IJCRR - 4(24), December, 2012
Pages: 17-31
Print Article
Download XML Download PDF
BIODIVERSITY OF ENTOMOPATHOGENIC FUNGI IN NEW CULTIVATED SOIL WITH THEIR USING TO CONTROL OF GALLERIA MELLONELLA
Author: Abd El- Ghany T.M., El-Sheikh H.H., Abd El-Rahman G. A., Abd El-Nasser A. M.
Category: General Sciences
Abstract:Chemical control for pests has been a serious concern for human and animal health; therefore this study was undertaken to screening of entemopathogenic fungi that are effective against against Greater wax moth Galleria mellonella. entemopathogenic fungi were isolated from different agricultural soil samples by plate dilution and baiting techniques. The crude extract of isolated fungi were screened against G. mellonella. Applied different fungal crude extracts of iolates achieved variable results on both larval and adult mortality percentages. The effect of crude extracts of Beauveria bassiana (Siwa isolate), Aspergillus tamarii, Fusarium solani, F. oxysporum, F. chlamydosporum and B. bassiana (El-Farafra isolate) giving total death percentages (82.48, 45.36, 37.12, 49.48, 53.6 and 74.22, respectively) to G. mellonella larvae comparing with the control treatment 0%. The results exhibited significant effects of B. bassiana (El-Farafra and Siwa Oasis isolates) against G. mellonella (larval and pupal weights). The % of larvae death increased with increasing the concentration of crude extract of B. bassiana (El-Farafra and Siwa isolates). Also pupal death percentage of G. mellonella increased while adult emergence decreased with increasing concentration of crude extract of El-Farafra and Siwa isolates of B. bassiana. On the other hand, crude extract of B. bassiana (El-Farafra isolate) was more active than B. bassiana (Siwa isolate) at high concentration (50 mg/ 0.5ml) while at other concentration crude extract of B. bassiana (Siwa isolate) was more active than B. bassiana (El-Farafra isolate). At the same time, different fractions of B. bassiana (Siwa isolate) extract were tested to determine the active metabolite. The bioactive compound was separated and identified as Beauvericin with using Thin layer chromatography, Fourier Transform Infrared (FT-IR) and analytical X ray.
Keywords: Soil; Entomopathogenic Fungi; Beauvericin; Bioactivty.
Full Text:
INTRODUCTION
Of the nearly one million known species of insects, about 15.000 species are considered pests and about 300 require some form of control. Fortunately, most insect pests have pathogenic microorganisms associated with them. There are two aspects of economic problems caused by insects. One concerns the loss of production that results from damage to crops and to the health of human and domestic animals, the other concerns the cost of attempt to prevent or control such production lose. At the same time with increasing agriculture, insects become more and more important competitors of human food damaging or even destroying the crops (Bale et al., 2008). The use of agrochemicals, although decreasing the attack of insects and phytopathogenic microorganisms, still represents a high risk to field workers and consumers. Over the last 25 years, chemical pesticides have become less attractive for numerous reasons including increased cost, the development of pesticide-resistant insects and weeds, concerns raised about human health hazards, and deleterious effects upon non-target organisms (Evans, 2008). Nowadays biological control as a practical science is very appreciated and as a solvent for long term usage of chemical pesticides problem is completely notified. Biological control is regarded as a desirable technique for controlling insects, due to its minimal environmental impact and preventing the development of resistance in vectors. Biological control (or biocontrol, which is synonymous) has been defined a number of times (Eilenberg et al., 2001). Entomopathogenic fungi have been found in many diverse habitats and associated with a broad range of insect hosts (Samson et al., 1988). These habitats include aquatic, forest and agricultural ecosystems that are of direct importance to insect-vector control, silviculture and crop protection (Bing et al., 2008), associated with insects (Pereira et al., 2009). Numerous laboratory and field studies indicate that entomopathogenic fungi (EPF) can provide a safe and effective control of many important insect pests (Lazreg et al., 2007; Stafford and Allan, 2010). The soil environment is usually the conventional isolation site for hypocrealean entomopathogenic fungi (Keller and Zimmerman, 1989; Hajek, 1997 (, and several species can be found in both cultivated and more natural habitats (Vanninen, 1996; Klingen et al., 2002). Bidochka et al. (1998) found Beauveria. bassiana to be affiliated with shaded and uncultivated habitats (i.e., forests) and B. bassiana also occurred frequently in hedgerow soils at a Danish locality (Meyling and Eilenberg, 2006). Agamy (2002) recovered the entomopathogenic fungus B. bassiana from soil samples from vegetable fields in El-Badrashin, Egypt. Eight fungi with differing incidence rates were isolated by Balogun and Fagade (2004) from Zonocerus variegates (Nigeria). These are Fusarium sp., B. bassiana, Metarhizium sp., Aspergillus flavus, Penicillium sp. , A. niger and Mucor sp. Fungi are known to produce a vast array of secondary metabolites that are gaining importance for their biotechnological applications (Siddhardha et al., 2009). Fungal secondary metabolites with high therapeutic value as antibiotics, cytotoxic substances, insecticides, compounds that promote or inhibit growth, attractor, repellent etc (Demain, 1999; Vey et al., 2001). Wasti and Hartmann (1978) investigated the effect of two toxins derived from B. bassiana, and other fungi on larvae of Galleria mellonella (L.) and Lymantria dispar (L.) by topical application and intra haemocoelic injection, the toxins from B. bassiana was the most effective. Fuguet et al. (2004) investigated the production of macromolecular insecticidal toxins by two selected strains of B. bassiana. Entomopathogenic fungi secrete a wide range of toxic metabolites in artificial culture and, in a few cases, in vivo (Vey, 1998). As B. bassiana is known to produce low mol. wt compounds such as beauvericin and bassianolide (Vey et al., 2001), toxicity was expected in dialysates prepared from filtrates, yet the dialysates had no lethal effect on the insect host. A toxic protein, bassiacridin, was purified from a strain of the entomopathogenic fungus B. bassiana isolated from a locust (Quesada and Vey, 2004). The work aimed to screening of entomopathogenic fungi from new cultivated soil with identification of bioactive compound of highly potency isolates .
