of 35 % crude protein, crude fat 4% and crude fiber 9% which was commercially available.
EXPERIMENT SETUP/TREATMENTS
The fish were divided into 3 equal groups. Group I [Treatment 1 (T1) – fishes are fed with L.plantarum blended granular feed]; Group II [Treatment 2 (T2) – fishes are fed with B.megaterium blended granular feed]; Group III [Treatment 3 (T3) – fishes fed with L.plantarum and B.megaterium blended granular feed]; Group IV [Control (Control) – fed with normal feed]. Each treatment i.e.T1, T2, and T3 were divided into two equal numbers of fishes during the probiotic and pathogen treatments. Such as T1 and C1, T2 and C2, T3 and C3. All T1, T2, and T3 were fed with pathogen blended granular feed after 20 days of probiotic treatment. Whereas C1, C2, and C3 were fed with only respected probiotic blended granular feed. Each group contains 5 fishes. Stocking density is 5 L per fish (fish/5 L water).
Culturing of Probiotic Bacteria
In the present study Lactobacillus plantarum and Bacillus megaterium was a probiotic bacteria, which have obtained from IMTECH, Chandigarh, India. And these cultures were sub cultured in the nutrient agar slant. L.plantarum was cultured in lactic acid broth and B.megaterium was cultured in nutrient broth. After incubation period the broth culture were centrifuged and collected cells were washed twice with saline. Pellet was mixed properly with the 50 g of granular feed. Likewise prepared B.magaterium feed mixture. Mixed equal volume of both bacteria blended feed gives combination of the two probiotic bacteria (B.magaterium, L.plantarum). Fishes were fed once daily along with normal feed at a fixed feeding rate 3% (i.e. 1 – 1.5 g) of the body weight of fish. The feed given rate were adjusted at 10 days intervals after fish were weight. Each tub (fish tank) were cleaned once in 2 days interval to remove fish feces remaining feed with complete water change i.e. refilled fresh water to fixed volume. The experiment (probiotic feeding) runs for 25 days (i.e. before pathogen treatment). The growth parameters and rate of feed intake was calculated according to Tekinay and Davis (2001) method.
Inducing disease by A. Hydrophila
In this study Aeromonas hydrophila is a predominant bacterium which obtained from Department of zoology, Bharathiar University, Coimbatore and gram staining and biochemical tests are done for the culture confirmation. A.hydrophila was sub cultured in the nutrient broth (50 ml) and incubated 48 hrs at 30? C. After incubation period the broth culture were centrifuged and collected cells were washed twice with saline. Pellet was mixed properly with the feed. After 25 days of probiotic treatment pathogen (A.hydrophila), blended feed was introduced into respected treatment fishes (T1, T2, T3) via feed. Then carefully monitor the mortality rate of the fish was observed during this period. Antagonistic test The in - vitro probiotic activity was done using agar diffusion method (Muller – Hinton agar plates) and the inhibition zone was determined (Ruiz et al., 1996). Biochemical analysis of fish (Protein and Carbohydrate) The initial and final (before and after pathogen treatment) biochemical (such as protein and carbohydrate) analysis were estimated in fish Catla catla (Hamilton 1822). The protein content of fish was estimated by Folin – ciocalteau method. The carbohydrate estimation was done by anthrone method. Fish blood was drawn from the heart region by cardiac puncture by using a sterile syringe previously rinsed with EDTA as an anti coagulant. The collected blood was diluted with EDTA (2 mg/ml). Total number of Red blood cells and White blood cells in the blood were counted by haemocytometric method. Blood plasma also collected. The protein content of plasma was estimated by Folin – ciocalteau method. Preparation of bacterial antigen from A.hydrophila: A.hydrophila was cultured in the LB broth for 24 hours at 37?C. After incubation of bacterial broth culture was centrifuged. Collected the pellet was washed twice with saline. Finally pellet was redissolved in 5 ml of saline. This bacterial suspension was treated with ultrasound 20 minutes at 10 seconds intervals on ice by sonicator and then centrifuged at 16,000 rpm for 30 minutes. The supernatant was used as antigen and stored in the refrigerator. Immuno electrophoresis: The immuno electrophoresis (IEP) technique combines electrophoresis and double immunodiffusion (DID), and helps resolve an antigen mixture and identify it. Microbial analysis: The samples from skin/fin/scales and internal organs (like kidney, stomach and intestine) and gills were taken for the further microbial analysis. Purification and identification of the isolates were done using sub – culture techniques and biochemical tests (carbohydrate fermentation, Indole test, Methyl red test, VP test, starch hydrolysis, TSI agar test, H2S production test, Urea utilization test etc.) according to the Bergey’s manual (Bergey et al., 1984) Morphology of the isolates was examined using staining technique (such as Gram’s staining).
