IJCRR - 5(20), October, 2013
Pages: 22-31
Date of Publication: 02-Nov-2013
Print Article
Download XML Download PDF
MIKANIA SCANDENS (L.) WILLD. - A PLANT BASED FUNGICIDE AGAINST ALTERNARIA LEAF SPOT DISEASE IN SESAMUM ORIENTALE L. : SOME OBSERVATIONS
Author: Lubaina A.S., Murugan K.
Category: General Sciences
Abstract:Background: Phytochemicals are a large group of plant-derived compounds responsible for several physiological functions including defence. Polyphenols are ubiquitous in plants. Aim: Formulation of a plant based control in sesame against Alternaria leaf spot disease. Methodology: Methodologies include culturing of pathogen, aqueous extraction of Mikkania scadens by hydrodistillation, quantification of phenols and IR finger printing of infected plants. Results: Inoculation of 20 \?l conidial suspension of Altenaria sesami followed by treatment of sesame plants with 7.5% M. scandens extract delayed the disease outbreak and increased the resistance. Remarkable level of free and cell wall-bound phenolics was noticed in the M. Scadens treated sesame plants. Subsequently, the total phenol was fractionated revealed the presence of a pool of phenolic acids and were identified by comparing with the peaks of the internal standards. Infra-red spectral analysis showed the bands around 3370 cm-1 represent O-H and N-H stretching vibrations that are mainly generated by alkaloids. The bands between 3000 and 2800 cm-1 represent C-H stretching vibrations of triglycerides. Bands between 1800 and 1500 cm-1 contained amide-I, amide-II bands, but overlapped with other absorption bands within this region. Amide-III, the functional group of nucleic acids also contributed to these absorption bands in the leaves. Conclusion: Plant based control measures are an eco-friendly method that effectively induce resistance in the plants.
Keywords: Alternaria sesami, Sesamum orientale, Mikania scandens, Biocontrol, Phenolic acids.
Full Text:
INTRODUCTION
Phenolics are low molecular secondary metabolite, essential for growth, development and defense in plants1. Wound, pest, pathogen attack, herbivory, and infection by symbionts such as Rhizobium, mycorrhiza can induce the synthesis and release of different types of phenolics. These metabolites are potential to function as phytoalexins, phytoanticipins and gene inducers related with many physiological events including nodulation in legumes2-3.
Defences include constitutive barriers such as thick lignified cell walls, waxy coating or cuticularization, sclerification, suberification and formation of trichomes. These modifications not only protect the plant from microbial invasion, but also provide strength and rigidity. In addition to these, plants have the ability to sense invading organisms and react with inducible defences including the formation of toxic chemicals such as quinones, catabolic enzymes like NADPH oxidase, superoxide dismutase, reactive oxygen species/free radical formation and cellular oxidative burst. In some cases, pathogen is capable of suppressing the initial defence, plants may respond with another line of defence: the hypersensitive response (HR). The HR is characterized by deliberate plant cell suicide at the site of infection4-5.
Alternaria leaf spot disease caused by Alternaria sesami a single major threat to sesame leads to poor growth followed by reduction in yield. The common practice to control the disease is the application of chemical fungicides causing environmental problem. This has stimulated the investigation to find an alternative strategy for the control of the disease6. Mikania scandens (L.) Willd. (Asteraceae), a herbaceous climbing weed growing throughout the plains of India. Aqueous leaf extracts of this plant have been used in folk medicine to treat many skin disorders7. The present study aims to evaluate the induction of induced resistance in Sesamum orientale against A.sesami by the application of plant based control by- Mikania scandens.
MATERIALS AND METHODS
Plant materials
Sesame cultivar Thilarani (susceptible to A. sesami) and Sesamum orientale L. var. malabaricum, wild species seeds are collected from Regional Agriculture Research Station, Kayamkulam, Kerala.