MATERIALS AND METHODS
Sites of collection samples
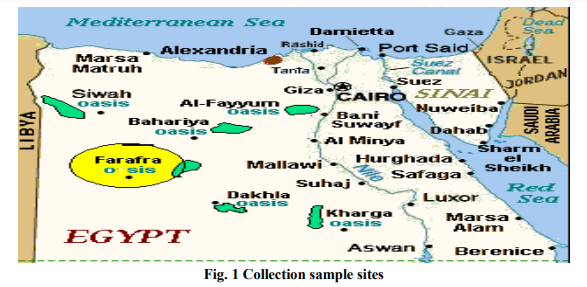
Eighty new cultivated soil samples were collected from four different regions (Siwa Oasis, El Baharia Oasis, El Farafra Oasis and Matrouh region ) with four location distributed in the Western desert and cultivated with different fruits and crops. Surface soil samples (5-15 cm depth) were collected from underneath different field plants for isolation of entomopathogenic fungi.
Media used and isolation techniqes
Czapek-Dox Agar (CzDA), Malt Extract Agar (MEA), Potato Dextrose Agar (PDA) media were used for isolation, identification and growth of isolated fungi. Two methods were used for entomopathogenic fungi , Soil Plate method according (Warcup, 1950) and Insect bait method with using newly hatched larvae of Galleria mellonella which were supplied from Plant Protection Department, Desert Research Center, Egypt. Insect-associated fungi were isolated from soil samples by using the ‘Galleria bait method’s (Marston and Brown 1974; Zimmermann, 1986; Woodring and Kaya, 1988; Meyling, 2007). Identification of fungal isolates Both light and high-vacuum JEOL JSM-5500LV scanning electron microscope were used for examination and identification of the fungal isolate according to Raper and Fennel (1965) and Samson et al. (1995) in the Regional Centr of Mycology and Biotechnology, Al Azhar University, Egypt. Fungal growth and their toxicity against Galleria mellonella Fifteen isolates were used to bioassay. Each isolate was cultured on potato dextrose broth medium at 28 ±2 oC. After inocubation period eight days, the growth medium was extracted by n-butanol. The butanolic extract was filtered on anhydrous of sodium sulphate and then concentrated. Different concentrations of the crude extract was tested to toxicity against 15 days old of G. mellonella larvae at different perids, the percentages of mortality were counted. Fractions of crude extract of more active isolates were prepared and examined against 15 days old Galleria larvae, 10 fractions was tested at concentration of 0.1 mg active ingredient from each fraction /1 ml distilled water. Determination of LC 50 was calculated according to Finney, (1964). Fractions of crude extract were used to determine the active fraction.Thin layer chromatography, Fourier Transform Infrared and Philips analytical X-ray were used to identify the active metabolite .
RESULTS
Screening of entomopathogenic fungi
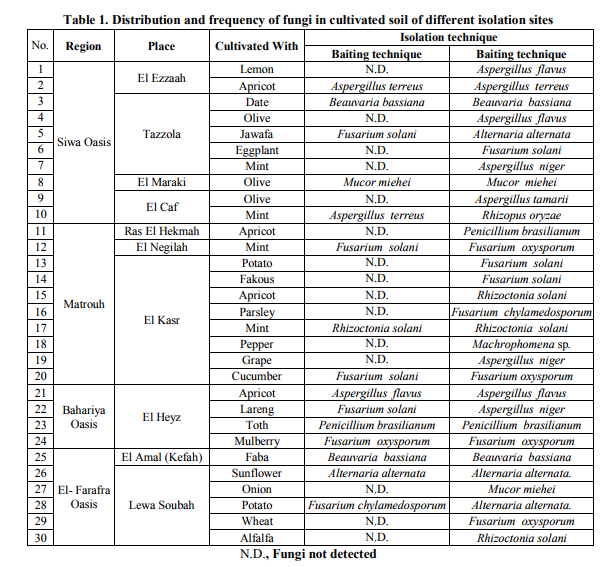
Different fungal isolates were isolated from different regions cultivated with different plants (Table 1 and Fig. 1) with Baiting and Plate dilution. Thirty isolates were isolated by Plate dilution while 15 isolates only were isolated by Baiting technique. In case of the soil sample that was taken from under the jwava trees in Tazzola site (located in Siwa Oasis), Fusarium solani was isolated by baiting technique while Alternaria alternata was isolated by plate dilution. The same observation was repeated for the soil samples from under the mint of El-Caf and El-Negilah sites, cucumber of El-Kasr, lareng of El-Heyz and from the soil sample of potato plantation that cultivated in El-Lewa Sobah village. On the contrary, certain fungi were isolated by both techniques. This observation was done on A. terreus that isolated from the soil under apricot trees in El-Ezzaah site and Beauvaria bassiana that isolated from the soil under date trees in Tazzola. Some fruit trees or crops that cultivated in more than one place had a high diversity of their soil-associated fungi. Where, in the soil samples of apricot trees that cultivated in ElEzzaah, Ras El-Hekmah, El-Kasr and El-Hayz sites, each of A. terreus, P. brasilianum, R. solani and A. flavus were detected respectively.