RESULTS
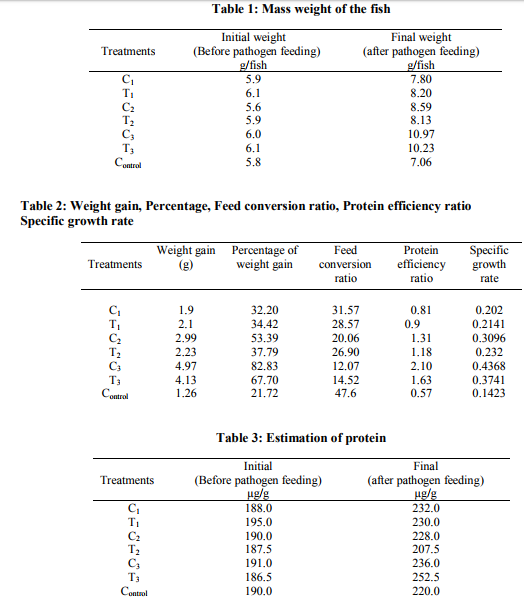
Growth/Mass weight
The parameter like weight, active feeding status were done and tabulated in Table-1. The initial growth in term of weight ranges between 5.6 to 6.1 gms. Length is also measured during initial stage, which range between 6.5 and 7.2 cms. With reference to mass weight of the fish, there is a slow and steady growth was observed in control. i.e. the initial weight of the fish shows 5.8 gm on average and over a period of 60 days the fish has been gained a weight of 1.26 gm. Then coming to probiotic bacteria feed treatment (L.plantarum, B.megaterium and combination of L.plantarum and B.megaterium). The combine treatment of L.plantarum and B.megaterium shows very high growth rate, i.e. 4.97 g was gained an average within 60 days time. And individual probiotic bacteria treatments - C2 (B.megaterium) treatment shows 2.99 gm of weight gained within the 60 days with comparing to control. C3 (combination of L.plantarum, B.megaterium) treatment gain 4.97 gms and T2 and T3 shows 2.23 gm and 4.13 gm, respectively with compared to control. In the probiotic and pathogenic treatment the pathogen (A.hydrophila) is an opportunistic bacterium, which can control by the above said probiotic bacteria, the Table 2 shows there is gradual weight gained by T3 treatment, combine probiotic bacteria along with pathogen. It clearly shows that the pathogen in presence of probiotic bacteria won’t establish the symptoms and won’t cause disease, (because only in absence of probiotic bacteria), the purely treated pathogen along with normal feed was fed for a group of fish (5 numbers), subsequently one after other within 40 days all the 5 fishes were died. Here no probiotic treatment. The feed conversion ratio (FCR), Protein efficiency ratio (PER) and Specific growth rate (SGR) was been tabulated in Table 2. The feed conversion ratio is higher in the C1 (L.plantarum treatment) followed by C2 (B.megaterium treatment). When comparing the treated and untreated fishes for the PER analysis test. Protein efficiency ratio in combined bacterial treatment shows higher ratio (>25%) more than that of control.
