IJCRR - 4(24), December, 2012
Pages: 59-63
Print Article
Download XML Download PDF
ACTIVITIES OF SOME ANTIOXIDANT ENZYMES AND LIPID PEROXIDATION IN LIVER CANCER PATIENTS
Author: Jain Suman, Sharma Renu, Sharma Suman, Varadkar A.M. Department of Biochemistry and Physiology, Geetanjali Medical College and Hospital
Category: Healthcare
Abstract:Introduction: Liver cancer is the leading cause of death due to malignancies worldwide. The extent of free radical induced oxidative stress can be exacerbated by the decreased efficiency of antioxidant mechanisms, The present study was conducted to investigate the extent of oxidative stress and the levels of antioxidants in the circulation of liver cancer patients. Method: Plasma thiobarbituric acid reactive substances (TBARS) and level of some antioxidant enzymes such as superoxide dismutase (SOD), catalase, and non enzymatic antioxidants vitaminC and vitamin E were estimated in the circulations of 50 liver cancer patients and an equal number of age matched normal subjects. Results: Significantly increased concentrations of plasma TBARS, significantly lowered levels of SOD, CAT, VitC and VitE were observed in liver cancer patients and may be due to their increased utilization to scavenge lipids peroxides as well as their sequestration by tumor cells. Increase levels of lipid peroxidation may be due to excessive oxidative stress.
Keywords: Antioxidants, Peroxidative stress, Malondialdehyde (MDA), Superoxide Dismutase (SOD), catalase, vitaminC, vitaminE
Full Text:
INTRODUCTION
Globally, primary liver cancer ranked seventh in men in 1975 and by 1990 it reached the fifth position with a 73% increase in the burden. (1) According to WHO more than 6,22,000 people across the world die because of the fatal disease. The disease is very much related to Hepatitis B and C virus. The exposure of these viruses is the primary reason of this disease, but smoking and liquor is also among the main reasons of liver cancer. (2) Role of oxidative damage in carcinogenesis is increasingly being appreciated. Free radicals are species capable of independent existence containing one or more unpaired electrons in their outer most orbital. Majority of tumors are caused by environmental factors which act almost always via free radicals. They are involved in both initiation and promotion of multistage carcinogenesis by damaging DNA, resulting in mutation protooncogene activation, inactivation or loss of tumor suppressor gene. (3) Oxidative stress plays important role in the pathogenesis of toxic liver diseases and also of some kinds of hepatic alterations (4) An antioxidant is the substance which delays or prevents oxidation of a substrates and constitutes the body’s primary defense against free radicals injury. Reducing the oxidative stress by supplementation of antioxidant could be effective option of chemoprevention and was suggested. (3) Oxidative stress caused by increased free radical generation and or decreased antioxidant level in the target cells and tissues has been suggested to play a important role in carcinogenesis. (5,6,7)
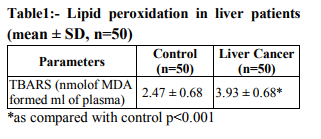
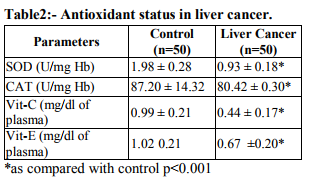
The levels of free radicals molecules are controlled by various cellular defense mechanisms consisting of enzymes Catalase (CAT), Superoxide Dismutase (SOD), Glutathione peroxidase and non enzymatic vitamin C and vitamin E glutathione components.(8) The present study is aimed to determine the extent of oxidative stress by measuring TBARS and antioxidant parameters in plasma of the patients with liver cancer. MATERIAL AND METHODS The study was conducted in R.N.T. Medical College and associated M.B. Hospital. Healthy subjects (50) & 50 liver cancer patients (outdoor & inward) were selected for this study. Patient Detailed present and past history of subjects was recorded on a separate proforma regarding their general information i.e. Age, sex, height, weight, caste, religion, dietary habits and education. The normal subjects were healthy medical students, staff members and attendant of patients, donors of blood bank of Maharana Bhupal Hospital/RNT Medical College Udaipur. Biochemical Estimations Lipid peroxide was estimated by measurement of thiobarbituric acid reactive substances (TBARS) in plasma by method of Yagi. (9) The lipid peroxidation was estimated at 532nm. Superoxide dismutase (SOD) was assayed by the method of Kakkar et al (10) based on the 50% inhibition of the formation of NADH-phenazine methosulphate nitroblue tetrazolium formazan at 520nm. Catalase activity was assayed by the method of Sinha(11) Hemolysate was treated with H2O2 (0.2mol/L) and the reaction was arrested after 60 sec by the addition of dichromate – acetic acid reagent cooled and the intensity of color read at 620nm. Various aliquots of H2O2 were used as the standard. A system devoid of the enzyme served as the control. Plasma vitamin C (ascorbic acid) was estimated by the method of Roe and Kuether (12) in which dehydroascorbic acid is coupled with 2, 4- dinitrophenylhydrazine and when treated with sulphuric acid forms an orange colour compound which was measured at 520nm. Plasma vitamin E(α-tocopherol ) was estimated by the method of Baker and Frank (13) The method involves the reduction of ferric ions to ferrous ions by α-tocopherol and the formation of a colored complex with 2, 2-dipyridyl. Absorbance of the chromophore was measured at 520nm. The data for biochemical analysis are expressed as mean, Standared Deviation. Statistical comparisons were performed by student’s‘t’ test. RESULTS Table 1 shows the level of lipid peroxidation in plasma of normal and liver cancer patients. Lipid peroxidation as assayed by thiobarbuturicacid reactive substances (TBARS) level was significantly higher in liver cancer patients as compared with normal subjects. Table 2 shows the level of antioxidants in the circulation of normal and liver cancer patients. The enzymatic antioxidants such as Superoxide dismutase (SOD) and Catalase in the hemolysate were significantly lower in liver cancer patients as compared with normal subjects. Also the nonenzymatic antioxidants vitamin C and vitamin E in plasma were significantly lower in liver cancer patients Vs control.