Collection and isolation of the fungus
Leaves of sesame showing typical symptoms mainly on leaf blades as small, brown, round to irregular spots caused by A. sesami were collected from University of Agricultural Sciences, Bangalore. The fungal pathogen was isolated from the infected tissue and made pure culture using standard protocols and the identity was confirmed from Division of Plant Pathology, Indian Agricultural Research Institute, New Delhi.
Preparation of Mikania scandens leaf extract
Healthy leaves of Mikania scandens collected were thoroughly washed in running water followed by rinsing with distilled water and were shade dried at room temperature. 10 g of powdered dried leaves of Mikania scandens were hydro distilled in 100 ml sterile distilled water at 100?C for an hour and allowed to stand for 24 hrs at 4?C. The mixture was centrifuged at 4000 rpm for 10 min. and the extract (10%) was filtered through Whatman No.1 filter paper into a sterile filter flask and stored in sterile condition for further use.
Mikania scandens leaf extracts treatment
Mature sesame plants were inoculated with 20 µl of A. sesami conidial suspension (1×103 conidia ml-1) from pure culture and were covered with plastic bags for 24 h and placed under darkness to provide maximum humidity for penetration of fungus into host cells. This is followed by spraying 7.5% M. scandens aqueous leaf extract using a pneumatic hand sprayer from one week after fungal inoculation and repeated once in every 7 days for a period of 21 days. Control was maintained by infecting the Thilarani and wild sesame with 20 µl conidial suspension of A. sesami without any plant extract treatments. After the treatment, the leaves were harvested for all analysis.
Isolation and estimation of total phenolics
Free and cell wall-bound phenolics were extracted from (0.5g) leaves of M. scandens treated sesame (both the cultivar and wild) and control plants in 50% methanol (12 v/v) for 90 min at 80 oC according to the methodology of Haddadchi and Gerivani8. The absorbance of samples was measured at 725 nm.
Reverse Phase High Performance Liquid Chromatography (RP-HPLC) of phenols Phenolic components of the extract were further fractionated following the method of Sara Canas et al9 with appropriate standard phenolic acids. Phenolic acids in the sample were identified by comparing with the retention time of the standards.
IR spectroscopy
The leaves of each accession (approximately 3-4 cm) taken from fresh plants were pooled as one sample. The samples were immediately dried in an oven for 2 days at 60°C. Tablets for FTIR spectroscopy were prepared in an agate mortars, by mixing leaves powder (2 mg) with KBr (1:100 p/p). At least three spectra were obtained for each sample10. The band position of functional groups was monitored with Knowitall 7.8 software.
RESULTS AND DISCUSSION
Free and wall bound phenolic content
Aerial application of M. scandens leaf extract led to remarkable increase in both free and wall bound phenolics compared to control. Maximum free (1288 and 1394 μg gallic acid /g fresh wt. in Thilarani and wild respectively) and bound phenolics (1139 and 1278 μg gallic acid /g fresh wt. in Thilarani and wild respectively) were observed in sesame leaves treated with the aqueous extract. The results suggest that the topical application of the extract induce host secondary metabolism i.e. activating the phenyl propanoid pathway leading to the synthesis of polyphenols and there by activating defence mechanism in the host against infection. Bordbar et al11 reported a time dependent formation of phenolics in apple upon treatment with Trichoderma isolates.
Fractionation of total Phenols
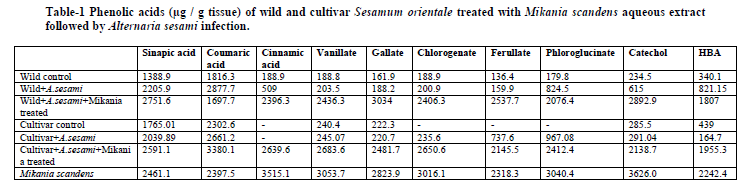
HPLC chromatogram of the treated wild and cultivar sesame showed a significant increase in the levels of phenolic acids such as sinapate, cinnamate, vanillic, gallic acid, chlorogenate, ferulate, p-hydroxybenzoic acids, paracatechol and coumarate compared to control suggesting its potentiality towards resistance (Table-1). It is evident from the chromatogram that (Fig- A & B) phenolic extracts of leaf samples contain the peaks of most of the standards indicating the functional compartmentation of phenolic acids induced by the plant based extract.
