IJCRR - 4(24), December, 2012
Pages: 01-06
Print Article
Download XML Download PDF
STUDIES ON MYCOFLORA ASSOCIATED WITH ROOTS OF SOLANUM INDICUM LINN. AND CHANGES IN CHEMICHAL CONSTITUENTS UNDER INFLUENCE OF RELATIVE HUMIDITY
Author: M. Rashidi, S. S. Deokule
Category: General Sciences
Abstract:The present investigation was designed to investigate associated fungi and deterioration of chemical constituents in drug Solanum indicum Linn. roots under influence of different relative humidities. Associated fungi isolated and identified according standard references. In the present study, total 20 fungi were associated with roots. F. semitectum, F. xysporum and F. solani showed high percentage incidence while minimum percentage incidence recorded in case of Sordaria fimicola, Neurospora sitophila, Thraustotheca sp., A. flavus, Aphanomyces sp., Chaetomium globosum and Rhizopus oryzae. The drug stored under the influence of different relative humidities viz. 30, 50, 75, 96 and 100% showed variation in percentage incidences of fungi as well as biodeterioration of chemical constituents such as sugars, proteins, phenols and glycosides. The drug stored under high relative humidity 96 and 100% and with the length of incubation days 45 and 60 days showed maximum deterioration of chemical constituents.
Keywords: deterioration, fungi, chemical constituents, percentage incidence.
Full Text:
INTRODUCTION
In Sanskrit plant Solanum indicum is called, "Brahati"and" Vrihati", and it is commonly called as "Brahanta", "Birhatta" and "Indian Nighte shade". This herbal plant belongs to the "Solanaceae" family. It is a common plant in all over world. Fruit and roots contain wax, fatty acids and alkaloids: Solanine and Solanidin. It is useful in asthma, dry cough, difficult parturition, chronic fibrile effection worms, scorpion sting also dysuria. The roots are one of the ingredients of Dashmoola Kautha of Hindi medicine. The roots take internally manifests strongly exciting qualities. It is employed in difficult parturition and in toothache and also used in fever, work complaints and colic (Kritikar and Basu, 1984). There are not any report concentrating on the subject mycoflora associated with roots of drug S. indicum and changes in their chemical constituents due to spoilage, therefore, this survey was carried out to determine the percentage incidence of fungi associated with the roots of this plant stored at different relative humidities and second part of this study involved determination of quality and biodeterioration of chemical constituent amounts in samples in relation to fungal contamination.
MATERIAL AND METHODS
The roots of S. indicum were collected from different localities. It was brought to the laboratory in separate polyethylene bags to avoid aerial contamination. Blotter test method, as recommended by International Seed Testing Association (1966) was adopted for isolation of fungi. Agar plate method was also used. The roots were sterilized with 2% NaOCl solution before plating. In order to evaluate the chemical changes in relation to fungi associated, the samples were stored in small muslin cloth bags under different RH levels i.e. 33, 55, 75, 96,100 % and at 28 ± 3?C temperature for 90 days (Wink and Sears, 1950). At an interval of 15 days, samples were taken out and washed thoroughly with sterilized distilled water and plated in Petri plates. The isolation of mycoflora was recorded from first day to 60th day of storage. Fungi were identified by using references such as Raper and Thom (1949), Barnet and Hunter (1972) and Nelson et al. (1983). Some parts of samples after washing with distilled water were dried in oven for chemical analysis. The changes take place in total proteins, phenols and glycosides amounts in the samples were estimated by the methods of Lowry et al. (1951), Singh et al. (1978) and Kokate et al. (2002). Anthrone method for total sugars and Dinitrosalicilic acid (DNSA) method for reducing sugars amount were also followed (Sadasivam and Manickam, 1992). Data from the experiment were analyzed by SPSS software in which statically significance was determined at 0.05 % probability levels.