DISCUSSION
Oxidative stress is due to a disturbance in the balance between the production of ROS and the efficiency of the antioxidant defense. (14) Potential targets for free radical attack includes lipids, proteins and nucleic acids. (15) The epoxides generated due to increased oxidative stress may spontaneously react with nucleophilic centres in the cell and thereby covalently bind to DNA and RNA and protein. Such reactions may lead to cytotoxicity and carcinogenicity depending on the properties of the epoxides. (16) Moreover, severe oxidative stress is not only known to cause DNA and mutations in tumor suppressor genes but play important role in the promotion of multistep carcinogenesis. (17, 18) The end product of lipid peroxidation malondialdehyde, due to its high cytotoxicity and inhibitory action on protective enzymes is suggested to act as a tumor promoter and a cocarcinogenic agent. (19) Increased lipid peroxidation is observed in the plasma of cervical cancer patients also. (20) In our present work we noticed increased levels of circulating TBARS in liver cancer patients which can be attributed to the deficiency of antioxidant defense mechanisms. The antioxidant enzymes SOD and CAT widely distributed in all cells are present in high amounts in erythrocytes. SOD protects against O2 - by dismutation of the highly reactive superoxide anion to O2 and to a less reactive species to H2O2 . (21) Catalase protects the cell from H2O2. (22) In our case, we have observed low levels of SOD and Catalase in liver cancer patients. This correlates with the increase peroxidation activity. A decrease in the activity of catalase and SOD could be due to increase in lipid peroxidation products Malondialdehyde (MDA) which forms cross links inactivating several membrane bound enzymes and increase in lipid peroxides could be responsible for decreased SOD levels. Decreases catalase could be due to use of the enzyme because of increased peroxidation. Antioxidant vitamin E present in the cell along cell membrane. (23) and vitamin C is radical scavenging antioxidant (24) present in all cells as a reducing agent. A lipid peroxidation and its positive correlation with antioxidant vitamin E and vitamin C has been documented (25). Many studies regarding ovarian and other cancers also show inverse relationship between circulating levels of vitamin C and vitamin E. (26,27) )Due to low economic status and trends of using less fruits and vegetables people here are malnourished and thus prone to malignancy and thus associated with cancers (28) and also in liver cancer. (29)
CONCLUSION
Comparative prospective study was undertaken between healthy and liver cancer subjects with respect to lipid peroxidation and antioxidant level. MDA (malondialdehyde) levels (2.47±0.68) in healthy and patients had 3.93± 0.68 were significantly higher than controls. SOD (superoxide dismutase) levels were significantly lowered in patients as compared to healthy controls. Catalase levels were lower in patients than healthy controls. Vitamin C and Vitamin E were significantly lower in patients than healthy controls.
AKNOWLEGEMENT
We sincerely thank R.N.T. Medical College and associated hospital, Udaipur for extending all the facilities for conducting the work. Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
References:
1. Perin N. Global variation in cancer incidence and mortality. Current science. 2001; l81: 465.
2. New hope for liver cancer patients. 6th June. 2007; Newstract India.
3. Khanna K, Nikunji N and Khanna H.D. A study of antioxidant and their preventive potential in cervical Dysplasia... Asian Journal of Obs and Gynae Practice. 2002; l6: 3-11.