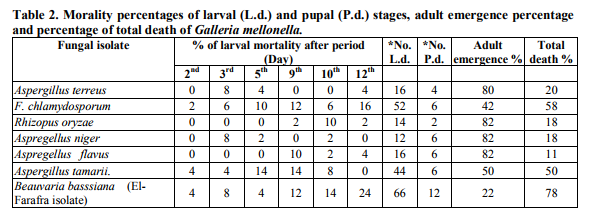
Effect of fungal crude extracts on both larval and adult stages
Applied different fungal crude extracts of iolates achieved variable results on both larvae and adult mortality percentages comparing with the check treatment. The effect of crude extracts of B. bassiana (Siwa isolate), A. tamarii, F. solani, F. oxysporum, F. chlamydosporum and B. bassiana (El-Farafra isolate) giving total death percentages (82.48, 45.36, 37.12, 49.48, 53.6 and 74.22, respectively) to G. mellonella larvae comparing with the control treatment 0 % (Table 2). On the other hand, the larvicidal effect of P. brasilianum, Mucor miehei, A. alternata, Macrophomena sp, Rhizopus oryzae and A. niger on G. mellonella larvae was weak. Fungal crude extracts, as shown in Table (2), were characterized by their latent effects on the pupal stage. The dead pupae ( Pupae which didn’t emerge as adult symbolized as P.d.). The % of pupal death was 12 % for B. bassiana (ElFarafra isolate) compared with 2 % F. oxysporum. The percent of viable pupae which turn out to be emerged as adult moths was also stated (Table 2). The highest moth emergence percentage was obtained from the larvae previously treated with P. brasilianum extract (86 %) while the least recorded one was 14% that achieved from the crude extract of B. bassiana (Siwa isolate).
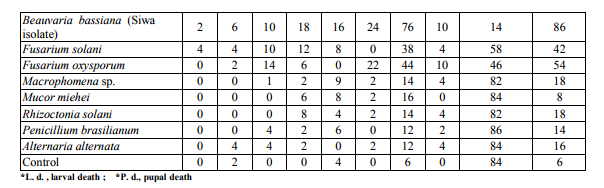
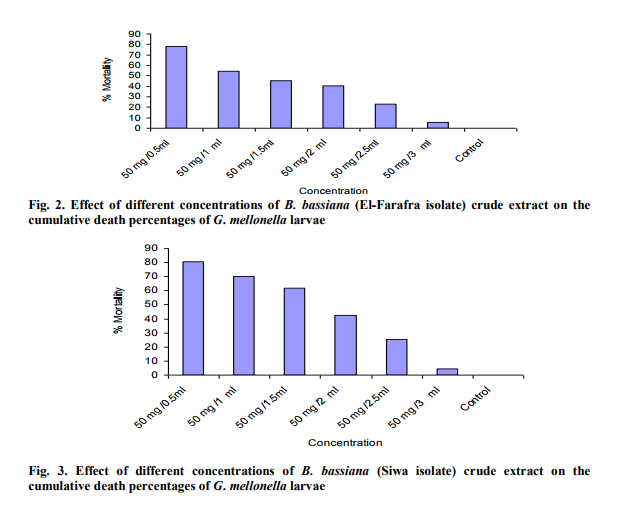
Effect of different concentrations of B. bassiana crud extract on G. mellonella larvae.