Pre-formed antibiotic compounds such as phenolic and polyphenolic compounds are common in plants and play an important role in defence against pest and pathogens12. The first proven report of phenolics providing disease resistance was the case of onion scales accumulating sufficient quantities of catechol (I) and protocatechuic acid (II) to prevent onion smudge disease caused by Colletotrichum circinans. The coloured outer scale of resistant onion varieties contain surplus of these phenolics to inhibit spore germination of C. circinans to below 2%, while susceptible varieties lack these compounds with high % of germination rate (90%) 13 . Similarly, adequate levels of chlorogenic acid account for the resistance of potato tubers against Streptomyces scabies, Verticillium alboatrum and Phytophthora infestans 14, while, its lower profile accelerate P. infestans and Fusarium solani var. Coeruleum growth. Low concentrations of benzaldehyde inhibited spore germination of Botrytis cinerea and Monilia fructicola 15
Hydroxycinnamates such as p-coumaric acid are major components of plant cell walls and are esterified with lignin, a key cell wall component16. Hydroxybenzoides have widespread application in food, pharmaceutical, fragrance and flavour industries17. Rot fungi are able to convert p-coumaric to p-hydroxybenzoate which has considerable ecophysiological and defence related implications18. Ferulic acid has immense role in plant cell wall formation and also an effective antioxidant and anticancer compound19.
Furthermore, fungicidal activity by phenolics leads to condensation reactions with proteins, amino acids, nucleic acids forming brown non palatable melanin like compounds. This reaction results in the formation of an impermeable barrier in the host plant, and also a decrease of nutrients essential to the fungal development. Beckman et al20 showed that oxidation products of 3-hydroxy-tryptamine, leads to inhibition of invasion of Fusarium oxysporum. Secondary responses also include the release of toxic phenols that are normally stored as less toxic glycosides in the vacuoles of the plant cells, lignin synthesis, trichomes, and phytoalexins.
Peters and Verma 21 reported that plant phenolics induce gene leading to the transfer of genetic information to the entire plant. Stachel et al22. identified monocyclic phenolics that induce expression of several virulence genes of A. tumefaciens. Induction was also found to depend on the structure of the phenolic compounds. Compounds related to acetosyringone were found to be effective inducers including chalcones. So the application of M. scandens leaf extract induces the phenyl propanoid pathway in sesame with the production of phenols which in turn induce resistance. Singh et al 23. reported that treatment of chickpea with Pseudomonas spp. increased the synthesis of cinnamic, ferulic and chlorogenate which showed antifungal effects against Sclerotium rolfsii.