RESULTS AND DISCUSSION
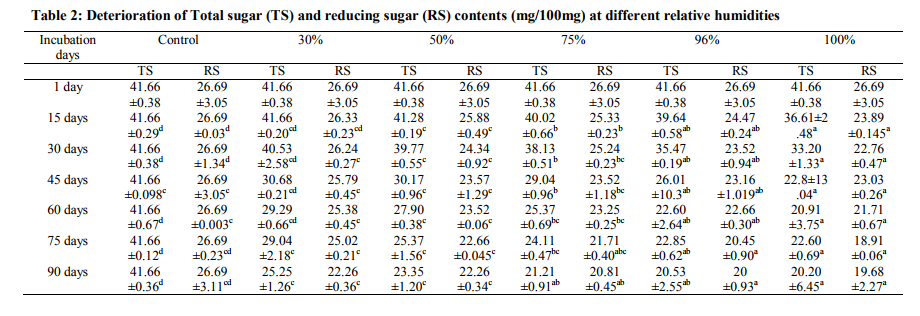
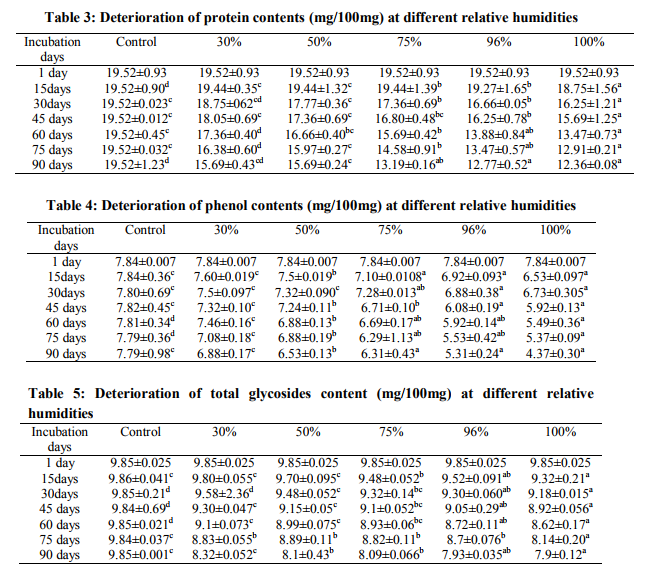
Total 20 fungi were isolated from the fresh roots of Solanum indicum in varying percentages (Table 1) such as F. semitectum 26.39%, F. oxysporum 10.92%, F. solani 10.82%, Theilavia terricola 9.25%, Scytallidium thermophilum 6.98%, Trichoderma sp. 6.05%, A. niger 5.11%, Papulaspora immerse 4.41%, F. lateritum 3.13%, Syncephalestrum racemosum 2.12%, Drechslera bicolor 1.93%, F. equiseti 1.66%, Sordaria fimicola 0.57%, Neurospora sitophila 0.67%, Thraustotheca sp. 0.76%, A. flavus 0.86%, Aphanomyces sp. 0.77%, Chaetomium globosum 0.38% and Rhizopus oryzae 0.28%. Fresh roots of S. indicum stored under different relative humidity and deterioration of sugars in each tested relative humidity observed (Table 2). In case of 30, 50, 75, 96 and 100 % RH, after 15 days of incubation TS (Total sugar) and RS (Reducing sugar) values observed 41.66, 26.33%; 41.28, 25.88%; 40.02, 25.33%; 39.64; 24.47% and 36.61, 23.89%, they showed more deterioration after 90 days of incubation period 25.25, 22.26%; 23.35, 22.26%; 21.21, 20.81%; 20.53, 20% and 20.20, 19.68%. Protein contents in the roots of S. indicum stored under 30 and 50 % RH reduced to 18.75, 17.36, 15.69 % and 17.77, 16.66 and 15.69 % after 30, 60 and 90 days of incubation days. At 96 and 100 % RH showed considerable deterioration 16.66, 13.88, 12.77% and 16.25, 13.47 and 12.36% after 30, 60 and 90 days of storage (Table 3). The total phenol content in roots was 7.84% (Table 4), this value of phenol was deteriorated to 4.37 % under 100 % RH and after 90 days of incubation period. Minimum reduction observed under 30 and 50 % RH and maximum reduction observed under 96 and 100 % RH. In all tested relative humidity, total amount of phenol, after 30, 60 and 90 days of incubation observed 7.5, 7.32, 7.28, 6.88 and 6.73% (30 days ); 7.46, 6.88, 6.69, 5.92, 5.49% (60 days); 6.88, 6.53, 6.31, 5.31 and 4.37% (90 days). The roots stored under 30 % RH showed reduction to 9.80% in total glycosides amount (Table5) after 15 days, this value deteriorated to 9.32% after 90 days of incubation. In case of 50 and 75 % RH also decline in total glycosides observed, 9.70, 9.48, 8.99, 8.1%; 9.48, 9.32, 8.93, 8.09 % after 15, 30, 60 and 90 days of storage. Similarly, under 96 and 100 % RH maximum reduction in total glycosides observed, after 15 days to 90 days of storage total amount of glycosides deteriorated from 9.52, 9.32% to 7.93 and 7.9% respectively Analysis of variance revealed that relative humidity and different incubation days were effective on reduction of sugars (TS, RS), proteins, phenols and glycosides value. The fungi isolated from drug roots of S. indicum in various % incidence spoils the roots and ultimately affect on changes of chemical constituents. During the period of storage (first day to 90th days and under 30 to 100% RH) biochemical analysis of roots were showed that samples stored at 30 and 50% RH recorded minimum deterioration of chemical constituents but under 75, 96 and 100% RH showed the significant reduction in the sugars, proteins, phenols and glycosides content. The prolong storage period is also effected on associated mycoflora and their growth on roots and continuously reduction of medicinal values. Changes in chemical constituents may be due to active interference of fungi in breakdown of constituents and utilization by them. Several workers have been showed deterioration of chemical constituents under storage due to spoilage of fungi in different plants (Deokule and Kabnoorkar, 2008; Kabnoorkar and Deokule, 2009; Dutta and Roy, 1987, Bilgarmi et al. 1978, Roy et al. 1987, Inman, 1962, Marx et al. 1966).