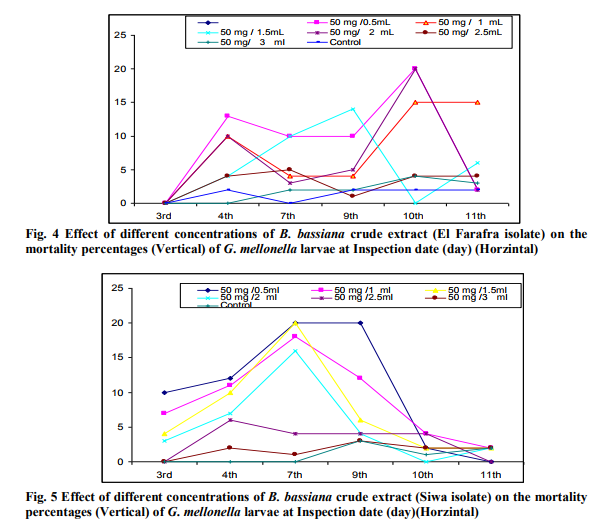
Highest mortality percentages of Galleria larvae was achieved with B. Bassiana particularly siwa isolate and therefore the effect of different concentrations of their extract on the 15 days old Galleria larvae was studied (Fig. 2 and 3). The % of larvae death increased with increasing concentration of crude extraction of B. bassiana (Siwa isolate) . B. bassiana (El-Farafra ) extract with 50 mg /3 ml caused the lowest cumulative larval death (11%) and 18 % when the applied concentration was 50 mg/2.5 ml. On the other hand, the fungal extract of 50 mg/0.5ml caused the highest larval mortality (55%). Focusing on the effect of applied concentrations of Siwa isolate on this parameter, similar trend could be observed. As the highest cumulative larval death percentage (64%) was achieved at the highest applied concentration, such percent was declined to 10% by applying 50 mg/3 ml concentration. Larval mortality with active extract of B. bassiana (El-Farafra isolate) increase by time of application in an ascending order (Fig. 4). Considerable larval death occurred after 96 hours after application while negligible death percentages were estimated before it. At 50 mg/0.5 ml concentration, the larval death percentage after 72 hours post treatment was 0 % then increased to record 13, 10 and 10 larval mortality percentages after the 4th, 7th and 9th days, respectively. Through the whole tested concentrations, the highest larval death percentage was observed after 10 days of application. With the application of B. bassiana (Siwa isolate) extract, the larval death percentages was recorded at 72 hours post treatment with most applied concentrations (Fig. 5), the high potency of B. bassiana (Siwa isolate) extract succeeded to show its influence faster than B. bassiana (El-Farafra isolate). The highest larval death percentages for the crude extract of Siwa isolate through all its applied concentrations was achieved after the 7th day of treatment.
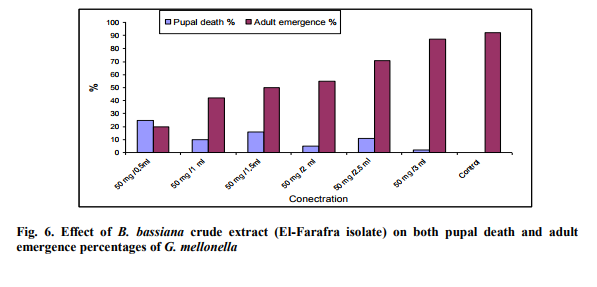
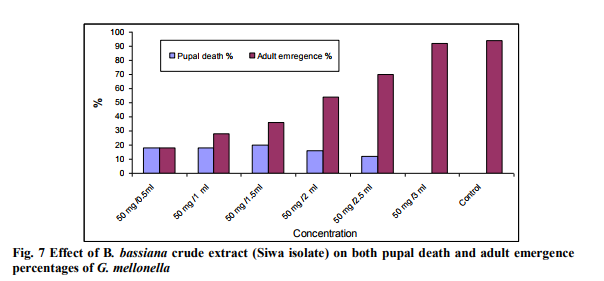
Effect of different concentrations of B. bassiana crud extracts on Pupal death and adult emergence of G. mellonella. Pupal death percentage of G. mellonella increased while adult emergence decreased with increasing concentration of crude extract of B. bassiana (El-Farafra and Siwa isolates). On the other hand, crude extract of B. bassiana (ElFarafra isolate) was more active than B. bassiana (Siwa isolate) at high concentration (50 mg/ 0.5ml) while at other concentration crude extract of Siwa isolate was more active than B. bassiana (El-Farafra isolate) (Figs. 6 and 7). Crude extract of El-Farafra isolate was currently active against Pupal of G. mellonella at low concentration 50 mg/ 3 ml while at this concentration crude extract of B. bassiana (Siwa isolate) was not active. Adult emergence at the lowest concentration (50 mg /3 ml) was 87% for B. bassiana (El-Farafra isolate) and 92% for B. bassiana (Siwa isolate) extracts.
Effect of B. bassaina crude extracts on larval and pupal weight gains Larval weight gain was forced to decline under the influence of both the vigor state of the Beauveria extract and the tested concentration. As the untreated larvae weighted about 0.23 gm the weight gain of treated ones recorded their minimum weight at 50 mg/0.5 ml concentration of B. bassiana extracts (0.05 and 0.01 gm of B. bassiana El-Farafra and Siwa isolates respectively). According to the calculated values of L.S.D., all applied concentrations of both extracts were significantly differed from the check trail. Such significant differences in the larval weights between treated and check ones declared the extending entomopathogenic influence of these extracts ( Table 3). The effect of B. bassiana (Siwa isolate) extract on the development and growth of G. mellonella adult was observed (Fig.8) ,where the abnormalities apperead on the body of adult G. mellonella compared with the control accompanied with a deficiency of weight.