IR spectral analysis
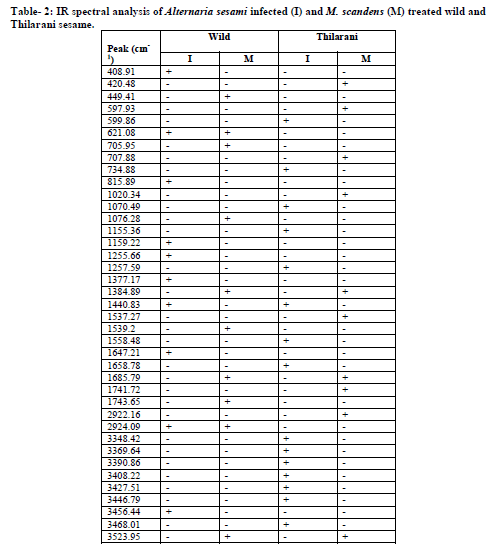
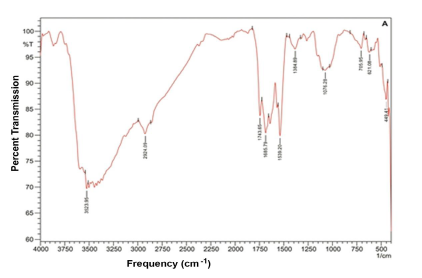
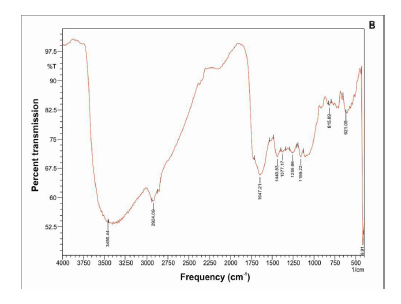
Specific spectral signatures and re?ectances are characteristic feature of plant – pathogen interaction. A strong broad absorption band around 3360-3390/ cm found in all samples may be due to the presence of hydrogen bond N-H stretching, characteristic of amino acids. The absorption band 2924 / cm, corresponding to C-H stretching of the CH2 groups, indicates the presence of various amino acids, this band may also be characteristic for the presence of aliphatic CH groups in these compounds. The absorption band at 1735/ cm, characteristic of C=O stretching, indicates the carbonyl groups. Bands 1643 /cm and 1560 /cm coupled with the presence of the band around 3368 /cm may be taken as indication of the presence of amino acids. The absorption band appears at 1439 /cm due to the aromatic ring C-C stretching. The absorption bands 1240/ cm and 1052 / cm are due to the stretching vibration of C-O group of esters and phenols. The band 606 / cm belong to C-C ring bending coumarin structure. Unique spectral biomarkers were found in M. scandens treated sesame compared to infected (Fig-2A, B, C & D).
Fourier transform infrared (FTIR) spectroscopy has the ability to quickly identify the presence of specific metabolites in plant materials.In FT-IR spectral peaks corresponding to diseased leaves, absorption bands occur at 3368/ cm, 3359/ cm, 2925/ cm, 1735 /cm, 1643/ cm, 1549/ cm, 1439/ cm, 1425 /cm, 1246/ cm, 1035 /cm and 606/cm (Table 2). Recently, the use of FT-IR spectroscopy in metabolomics has become increasingly important within the phytopathology field.
CONCLUSION
Plants respond to diverse environmental signals with a bewildering array of responses, which use constitutive and induced resistance affecting the susceptibility/resistance of the infected plant. Plant based products can induce the secondary metabolic pathway by activating the corresponding genomes. FTIR spectroscopy shows these changes of the plant before visible symptoms in the plants and therefore, can be used as a predictive method in the future.


References:
- Vogt T. Phenylpropanoid biosynthesis. Mol. Plant. 2010;3:2–20
- Mandal S.M, Mandal M, Das A.K, Pati B.R, Ghosh A.K. Stimulation of indoleacetic acid production in a Rhizobium isolate of Vigna mungo by root nodule phenolic acids. Arch Microbiol. 2009; 191: 389-393.
- Mandal S.M, Chakraborty D, Dey S. Phenolic acids act as signaling molecules in plant-microbe symbioses Plant Signal Behav. 2010; 5(4) 359-368.
- Khurana P.S.M, Pandey S. K, Sarkar D, Chanemougasoundharam A. Apoptosis in plant disease response: A close encounter of the pathogen kind . Curr. Sci. 2005; 88: 740-752.
- Jeandet P, Clément C, Cordelier S. Modulation of phytoalexin biosynthesis in engineered plants for disease resistance. Int. J. Mol. Sci. 2013 14 (7) 14136-14170.