4. Diplock AT. Antioxidant nutrient and disease prevention: An overview. Am. J. Cli. Nutr1991; 53:189-193.
5. Halliwell B and J.M.C. Gutteridge. Free radicals in biology and medicines. 3rd ed. UK: Oxford Science Publications 1990; pp192.
6. Rajnesh C.P, Manimaran A, Sashikala KR, and Adaikappan P. Lipid peroxidation and Antioxidant status in patients with breast cancer. Singapore Med. J. 2008 49(8): 640- 643.
7. Lopaczynski, W and S.H. Zeisel. Antioxidants: Programmed cell death and Cancer Nutr. Res. 2001; 21:295-307.
8. Parol M, Leonardduzzi G, robino G, Albana E, Poli G, Dianzani M.. On the role of lipid peroxidation in the pathogenesis of liver damage induced by long- standing cholestasis.; Free radic Biol Med. 1996; 20 (3): 351-359.
9. Yagi K. Lipid peroxides and human diseases. Chem. Phys. Lipids. 1996; 45: 337- 351.
10. Kakkar P.S, Das B B and Viswanathan P N. A modified spectrophotometric assay of superoxidedismutase. Indian J. Biochem. Biophys.1984; 21:130-132.
11. Sinha K.A. Colorimetric assay of Catalase. Ann. Biochem.1984; 47:387-394.
12. Roe J.H. and Kuether C A. Detection of ascorbic acid in whole blood and urine through the 2, 4 DNPH derivatives of dehydro ascorbic acid. J. Biol. Chem.1943; 147:399-407.
13. Baker H, Frank O, De Angelis B and Feingod S. Plasma tocopherol in man at various times after ingesting free or acetylated tocopherol. Nutr. Rep. Int. 1980; 21: 531-536.
14. Kang D.H. Oxidative stress, DNA damage and breast cancer. AACN Clin.2002; Issues, 13: 540-549.
15. W.A. Cancer and free radicals. Basic Life Sci. 1986; 39: 45-59.
16. Tampo Y. and Tsukamoto M. The antioxidant action of 2-methyl- 6- (pmethoxyphenyl3, 7- dihydroimidazo [1,2- alpha]pyrazin-3-one (MCLA), a chemiluminescence probe to detect superoxide anions. FEBS Lett1998; 430: 348-352.
17. Ahmed M.I, Fayed ST, Hossein H and Tash FM, Lipid peroxidation and antioxidant status in human cervical carcinoma. Dis Markers 1999; 15: 283-291.
18. Halliwell, B. And Gutteridge M C , Free Radicals in Biology and Medicine. 3rd ed. London: Oxford 1999 pp: 152.
19. Diplock A, Rice-Evans AC, Burton RH, Is there a significant role for lipid peroxidation in the causation of malignancy and for antioxidants in cancer prevention? Cancer Res.1994; 54: 19525-19526.
20. Otamiri T. and Sjodahl R. Increased lipid peroxidation in malignant tissues of patients colorectal cancer. Cancer1989; 61: 122-125.
21. Manju V, Kalaivanisailaja J and Nalini N. Circulating lipid peroxidation and antioxidant status cervical cancer patients: a case-control study. Clin. Biochem. 2002; 35: 621-625.
22. Speranza M.J, Bagley AC and Lynch R E. Cells enriched for catalase are sensitised to toxicities of bleomycin, adriamycin and paraquat. J Biol. Chem.1993; 268: 19039- 19043.
23. McCord, J.M. The evolution of free radicals and oxidative stress. Am. J. Med 2000;108: 652–659.
24. Mates, J.M, Perez-Gomez C and De Castro I N. Antioxidant enzymes and human disease. Clin Biochem.1999; 32: 595-603.
25. Wagner B.A, Buettner G R, Burns P. Vitamin slows the rate of free radical mediated lipid in cells. Arch. Biochem. Biophys. 1996; 334: 261-267.
26. Fairfield K M, Hankinson S E, Rosner B A, Hunter D J, Colditz G A and Willett W C. Risk of ovarian carcinoma and consumption of vitamin A, C and E and specific carotenoids; a prospective analysis. Cancer 2001; 92: 2318-2326.
27. Bidoli E, Lavecchia C, Talamini R, et al., Micronutrients and ovarian cancer: a case – control study in Italy. Ann. Oncol 2001; 12: 1589-1593.
28. Aaron T.F, Sara H O, Laura M, Neal S and Susan H. Dietary antioxidant supplements, and risk of epithelial ovarian cancer. Nutr. Cancer 2002; 40: 92-98.
29. Eastwood, M.A. Interaction of dietary antioxidants in vivo: how fruits and vegetables prevent disease? QIM.1999; 92: 527-530.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License