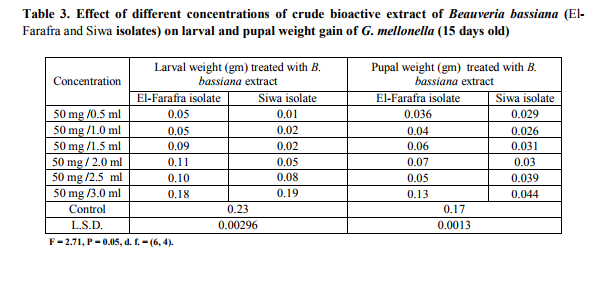
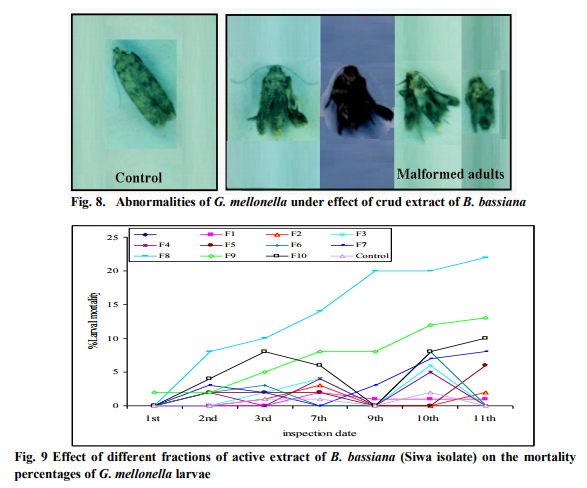
Effect of different fractions of crude extraction B.bassiana (Siwa isolate) on G. mellonella larvae
Fractions of B. bassiana Siwa extract the total larval death percentage of fraction 8, which exceeded 90% categorized this fraction as the highest entomopathogenic one. Fraction 9 by its recorded 50% total larval death was ranked in the second cohort, whereas the total larval death of the reminder fractions was ranged between 36 to 5 percentages that attained for fractions 10 and 1 respectively. Where, after the 2nd day of treatment the larval death percentage for this fraction (8%) was the highest one among the other fractions. For all testing fractions, the highest larval death percentages were attained either at the 10th or 11th day post treatment. For fractions 8 and 9, the recorded larval death during these inspection dates (10th or 11th day) was the highest among all fractions (Fig. 9).
Virulence of B. bassiana spore suspension on larval stage of G. mellonella
Mortality percentage in larvae of G. mellonella increased with increasing the time elapsed posttreatment, as well as the increasing concentration of spore suspension (Table 4 and 5). After the 3rd day of treatment, larval mortality was only recorded at the concentrations of 40 and 60 x106 spores/ml with almost low percentages (6 and 10 % respectively). For the intermediate concentrations (15 and 20x106 spores/ml), larval mortality was delayed to initiate 4th days post treatment by 4 and 6 % at 15 and 20x106 spores/ml respectively. Whereas larval mortality was commenced after the 5th day of application at the lowest concentrations (5 and 10 x106 spores/ml). As mentioned in Table (4), larval mortality percentages were of direct proportional relationship with the administered spore concentrations. The highest applied concentrations (40 and 60 x106 spores/ml) achieved 100 % larval mortality after the 10th day of application. At the later inspection date (12th day) the induced larval mortality percentages due to the lowest concentrations was ranged between 70 % for 15 x106 spores/ml and 44% for 5 x106 spores/ml (Table 4). On the same sequence, the bioassay test of B. bassiana fungal spores that isolated from siwa oasis was illustrated in Table (5), where larval mortality was commenced at 3 days post treatment. At the highest applied concentrations (40 and 60 x 106 spores /ml) larval mortality percentages achieved 100 % at the 9th day of application (Table 5).
Identification of active compound
The active extract of B. bassiana was fractinated and each fraction was tested as insecticid on Galleria larvae, Fraction 8 was the highest one among the other fractions and caused mortality percentage (91.83%). The fraction given promising results (Fraction 8) subjected to refractionation for purification and identification of active compound by using Thin layer chromatography, Fourier Transform Infrared (FT-IR) and analytical X ray. The data of fourier transform infrared of compound illustrated in the Table (6). X rayes indicated the presence of beauveicin as Colorless crystal, 1H NMR (CDCl3), δ 0.37 (3H, d, J = 6.6Hz, γ1-CH3, Hiv), 0.76 (3H, d, J = 6.6Hz, γ2- CH3, Hiv), 1.97 (1H, m, β-CH, Hiv), 2.93 (1H, dd, J = 11.9, 14.5Hz, β-CH(H), Phe), 2.98 (3H, s, N- CH3), 3.34 (1H, dd, J = 4.9, 14.5Hz, β-CH(H), Phe), 4.89 (1H, d, J = 8.5Hz, α-CH, Hiv), 5.51 (1H, dd, J = 4.8, 11.8Hz, α-CH, Phe), 7.20(5H, ar CH, Phe), 13C NMR (CDCl3), δ 17.4 (q, γ1- CH3, Hiv), 18.2 (q, γ2- CH3, Hiv), 29.6 (d, β- CH, Hiv), 32.2 (q, N- N- CH3), 34.7 (t, β- CH2, Phe), 57.2 (d, α-CH, Phe), 75.4 (d, α-CH, Hiv), 126.7 (d, ar CH, Phe), 128.4 (d, ar CH, Phe), 128.8 (d, ar CH, Phe), 136.6 (s, ar C, Phe), 169.3 (s, CO), 169.8 (s, CO), HRFABMS m/z, [M+H]+ 784.4164 (calcd for C45H58N3O9, 784.4173). From the above data of X-rays explained that fraction may contain beavericin. From the mentioned data documented, this compound may beauvericin.