Biochemical analysis
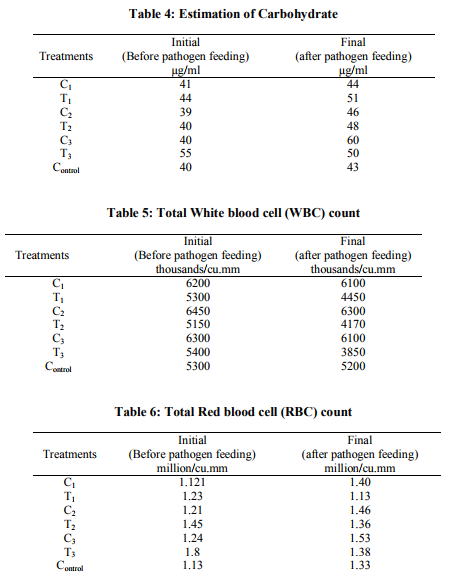
Estimation of Protein (Table 3): During the initial stages the control and treated fishes shows an average of the crude protein 190 µg/g of tissue sample and also the treatment lot shows between 189 - 191 µg/g. After the study period the final stage of the protein estimation was done. The protein concentration for different treatments is varying when compared with controls. In control initial quantum of protein was 190 µg/g of tissue. The same treatment fishes after 60 days with control feed shown 220 µg/g, the probiotic bacteria (L.plantarum and B.megaterium and combined L.plantarum and B.megaterium) shown a slight improvement with compared to the control one. i.e. it was estimated that 236 µg/g was been observed in C3 (combined treatment of L.plantarum and B.megaterium). Then coming to probiotic bacteria and pathogen treatments L.plantarum and combined probiotic bacteria treatment are shown almost the same results. B. megaterium was shown less than that of the control. This clearly indicates that either L.plantarum or the combined treatment (L.plantarum and B.megaterium) will always give the positive results and improvement over control once. Estimation of carbohydrate (Table 4): Analyzing carbohydrate from fish tissue, during the initial stage, almost all the treatments show that similar results, range between 39 to 55 µg/g of carbohydrates in tissue samples. At the time of feeding stage the carbohydrates in the fish sample is gradually increasing in all the treatments. But there is a higher significant growth was observed in L.plantarum treatment and the combined treatment (combined L.plantarum and B.megaterium), between 5 – 30% respectively. The same study of carbohydrate analysis was done during 60th day after the pathogen (A.hydrophila) treatment. Due to the presence of probiotic bacteria and its association with fish there is no much destruction caused by the pathogenic bacteria and it also never affect the protein profile or protein accumulation in its tissue. Rather, there is a significant increase in C3 and T3 (L.plantarum and B.megaterium with pathogenic treatment). Even in the B.megaterium treated (T3) fishes there is a significant increase in carbohydrate by 7% and along with pathogen 8%. But the combined treatment shows vary high percentage of carbohydrate. i.e. around 30% increase over the control.
Microbial analysis
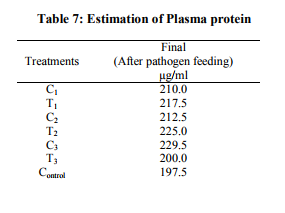
Four isolates of Gram-positive bacteria and four isolates of Gram – negative bacteria were isolated from the fish skin and internal organs (gonads, stomach, and intestine). Micrococcus spp., Lactobacillus spp., and Bacillus spp.,(one from skin and one from internal organs) are the Gram – positive bacteria isolated from the fish organs. Gram negative bacteria isolated from the fish organs are Pseudomonas spp., E.coli, Klebsiella spp., Aeromonas spp. Immunotechnological studies WBC count (Table 5): White blood cells plays a very important role in controlling the disease, the initial count of the WBC was between 5300 – 6200 cells/cu.mm an average. After 20 days interval (i.e. duration of probiotic treatment) again the WBC count was been carried out, during this stage the B.megaterium treatment shown the higher WBC count. i.e. 6450 cells/cu.mm). The above result reveals strong evidence that B.megaterium has slightly adverse affect when compare to L.plantarum. And also the immuno response study was been carried out after the pathogen treatment. i.e. after 60 days. The combined treatment shows the least WBC count when compared to control, L.plantarum and B.megaterium. This is clearly indicated that the T3 treatment were two probiotic bacteria and a pathogen (A.hydrophila) was given along with a feed that may be the reason for the decrease in WBC count. But the growth wise/feeding efficiency and activity wise the fishes were very healthy when it compared with other treatments. RBC count (Table 6): Before starting the treatment the initial of RBC during stage ranges 1.10 million/cu.mm to 1.45 million/cu.mm on average. The same trend of growth never observed at the end of the treatment. Were it shows the combined probiotic treatment without pathogen treatment shown very highest platelets (1.8 million/cu.mm RBC was founded). Then along with pathogen treatment (T3) has shown less when compared with other treatment. Based on the Table – 6 the author