IJCRR - 5(1), January, 2013
Pages: 58-63
Print Article
Download XML Download PDF
CARTILAGE METABOLIC MARKERS AND ANTIOXIDANTS: ITS CORRELATION IN OSTEOARTHRITIC PATIENTS OF SIKKIM
Author: Sonam Choden Bhutia, T. A. Singh, Mingma Lhamu Sherpa
Category: Healthcare
Abstract:Background: Oxidative stress has been implicated as a mediator of cartilage damage in patient with Osteoarthritis (OA). Aim: To assess the antioxidant markers like superoxide dismutase (SOD) , glutathione peroxidase (GPx) and uric acid (UA) in the blood and cartilage metabolic markers like hyaluronic acid (HA) and keratan sulphate (KS) in the synovial fluid of osteoarthritic patients and to find the correlation between these parameters in the blood and synovial fluid. Methods: This was an observational study conducted in the department of Biochemistry, Sikkim Manipal Institute of Medical Sciences (SMIMS), Gangtok. 75 osteoarthritic patients and healthy volunteers were taken for the study. Blood levels of antioxidants (SOD, GPx and UA) were assayed in both osteoarthritic and healthy volunteers. Cartilage metabolic markers like HA and KS were assayed in the synovial fluid. Result: Osteoarthritic patients showed statistically significant increase in KS and decrease in HA level indicating cartilage damage. Decrease in SOD and GPx activities indicates increase in oxidative stress. The difference in UA levels in the two groups was not statistically significant. This study did not establish any significant positive/negative correlation between the antioxidants in the blood and cartilage metabolic markers in the synovial fluid of osteoarthritic patients.
Keywords: Osteoarthritis, cartilage metabolic markers, Antioxidants, GPx, SOD
Full Text:
INTRODUCTION
Osteoarthritis (OA) is the leading cause of chronic disability worldwide between the fourth and fifth decade of life with a prevalence of 17% to 60.6% in India (1, 2). Knee is possibly the most commonly affected joint in most of the communities (3). Articular cartilage destruction in OA is believed to be a result of excessive loading, age-related changes, and metabolic imbalance in the tissues (4, 5, 6). The body fluid levels for direct and indirect biochemical markers of structural or metabolic changes in joint tissues has begun to provide clinically useful information. Cartilage derived molecules present in the synovial fluid may be markers of biosynthetic changes or of degradative changes (7). Keratan sulphate (KS) estimation has been proposed to be a potential marker of cartilage destruction in arthritis (8). Hyaluronic acid (HA) plays an important role in the protection of articular cartilage and soft tissue surfaces of the joint(9). Serum HA has been suggested as a useful indicator in assessing knee osteoarthritic activity (10). In knee OA the mechanism of cartilage matrix degradation is not clearly understood, but it is thought that Reactive Oxygen Species maybe one of the causes. Free radicals are formed in both physiological and pathological conditions in mammalian tissues (11).
Alteration of the Redox status is known to occur in rheumatic diseases (12,13) .To prevent ROS toxicity, our body possesses well co-ordinated antioxidant systems like superoxide dismutase (SOD), glutathione peroxidase (GPx) and non enzymic antioxidant like Uric acid (UA) among others. Antioxidants are compounds that dispose, scavenge and suppress the formation of free radicals or oppose their actions. It is reported that HA also acts as an antioxidant as it is an anti-inflammatory substance (14). To date various biomarkers of OA have been studied to potentially aid in the diagnosis and to assess minor changes in bone and cartilage of knee OA. Very few correlation studies between blood antioxidants and a cartilage metabolic marker have been published. The study aims to evaluate the activities of antioxidant enzyme SOD, GPx and non enzymic antioxidant level UA in the blood and cartilage metabolism markers like HA and KS in the synovial fluid of osteoarthritic patients and to determine the correlations between the parameters measured in blood and synovial fluid.
MATERIALS AND METHODS This study is an observational study and was conducted in the Department of Biochemistry, Sikkim Manipal Institute of Medical Sciences, Gangtok, Sikkim with due approval from the Institutional Ethics Committee. Participation in the study by all was solely voluntary, with prior detailed information and consent.