DISCUSSION
As an alternative to chemical pesticides, the entomopathogenic fungus Beauveria bassiana is currently under intensive study as a promising biocontrol agent for insects and other arthropod pests (Kirkland et al., 2004;Howard et al. 2010). In our results the Beauveria bassiana is the common entomopathogenic fungus that isolated from soil cultivated with fruits (Dates) and crops (Faba). Entomopathogenic fungi are widespread in agroecosystems. The hypocrealean species B. bassiana have broad host ranges in agroecosystems. Recent advanced researches elucidated aspects of the ecology of the fungi that are relevant for conservation biological control. It is suggested that B. bassiana is associated with insect hosts above ground (Meyling , 2007), also grows naturally in soil through out the world and acts as a potent parasite on various insect species (Arti and Nilofer , 2010). Recently, B. bassiana has been linked to plants as an endophytic fungus (Arnold and Lewis, 2005). Recently Amy et al. (2009) reported that entomopathogenic fungi are commonly found in forested soils. The variability in the isolated fungi in our study may be due to type of soil or type of cultivated plants. Keller and Zimmerman (1989) stated that, a number of factors, including soil type (texture, cation exchange capacity, organic matter content, pH, etc.), moisture (water availability) and the presence of a viable soil micro-flora can influence the persistence and/or efficacy of entomopathogenic hyphomycetous fungi. In this study, soil-associated fungi were isolated by two isolation techniques, Galleria bait method and soil plate method, such isolation methods were well documented by several authors (Zimmermann, 1986; Bing et al., 2008 and Amy et al., 2009). According to the pre-mentioned fungal categories, opportunistic fungi were the second group. Among the isolated fungi, Aspregillus spp., Fusarium sp., Mucor sp. are opportunistic isolates (Bing and Zhong, 2008). On the same trend, the later authors identified such opportunistic fungi as fungal species other than well-known insect pathogens but can infect insects. During their research they found that, the most commonly isolated species of this group were Fusarium oxysporum, F. solani, Aspergillus flavus and Mortierella spp. In the current study, G. mellonella larval mortality that induced by opportunistic fungal isolates may be due to the characteristic of the fast growing opportunistic fungi, which may infect, injured or weakened insect (Hajek et al., 1997). From our observation, most fungal crude extracts had an insecticidal activity and it was easy to conclude that, the applied concentration of the different crude extracts achieved variable results on both larval and adult mortality of G. mellonella. Then from this screening test, B. bassain (Siwa and El Farafra isolates) was the promising agent upon achieving the highest percent mortality 86 and 78% respectively comparing with other isolates. The obtained results by Quesada et al. (2006) indicated that, extracts from two two B. bassiana isolates gave significant mortalities when applied either on alfalfa leaf discs or incorporated into artificial diet. The toxic effect of such extracts may be due to the fungal exudates (toxins) that excreted into the in-vitro growth media. Maketon et al. (2008) stated that each of Metarhizium anisoplias, Beauveria bassiana, Verticillium lecanii and Hirsutella citriformis achieved high mortality percentages of cotton jassid. Concerning with weight gain of treated larvae and pupae, our results stated that the tested concentration significantly diminish both larval and pupal weight. Such weight deficiency may be attributed to the antifeedent and/or mouthpart paralysis of Galleria larvae that exposed to crude extract contaminated media. Roberts (1981) declared the effect of fungal extracts on insect growth. Weight deficiency may be attributed to the effect of the applied treatments in decreasing the larval ability to convert ingested and digested food into body matter. In other study, Wilps (1989) stated that the reduction in weight gain of treated larvae were explained according to the fact that, there were considerable decrease in the amount of toxin ingested food (eaten diet) to that portion converted into body weight. As previously mentioned in our results the active ingredient that responsible for achieving theses symptoms was beauvericin. Griffin (1994) stated the following fact, fungi secret wide array of compounds that are biologically active against other organisms. Beauveria bassiana which has entomopathogenic properties may secret bassianin, beauvericin, bassianolide, beauverolides and tenellin (McCoy,1996).
CONCLUSION
B. bassiana that isolated from Siwa and El Farafra Oases achieved 82.48 and 74.22 total death % of G. mellonella, respectively (the highly potent isolates), their extracts adversely affected larval mortality. Comparing with control treatment, both extracts successfully decreased the larval weigh gain. The obtained data shed light on the higher efficacy of B. bassiana (Siwa isolate) extract as entomopathogenic agent than B. bassiana (ElFarafra isolate). The acftive compound was separated and identified as beauvericin. At the same time the larval mortality percentages were of direct proportional relationship with the administered spore concentrations of B. bassiana.