found out that there is no much significant role played by B.megaterium. Under the pathogenic treatment the mechanism of infection is taken place and same time L.plantarum along with B.megaterium without pathogen shown highest RBC count (1.53 million/cu.mm) when compared to other treatments. The treatment were only pathogen alone given (without probiotic bacteria 1 and 2), none of the fishes was been survived till the end of the study. All fishes were died within 40 days. Plasma protein estimation (Table 7): The initial plasma protein is also estimated, to compare with the final stage of plasma protein. Here also it is directly correlated to that of the total crude protein in it system. For example T1 and T3 treatment is almost (i.e. 217.5 and 200 µg/ml of plasma protein respectively) than that of T2 treatment plasma protein i.e.225 µg/ml was recorded. Coming to probiotic bacteria treatment the highest plasma protein was observed in C3 treatment were the combined bacterial blended granules feed were fed i.e., 229.5 µg/ml. Immuno electrophoresis: The whole cell antigen was been extracted from A.hydrophila and further centrifuged, separated and purified. Then by taking the serum of the fish which was under the treatment of L.plantarum and B.megaterium and A.hydrophila. The immuno electrophoresis result was positive, by band formation. Which is shows that the fish under treatment, developed antibodies against the pathogen. It may be a high complementary/competent antibody against the A.hydrophila antigen.
DISCUSSION
The aim of the study was to analyze and to find out the effect of few probiotic bacteria (L.plantarum and B.megaterium) on growth performance and immuno response of Catla catla. There are much remarkable differences between the control and L.plantarum and B.megaterium treatments with reference to growth parameters. To identify the bacterial species associated with the Catla catla, before treatments as well as after treatment. Before the treatment only very few gram positive/gram negative harmless bacteria were associated. Observation of micrococcus sp., E.coli, Pseudomonas sp., Enterobacter sp., klepsiella sp., were been reported. Whereas after the treatment, these initial bacteria were not predominant whereas L.plantarum, B.megaterium and here and there the pathogenic bacteria A.hydrophila was recorded. The current study of biochemical test author proves that when probiotic bacteria is predominantly associated with living system there is a chance of elimination of unwanted microorganism in the biological system. This is clearly evidence in the presence study. The similar study to that of carried out by Austin and Austin (1993). A wide range of Gram-positive (Bacillus, Carnobacterium, Enterococcus, Lactococcus, Lactobacillus, Micrococcus and Streptococcus) and Gram-negative bacteria (Aeromonas, Alteromonas, Photorhodobacterium, Pseudomonas and Vibrio) have been isolated from fish and has evaluated as probiotics in aquaculture (Irianto A. and Austin B. 2002). The final weight, weight gain, specific growth rate, survival rate, feed intake and protein efficiency ratio were increased among Catla catla fed a diet containing L.plantarum, so it may be considered as a growth promoter in fish aquaculture. The effect of probiotic feed in dietary protein and total plasma levels (from 30 – 40%) crude protein) were compared along with the dual treatment (pathogen and probiotic treatments). From data presented on the tables shows that during the time of probiotic bacteria feed as well as at the end of the experiment, the combined effect of probiotic bacteria is shows significant level of the protein content when compared with control as well as single probiotic bacteria feed. This work is also supported by Slah mesalhy Aly et al (2008). During the present experimental conditions the study shows that 20 – 25% of the best growth performance of Catla catla fingerlings. Feed conversion ratio is also decrease with increase with stocking density. The feed conversion ratio observed with fish reared at the lowest stocking density and fed the 30 crude protein diets. These results are similar those reported by, Essa and Nour (1998) and Zaki (1993). In the present study, L.plantarum and B.megaterium showed inhibitory effects in vitro against A.hydrophila. However, L.plantarum gave larger inhibition zone (4 cm) than B.megaterium (1.8 cm). Lewus et al. (1991) reported that the bacteriocins