- Itako A.T, Schwan-Estrada K.R.F, Stangarlin J.R, Tolentino J.B, Cruz M.E.S. Control of Cladosporium fulvum in tomato plants by extracts of medicinal plants. Arquivos Do Instituto Biológico. 2009; 76: 75-83.
- Perez-Amador M.C, Ocotero V.M, Balcazar R.I, Jiménez F.G. Phytochemical and pharmacological studies on Mikania micrantha H.B.K. (Asteraceae). Int. J. Exp. Bot. 2010; 79: 77-80.
- Haddadchi G.R, Gerivani Z. Effects of phenolic extracts of canola (Brassica napuse L.) on germination and physiological responses of soybean (Glycin max L.) seedlings. Int. J. Plant Prod. 2009; 3(1): 63-73.
- Sara Canas, Nelson Grazina, Pedro Belchior A, Isabel Spranger M, Bruno de Sousa R. Modelisation of heat treatment of portuguese oak wood (Quercus pyrenaica L.). Analysis of the behaviour of low molecular weight phenolic compounds, Ciencia Tec Vitiv. 2000; 15: 75.
- Batten J. D. Plant analysis using near infrared reflectance spectroscopy: the potential and the limitations, Australian J. Exp. Agr. 2009; 38(7): 697 – 706.
- Bordbar F.T, Etebarian H.R, Sahebani N, Rohani H. Control of postharvest decay of apple fruit with Trichoderma virens isolates and induction of defense responses. J. Plant Prot. Res. 2010; 50(2): 146-152.
- Boudet A.M. Evolution and current status of research in phenolic compounds. Phytochem. 2007; 68: 2722–2735.
- Walker J.C, Stahmann M.A. Chemical nature of disease resistance in plants. Ann. Rev. Plant Physiol. 1955; 6: 351-366.
- Lee S, Le Tourneau D.J. Chlorogenic acid content and verticillium wilt resistance of potatoes. Phytopathol. 1958; 48: 268-274.
- Wilson C.L, Wisniewski M.E. Biological control of postharvest diseases of fruits and vegetables: An emerging technology. Annu.Rev. Phytopathol. 1989; 27: 425-441
- Mac Adam W, Grabber J. H. Relationship of growth cessation with the formation of di ferulate cross-links and p-coumaroylated lignins in tall fescue leaf blades. Planta 2002; 215(5): 785–793.
- Chakraborty D, Sircar D, Mitra A. Phenylalanine ammonia-lyase-mediated biosynthesis of 2-hydroxy-4-methoxybenzaldehyde in roots of Hemidesmus indicus. J. Plant Physiol. 2008; 165(10): 1033–1040.
- Sachan A, Ghosh S, Mitra A. Transforming p-coumaric acid into p-hydroxybenzoic acid by the mycelial culture of a white rot fungus Schizophyllum commune, African J. Microbiol. Res. 2010; 4(4): 267–273.
- Renger A, Steinhart H. Ferulic acid de hydro dimers as structural elements in cereal dietary fibre. European Food Res. Technol. 2000; 211(6): 422–428.
- Beckman C.H, Mace M.E, Halmos S, Mc Gahan M.W. Physical barriers associated with resistance in Fusarium wilt of bananas. Phytopathol. 1961; 51: 507-515.
- Peters N. K, Verma D. P. S. Phenolic compounds as regulators of gene expression in plant-microbe interactions. Molecular Plant-Microbe Interact. 1990; 3 (1): 4-8.
- Stachel S.E, Montagu M.M.E, Zambryski P. Identification of the signal molecules produced by wounded plant cells that activate T –DNA transfer in Agrobacterium tumifaciens. Nature. 1985; 318: 624-629.
- Singh U.P, Samara B.K, Singh D.P. Effect of plant growth-promoting rhizobacteria and culture filtrate of Sclerotium rolfsii on phenolic and salicylic acid contents in chick pea (Cicer arietinum L.). Curr. Microbiol. 2003; 46: 131-140.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License