Acknowledgements:
The authors acknowledgement the team researchers of Regional Center for Mycology and Biotechnology, Al-Azhar University and Plant protection Departement Desert Research Center, egypt for providing necessary laboratory facilities.The authors are also grateful to authors and publishers of all those articles, from where the literature for this article has been reviewed and discussed.
References:
1. Agamy, E. A. 2002. Entomopathogenicity of Beauveria bassiana (Bals.) Vuillemin to early larval instars of the European corn borer, Ostrinia nubilalis (Hbn.) (Lepidoptera, Pyralidae). Egypt. J. Biol. Pest Contr., 12(1), 67-70.
2. Amy, R. T.; Jessica, L. M.; Shannon U. M.; Thomas, J. D.; Richard, C. F.; Michael,M.; Sadia, S.; Dieshia, R. and Kirby C. S.. 2009. Isolation of Entomopathogenic Fungi From Soils and Ixodes scapularis (Acari, Ixodidae) Ticks, Prevalence and Methods Journal of medical entomology. 46, 557-565. Arnold, A.E. and Lewis, L. C. 2005. Ecology and evolution of fungal entophytes, and their roles against insects. In, Vega, F.E., Blackwell, M. (Eds), Insect-fungal Associations, Ecology and Evolution. Oxford University Press, New York, 74-96.
3. Arti, P. and Nilofer, S. 2010. Evaluating prospects of fungal biopesticide Beauveria bassiana (Balsamo) Against Helicoverpa Armigera (Hubner), An ecosafe strategy for pesticidal pollution. Exp. Biol. Sci. Vol. 1 (3), 596-601.
4. Bale, J. S.; Van Lenteren, J. C. and Bigler, F. 2008. Biological control and sustainable food production. Philos. Trans. Roy. Soc. Ser. B, 363, 761-776.
5. Balogun, S. A. and Fagade, O. E. 2004. Entomopathogenic fungi in population of Zonocerus variegatus (l) in Ibadan,Southwest, Nigeria. Afr. J. Biotechnol., 3 (8), 382-386.
6. Bidochka, M. J.; Kasperski, J. E. and Wild, G. A. M. 1998. Occurrence of the entomopathogenic fungi Metarhizium anisopliae and Beauveria bassiana in soils from temperate and near- northern habitats. Can. J. Bot., 76, 1198-1204.
7. Bing, D. S.; Han,Y. Y.; Amanda, J. C. and Xing-Zhong, L. 2008. Insect-associated fungi in soil of field crops and orchards. Crop protect., 27, 1421-1426.
8. Bing, S. and Zhong, L. , 2008. Occurrence and diversity of insect-associated fungi in natural soils in China. Applied soil ecology, 39, 100-108.
9. Demain, A.L. 1999. Pharmaceutically active secondary metabolites of microorganisms, Appl. Microbiol. Biotechnol., 52, 455-63.
10. Eilenberg, J.; Hajek, A. and Lomer, C. 2001. Suggestions for unifying the terminology in biological control. Biocontrol., 46, 387-400.
11. Finney, D.J. 1964. Probit analysis. 2nd Ed., Cambridge. Univ. Press. England. 318 pp.
12. Fuguet, R.; Theraud, M. and Vey, A. 2004. Production in vitro of toxic macromolecules by strains of Beauveria bassiana, and purification of a chitosanase-like protein secreted by a melanizing isolate. Comp. Biochem. Physiol. C Toxicol. Pharmacol., 138(2), 149-161.
13. Griffin, D. H. 1994. Fungal physiology. Wiley-liss, New York, 458 pp.
14. Hajek, A.E. 1997. Ecology of terrestrial fungal entomopathogens. Adv. Microb. Ecol. 15, 193-249.
15. Howard, A.F.; Nguessan,R.; Koenraadt, C.J.; Asidi, A.; Farenhorst. M.; Akogbeto,M.; Thomas, M.B. and Knols,B.G. 2010. The entomopathogenic fungus Beauveria bassiana reduces instantaneous blood feeding in wild multiinsecticide-resistant Culex quinquefasciatus mosquitoes in Benin, West Africa. Parasit Vectors, 3, 87.
16. Keller, S. and Zimmerman, G. 1989. Mycopathogens of soil insects. In, Wilding, N., Collins, N. M., Hammond, P. M. and Webber, J. F. (ed.), Insect-Fungus Interactions. Acad. Press, London, 240-270.
17. Kirkland, B. H.; Westwood, G. S. and Keyhani, N. O. 2004. Pathogenicity of entomopathogenic fungi Beauveria bassiana and Metarhizium anisopliae to Ixodidae tick species Dermacentor variabilis, Rhipicephalus sanguineus, and Ixodes scapularis. J Med Entomol., 41, 705-711.
18. Klingen, I.; Meadow, R. and Aandal, T. 2002. Mortality of Delia floralis, Galleria mellonella and Mamestra brassicae treated with insect pathogenic hyphomycetous fungi. J. Appl. Entomol., 126(5), 231-237.