which produced by lactic acid bacteria had inhibitory effect against A. hydrophila pathogen protein. From the present study L.plantarum had a probiotic effect in vitro and in vivo against A. hydrophila, while B.megaterium had a probiotic effect in vitro and it small extinct in the in vivo. Our results agree with Chang and Liu (2002) who indicated that Bacillus toyoi suppressed the growth of Edwarsilla tarda in vitro, but did not reduce mortalities in eels due to edwardsillosis in vivo. In the present study author investigate the A.hydrophila cells are directly agglutinated with the fish (Catla catla) serum. In this regarding, further attentions are needed. Also much reference is needed, about the antigen – antibody interaction. Much less work has been directed at the immunological enhancement of defense mechanisms of fish by probiotic bacteria or the protective mechanisms of probiotic bacteria in fish (Nikoskelainen et al., 2003). Also less work has been directed at the blood parameters. In the present study, shows the decrease of RBC values in all treatments (T1, T2 and T3). But in the all controls (C1, C2 and C3) the RBC values are increased. These results are in agreement with that of Palikova et al (2004) who observed pathomorphological findings (hemorrhages in the skin, eyes, and hepatopancreas and in swim bladder) in the common carp after exposure to Cyanobacteria extract. WBC values are decreased in T1, T2 and T3 treatments and controls C1, C2 and C3 there is no much difference. White blood cells are very important role in controlling the disease. Decreased WBC levels at all the test (T1, T2 and T3) groups, due to effect of pathogenic bacteria (A.hydrophila). Much less work has been directed at the number of WBC and also in their action against the pathogenic organism during the probiotic bacterial treatment. The plasma total proteins showed decreased significance in fish fed with diet containing L.plantarum and mixture of L.plantarum and B.megaterium. These results agree with those of Cruz et al. (1989) who found lower total protein in plasma of Salmo gairdneri when injected with Vibrio anguillarum extracellular products intramuscularly.
CONCLUSION
The treatment of L.plantarum and combination of L.plantarum and B.megaterium was clearly revealed that it is beneficial for cultured when administered as a food additive/supplements. It is argued that such probiotic has a role in disease control strategies, growth promotion and it improves the blood platelets and biochemical parameters among Catla catla in aquaculture. However, a mixture of both bacterial species improved the protein content of fish. Many questions remain unanswered in the field of probiotics; a growing area of research indicates that they may be effective in treating or preventing a wide range of diseases in both humans and animals. The potential benefits of consuming probiotic bacteria include wide scale immuno – modulation as in auto – immune diseases and small scale suppression of specific intestinal pathogens. The list of targets is likely to grow as our understanding of the mechanisms behind probiotic activity continues to develop. Individual strain of probiotic bacteria was identified and carefully characterized for application of the aquaculture and other therapeutic uses. And also author suggested the more work on histopathological and immunological studies of diseased fish’s gives answers to the unanswered questions about the probiotics activity against the pathogens, in the field of aquaculture.
References:
1. Grisez, L. and Ollevier, F. 1995: Vibrio (Listonella) anguillarum infection in marine fish larviculture. In:Lavens, P., Jaspers, E., Roelande, l. (Eds.), Larvi 91-fish and crustacean larviculture symposium. Eur. Aquac. Soc. Gent. p. 497, Special publication no. 24.
2. Aoki, T.; Kanazawa, T. and Kitao, T. 1985: Epidemiological surveillance of drugresistant Vibrio anguillarum strains. Fish Patho., 20: 199-208.
3. Gomez-Gil, B.; Roque, A. and Turnbull, J.F. 2000: The use and selection of probiotic bacteria for use in the culture of larval aquatic organisms. Aquac.191: 259-270.
4. Verschuere, L.; Rombaut, G.; Sorgeloos and Verstraete, W. 2000: Probiotic bacteria as biological agents in aquaculture. Micro. And Mole. Biol. Rev., 64 (4): 655-671.
5. Salminen, S., A. C. Ouwehand, Y. Benno, and Y. K. Lee. 1999. Probiotics: how should they be defined? Trends Food Sci. Technol. 10:107–110.