Subjects: 75 clinically diagnosed patients with primary knee osteoarthritis of age group (40-80 years) were selected from patients attending Sir Thutop Namgyal Memorial Hospital (STNM) and Central Referral hospital (CRH), Gangtok. 75 healthy volunteers were taken for the statistical comparison for the parameters analyzed in the blood. No comparison could be made in healthy volunteer’s synovial fluid due to ethical concerns. Classification of the cases: Osteoarthritis was classified according to the classification criteria described by American College of Rheumatology (15). Exclusion criteria: Smokers, alcoholics, diabetics, hypertensive and patients suffering from any other systemic disease including patients with post traumatic osteoarthritis were excluded from the study. Sample collection and processing: Heparinised venous blood samples were collected and part of it was centrifuged and plasma was used for the estimation of UA (Uricase Method, RFCL, India) (16). The other part of the venous whole blood was hemolysed and used to estimate the activity of SOD by Ransod kit, and activity of GPx by Ransel kit (Both from Randox laboratory ltd, UK) (17,18). Synovial fluid was aspirated and centrifuged to remove the debris and the supernatant was used for the estimation of HA and KS by sandwich ELISA (Blue Gene Biotech Co ltd, Shanghai) (19). Since we did not have any healthy synovial fluid for comparison we have used the reference range obtained from Bluegene (19).
Statistical analysis SPSS (version 16) was used for our statistical analysis. The results were analyzed by using Independent‘t’ test for comparing the means between osteoarthritic patients and healthy controls. Pearson’s correlation was used to study the relation between the antioxidants and cartilage metabolic markers in osteoarthritic patients. P<0.05 was taken to be significant for our study.
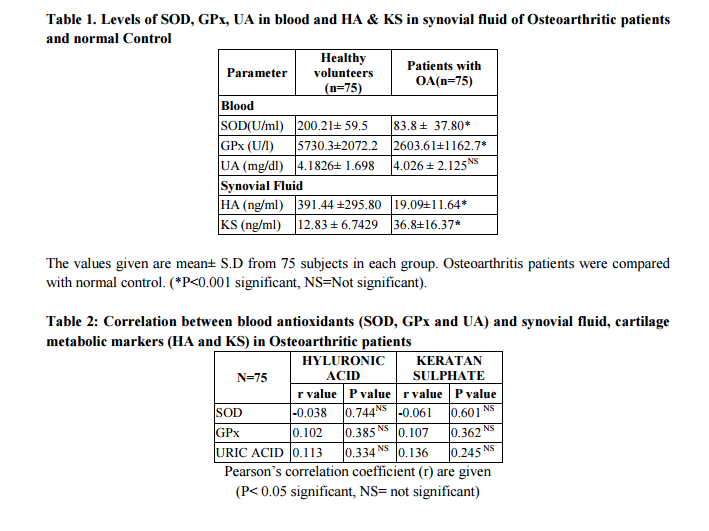
RESULT The findings of antioxidant levels in blood (SOD, GPx and UA) and cartilage metabolic markers (HA and KS) in synovial fluid and their comparison between healthy volunteers and Osteoarthritic patients are shown in table 1. The mean blood concentration of GPx, and SOD, were significantly decreased when compared with control group. No significant difference in UA levels between the two groups was observed. The mean synovial fluid concentration of HA, was significantly decreased in OA in comparison with the reference range and KS was significantly increased in OA patients when compared with reference range. Redox status of an individual has been implicated with risk of developing osteoarthritis by various studies (21). In our study, SOD showed insignificant negative correlation with both HA and KS. Glutathione peroxidase enzyme on the other hand showed positive correlation with both HA and KS that was statistically insignificant (p value>0.05) (table 2).
DISCUSSION Different studies worldwide have shown alteration in redox status and the shift towards oxidative stress leading to decrease in antioxidant levels as they are oxidized by the free radical and this is the protective response of the body to any oxidative damage. Synovial cavity damage has been associated with oxidative stress by some studies (20). In our study significant decrease in antioxidant enzymes SOD and GPx is in consort with few other studies (21, 22). UA, though being an antioxidant, no significant difference was observed in our study and this could be due to the complex, selective antioxidant capacity of uric acid (23). Our study findings are consistent with the findings of insignificant changes in UA and no association of serum uric acid in OA (24, 25).