19. Lazreg, F.; Shaukat, A.; Shunxiang, R. and Muhammad, A. 2007. Biological characteristics and pathogenicity of Verticillium lcanii against Bemisia tabaci (Homoptera, Aleyrodidae) on eggplant Pak. Entomol. 29(2), 63-72.
20. Maketon, M.; Orosz-Coghlan, P. and Hotaga, D. 2008. Field evaluation of metschnikoff (Metarhizium anisopliae) sorokin in controlling cotton jassid (Amrasca biguttula biguttula) in Aubergine (Solanum aculeatissimum). Int. J. Agr. Biol., 10(1), 47-51.
21. McCoy, C. W. 1996. Pathogens of eriophyoid mite. In, Lindquist, E.E., M.W. sabelis and J. Bruin (eds.) Eriophyoid mites, their biology, natural enemies and control., 481-490.Elsevier sciences publication, Amsterdam.The Netherlands.
22. Meyling, N. V. 2007. Methods for isolation of entomopathogenic fungi from the soil environment laboratory manual. http,//orgprints.org/11200/01/11200.pdf.
23. Meyling, N. V. and Eilenberg, J. 2006. Isolation and characterisation of Beauveria bassiana isolates from phylloplanes of hedgerow vegetation. Mycol. Res., 110, 188-195.
24. Pereira1, E. S.; Sarquis, M.M.; Ferreira R. L.; Hamada,N. and Alencar,Y. 2009. Filamentous Fungi Associated with Mosquito Larvae (Diptera, Culicidae) in Municipalities of the Brazilian Amazon. Neotropical Entomology, 38(3),1-8.
25. Quesada- M. E.; Maranhao, E. A. A.; Garcia, V. P. and Alvarez, S. C. 2006. Selection of Beauveria bassiana isolates for control of the whiteflies Bemisia tabaci and Trialeurodes vaporariorum on the basis of their virulence, thermal requirements, and toxicogenic activity. Biol. Contr., 36(3), 274-287.
26. Quesada, M.E. and Vey, A. 2004. Bassiacridin, a protein toxic for locusts secreted by the entomopathogenic fungus Beauveria bassiana. Mycol. Res., 108 (4), 441- 452.
27. Raper, K.B. and Fennell, D.I. 1965. The genus Aspergillus. Williams and Wilkins Company, Baltimore, MD.
28. Roberts, D. W. 1981. Toxin of entomopathogenic fungi. In, Buregs H. D. (ed.) Microbial control of pests and plant diseases 1970-1980. Acad. Press, New York, 441 - 464.
29. Samson, R. A.; Evans, H. C. and Latge, J. P. 1988. Atlas of Entomopathogenic Fungi. Springer-Verlag, New York, p.187.
30. Samson, R.; Hoekstra, E.; Frisvad, J. and Filtunborg, O. 1995. Intoduction of food borne fungi. Baarn and lyngby.
31. Siddhardha, B.; Prabhakar, P.; Suryanarayana, M. and Venkateswarlu Y., 2009. Isolation and biological evaluation of two bioactive metabolites from Aspergillus gorakhpurensis .Rec. Nat. Prod.3,3,161-164.
32. Stafford, K. C. and Allan, S. A. 2010. Field applications of entomopathogenic fungi Beauveria bassiana and Metarhizium anisopliae F52 (Hypocreales, Clavicipitaceae) for the control of Ixodes scapularis (Acari, Ixodidae). J. Medical Entomol., 47, 6, 1107-1115.
33. Vanninen, I. 1996. Distribution and occurrence of four entomopathogenic fungi in Finland, Effect of geographical location, habitat type and soil type. Mycol. Res., 100, 93-101.
34. Vey, A. 1998. Researches on insecticidal mycotoxins as a contribution to the development of biological control and sustainable agriculture. OILB. WPRS. Bulletin 21, 71–76.
35. Vey, A.; Hoagland, R. and Butt, T. M. 2001. Toxic metabolites of fungal biocontrol agents. In Fungi as Biocontrol Agents Progress, problems and potential (T. M. Butt, C. W. Jackson and N. Magan, eds), 311–346. CABI Publishing, Wallingford.
36. Warcup, J. H. 1950. The soil plate method for isolation of fungi from soil. Nature, 166, 117-118.
37. Wasti, S. S. and Hartmann, G. C. 1978. Microbial toxin activity on larvae of the gypsy moth and greater wax moth. Z. Angew. Entomol., 85(2), 192-196.
38. Wilps, H. 1989.The effect of extracts from the neem tree Azadirachta indica on flight activity, food uptake, reproduction and carbohydrate metabolism in the dipteran Phormia terraenove (Dip., Muscidae). Zool. Johr. Abt. Fur. Allg. Zool., 93,271-282.
39. Woodring, J. L. and Kaya, H. K. 1988. Steinernematid and Heterorhabditid Nematodes, A handbook of biology and techniques, Fayetteville, Arkansas, Arkansas Agricultural Experiment Station,1-30.
40. Zimmermann, G. 1986. The Galleria bait method for detection of entomopathogenic fungi in soil. J. Appl. Entomol., 102, 213- 215.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License