6. Swain, P.; Sahoo, P.K.; Ayyappan, S. 2006: Fish and Shellfish Immunology: An Introduction. Narendra pub. Co, New Delhi. pp: 225 – 244.
7. Ventura, M.T.and J. M. Grizzle. 1987. Evaluation of portals of entry of Aeromonas hydrophila in channel catfish. Aquaculture . 65: 205 - 214.
8. Eissa, I. A. M., A. F. Badran, M. Moustafa, and H. Fetaih. 1994. Contribution to motile Aeromonas septicaemia in some cultured and wild freshwater fish. Veterinary Medical Journal Giza. 42: 63 - 69.
9. Cipriano, R.C. 2001Aeromonas hydrophila and motile aeromonad septicemias of fish, Fish disease leaflet 69, Fish and wildlife service division of fishery research, Washington,D.C.
10. Tekinay,A.A. Davis, S.J. 2001. Dietary Carbohydrate level influencing feed intake, nutrient utilization and plasma glucose concentration in Rainbow Trout. Turk. J. of Vet.Sci. PP.657 – 666.
11. Ruiz, C.M.; Roman, G. and Sánchez, J.L. 1996: A marine bacterial strain effective in producing antagonisms of other bacteria. Aquac. Intern. 4: 289-291.
12. Bergey, D.; Sneath, P. and John, H. 1984, Bergey’s Manual of Systematic Bacteriology. Williams and Wilkins, Baltimore. Vol. I section 5.
13. Austin, B. and Austin, D.A. 1993: "Bacterial Fish Pathogens: Diseases in Farmed and Wild Fish. 2nd ed. Ellis Horwood Ltd., Chichester, England.
14. Irianto A. and Austin B. 2002: Probiotics in aquaculture (Review). J. Fish. Diseases, 25: 633-642.
15. Salah Meshaly Ali,: Azza M. Abd – EI – Rahman, Gearge John and Mohamad F. Mohamed. 2008. Characterization of some bacteria isolated Orechromis niloticus and their potential use as probiotics. Aquaculture. 277 (1 – 2): 1-6.
16. Essa, M.A. and Nour, A.M., August, 1988. Effect of stocking density and supplementary feeding on growth rate, food utilization and cost of tilapia hybrid production (Oreochromis niloticus X O. aureus) in cages. Proc.lst Conf. Develop.Fish. Res. Alexandria, Egypt, 6-8, pp. 75-82.
17. Zaki, M.A.A. 1993. Some factors affecting growth performance, feed and nutrient utilization of common carp (Cyprinus Carpio L.) in earthern fresh water ponds. Ph,.D. Thesis.Fac.. Agric.Alex. Unív. Alexandria, Egypt, 135 pp.
18. Lewus, C.B.; Kaiser, A. and Montville, T.J. 1991: Inhibition of food-born bacterial pathogens by bacteriocins from lactic acid bacteria isolated from meat Appl. Environ. Microbiol. 57: 1683-1688.
19. Chang, C.I. and Liu, W.Y. 2002: An evaluation of two probiotic bacterial strains, Enterococcus faecium, SF68 B. toyoi, for reducing edwardsiellosis in cultured European eel, Anguilla anguilla L. J. Fish Dis., 25: 311-315.
20. Nikoskelainen, S.; Ouwehand, A.C.;Bylund, G.; Salminen, S. and Lilius, E. 2003: Immune enhancement in rainbow trout (Oncorhynchus mykiss) by potential probiotic bacteria (Lactobacillus rhamnosus). Fish and Shellfish Immun. 15: 443-452.
21. Palikova, M.; Navratil, S.; Krejcf, R.; Sterba, F.; Tichy, F. and Kubala, L. 2004: Outcomes of repeated exposure of carp (Cyprinus carpio L) to Cyanobacteria extract. Acta. Vet. Brno, 73: 259- 265.
22. Cruz, M. C.; De - La. And Mroga, K. 1989: The effect of Vibrio anguillarum extracellular products on Japanese eels. Aquaculture, 80 (3-4): 2010210.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License