The decreased levels of HA in OA in our study is similar to previous studies (26, 27). The decrease may be due to dilutional effects, reduced hyaluronan synthesis and free radical degradation (28). Different studies have projected that KS levels increases in OA (29, 30) and stated that it is a promising marker of early cartilage breakdown (31, 32). This study showed no significant correlation between the antioxidants in the blood and the cartilage metabolic markers in the synovial fluid. This may be due to the fact that the assays for antioxidants were done in the blood and the cartilage markers in the synovial fluid and blood antioxidants may not truly reflect local redox status.
CONCLUSION Our results suggest that even though the antioxidants are significantly lowered in blood, it did not show any significant positive/ negative correlation with the synovial fluid cartilage metabolic markers in study. This may be due to the limitations of our study where the antioxidant assay and cartilage metabolic markers were studied in different body fluids and the total antioxidant status (TAS) assay was not done. This limitation was due to the fact that we could not get healthy volunteers for synovial fluid analysis due to ethical reasons. Further studies would be required to compare the total antioxidants in blood and synovial fluid and correlating the findings both in serum and synovial fluid to get a true picture of the role of redox status in osteoarthritis. The significant decrease in antioxidant enzymes and hyaluronic acid and increase in keratan sulphate level supports the plausible role of oxidative stress in osteoarthritis and may pave way for developing different preventive and therapeutic strategies.
ACKNOWLEDGEMENTS Authors acknowledge the great help received from the scholars whose articles cited and included in references of this manuscript. The authors are also grateful to authors/ editors/ publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
References:
1. Lutzner J, Kasten P, Gunther KP, Kirschner S.Surgical options for patients with Osteoarthritis of the knee. Nat Rev Rheumatol 2009; 5:309-16.
2. Sharma MK, Swami HM, Bhatia V, Verma A, Bhatia SP, Kaur G. An Epidemiological study of correlates of Osteoarthritis in Geriatric population of UTChandigarh. Indian J.Community Med.2007; 32:77-8
3. Syed A.HAQ, Fereydoun Davatchi. Osteoarthritis of the knees in the COPCORD world International Journal of Rheumatic Disease.2011; 14(2): 122-129.
4. T.Aigner, J.Haag, J.Martin, J.Buckwalter. Osteoarthritis: aging of matrix and cellsgoing for a remedy. Current Drug Targets 2007; 8(2): 325-331.
5. T.Aigner, N Gerwin. Growth plate cartilage as developmental model in osteoarthritis research potentials and limitations. Current Drug Targets 2007;8 (2):377-385
6. P. M. Van der Kraan, E. N. Blaney Davidson, W. B. van den Berg. A role for age-related changes in TGF beta signaling in aberrant chondrocyte differentiation and osteoarthritis. Arthritis Research and Therapy 2010; 12(1):201-214.
7. Thonar EJ-MA, Manicourt D-H. Noninvasive markers in Osteoarthritis. In Osteoarthritis: Diagnosis and Medical/ Surgical Management, 3 ed. Edited by Markowitz R, Goldberg V, Vowell DS, Altman RD, Buckwalter J. Philadelphia, and PA: WB Saunders; 2001:293-313.
8. Budsberg SC, Lenz ME, Thonar E. Serum and synovial fluid concentrations of keratan sulfate and hyaluronan in dogs with induced stifle joint osteoarthritis following cranial ligament transaction. Am J Vet Res 2006; 67: 429-32.
9. Van den Bekerom MP, Lamme B, Sermon A, Mulier M. What is the evidence for Viscosupplementation in the treatment of patients with hip osteoarthritis? Systematic review of the literature.Arch Orthop Trauma Surg 2008; 128(8):815-823.
10. Turan Y, Bal S, Gurgan A, Topac H, Koseoglu M. Serum hyaluronan level in patients with knee Osteoarthritis .Clin Rheumatol 2007; 26:1293-8.
11. TAS F,Hansel H,Belce A,Ilvan S, Argon A, Camlica H , et al. Oxidative stress in ovarian cancer. Med Oncol.2005; 22(1):11- 15.
12. Mezes M, Bartosiewicz G. Investigations on vitamin E and lipid peroxide status in rheumatic diseases .Clin Rheumatol 1983; 2:259-63.
13. Ostalwska A,BirknerE,Wiecha M, Kasperczyk S,Kaspercyzk A,Kapolka D et al. Lipid peroxidation and antioxidant enzyme in synovial fluid of patients with primary and secondary Osteoarthritis of the knee joint. Osteoarthritis Cartilage 2006; 14:139-45.