CONCLUSION
The result of this investigation indicate that the maximum reduction of chemical constituents is after 60th to 90th days of incubations while total percentage incidence of fungi are also maximum in mycoflora associated with samples. The result indicates that the quality of herbal drugs may be retained by storage condition at lower RH (below 50% RH) and short storage period.
ACKNOWLEDGMENT
Authors thank to the head of the Department of Botany, University of Pune for providing necessary facilities and encouragement. Authors acknowledge the great help received from the scholars whose articles cited and included in references of this manuscript. The authors are also grateful to authors/ editors/ publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed. Authors are grateful to IJCRR editorial board members and IJCRR team of reviewers who have helped to bring quality to this manuscript.
References:
1. Barnet HL, Hunter BB. Illustrated Genera of Imperfect Fungi. Minneapolis Burgress Publishing Company. Minneapolis; 1972
2. Bilgarmi KS, Sinha RK, Prasad T. Effect of fungal flora on the seed contents of moong. Indian Phytopatho. 1978; 31: 476-479.
3. Deokule SS, Kabnoorkar PS. Studies on phytochemical constituents of drug Punarnava under storage condition due to fungi. J. I. B. S. 2008; 87(3and4): 256 - 261.
4. Dutta GR, Roy AK. Mycoflora associated with Strychnous seed deterioration of their active principles under storage, Indian Phytopath. 1987; 40 (4): 520.
5. Inman RE. Disease development, disease intensity and Carbohydrate levels in rusted bean plants, Phytopath. 1962; 52; 1207.
6. International seed testing association. International rules for seed health testing, Proc. International seed test assoc. 1966; 31; 1.
7. Kabnoorkar PS. Deokule SS. Studies on changes in chemical constituent’s roots of Boerheavia diffusa infested with Aspergillus parasiticus and A. niger under storage. Geobios 2009; 36: 206-210.
8. Kiritikar KR. Basu BD. Indian Medicinal plants 2nd edition. Vol1. Published by L. M. Basu, Allahabad;1984
9. Kokate CK, Purohit AP, Gokhal SB. Pharmacognosy, Nirali Prakashan 18th Ed. 2002; 238.
10. Lowry OH, Rosenberg NJ, Fan AL, Randal RF. Protein measured with the folinciocalteu reagent, J. Biol. Chem 1951; 193: 265.
11. Marx AF, Beck HC, Vananderwaard WF, Flines J. Microbial deterioration of materials. In ‘Microbial biodeterioration’ (ed A.H. Rose) Academic press London, 1966.
12. Nelson PE, Tossoum TA, Marasa WFO. Fusarium Species. An Illustrated Manual for Identification . The Pennsylvania State University Press. U. S. A. 1983; 193
13. Raper KB, Thomas C. A manual of Penicillia Williams and Wilkins. Battimore, 1949; 850
14. Roy AK, Chourasia HK, Kumari N. Association of mycoflora with some crude herbal drug of Bhutan. Indian Bot. Reptr 1987; 6(1): 48.
15. Sadasivam S. Manickam A. Biochemical Methods of Agricultural Sciences. Wiley Estern Ltd, New Delhi, India. 1992.
16. Singh M, Singh SS, Sanwal GG. A new colorimetric method for the determination of phenolics, Indian J. Exp. Biol. 1978; 16(4); 712.
17. Wink SA, Sears GR. Instrumentation studies LVII equilibrium relative humanities above saturated salt solutions at various temperatures, Tapp 1950; 33(9) 96A.

|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License