14. Campo GM, Avenoso A, Nastasi G, Micali A, Prestipino V, Vaccaro M, et al. Hyaluronan reduces inflammation in experimental arthritis by modulating TLR-2 and TLR-4 cartilage expression. Biochem Biophys Acta 2011 Sep; 1812 (9):1170-81.
15. Altman R, Asch E,Bloch D,Bole G, Borenstein D, Brandt k et al. Development of criteria for the classification and reporting of Osteoarthritis: Classification of osteoarthritis of the knee. Arthritis Rheum 1986; 29:1039-1049.
16. Trinder P. Ann.Clin.Biochem 1969; 6:24- 27.
17. Woolliams JA, Wiener G, Anderson PH, Mc Murray CH. Research in Veterinary Science 1983; 34:253-256.
18. Paglia, D.E and Valentine, W.N., J.Lab. Clin. Med 1967; 70:158.
19. Shanghai Bluegene Biotech CO., Ltd. Human Hyaluronic acid Elisa kit and Human keratan sulphate Elisa kit. Cat number- E01H0004, E0590HU. lot #20110815.
20. Hooiveld, M.J.J., Roosendaal G, Vanden Berg, H.M, Lafeber et al. J.W.J Poster 47th Annual meeting. Orthopaedic Research Society, San Francisco, California. 2001: P- 455.
21. M Maneesh, Jayalekshmi, T Suma, S Chatterjee, A Chakrabarti, T.A.Singh. Evidence of oxidative stress in Osteoarthritis; Indian Journal of clinical Biochemistry 2005; 20(1):129-3.
22. Kalaci A, Yilmaz HR, Aslan B, Sogut S, Yanat AN, Uz E .Effects of hyaluronan on nitric oxide levels and superoxide dismutase activities in synovial fluid in knee osteoarthritis. Clinical Rheumatol 2007 Aug; 26(8):1306-11.
23. Sautin, Yuri; Johnson, Richard. "Uric Acid: The Oxidant-Antioxidant Paradox". Nucleosides, Nucleotides and Nucleic Acids 2008; 27 (6): 608–19.
24. G.M.Rao, Sreelaxmi, A.Naser, Vandana. Reduced blood glutathione in erythrocyte stability in osteoarthritis. Biomedical Research 2005; 16(3):201-203.
25. Y Sun, H Brenner, S Sauerland, K.P.Gunther, W.Puhl, T.Sturmer. Serum uric acid and patterns of radiographic osteoarthritis – the Ulm Osteoarthritis Study. Scandinavian Journal of Rheumatology 2000; 29 (6):380-386
26. Dahl B, Dahl LM, Engstrom-Laurent A, Granath K. Concentration and molecular weight of sodium hyaluronate in synovial fluid form patient with Rheumatoid Arthritis and other arthropies .Ann Rheum Dis1985;44:817-22
27. Balazs, EA. The physical properties of synovial fluid and the specific role of Hyaluronic acid. In: Helfet AJ, edition.In. In Disorders of the knee.Philadelphia: JB Lippincott; 1982.pp.61-74.
28. Van den Bekerom MP, Mylle G, Rys B, Mulier M. Viscosupplementation in symptomatic severe hip osteoarthritis: a review of the literature and report on 60 patients. Acta Orthop Belg 2006; 72 (5):560-568.
29. Mehrabaan F, Finnegan CK, Markowitz RW.Serum keratan sulfate quantitative and qualitative comparison in inflammatory versus no inflammatory arthritides. Arthritis Rheum 1991; 34:383-92.
30. Campion GV, Mc Crae F, Schnitzler TJ, Lenz ME, Dieppe PA, Thonar EJ. Levels of keratan sulphate in the serum and synovial fluid patients with osteoarthritis of the knee .Arthritis Rheum1991; 34:1254-9.
31. Wakiteni S, Nawata M, Kawaguchi A, T Okabe, k.Takaoka, T.Tsukchiya, et al. Serum keratan sulphate is a promising marker of early cartilage breakdown. Rheumatology 2007 Nov; 46(11):1652-6.
32. Thonar EJ, Lenz ME, Klintworth GK, Caterson B, Pachman LM, Glickman P, et al. Quantification of keratan sulfate in blood as a marker of cartilage catabolism. Arthritis Rheum 1985; 28:1367-76.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License