IJCRR - 6(3), February, 2014
Pages: 35-45
Print Article
Download XML Download PDF
PROTEIN ISOLATES : PRODUCTION, FUNCTIONAL PROPERTIES AND APPLICATION
Author: Umar Garba, Sawinder Kaur
Category: Healthcare
Abstract:Protein isolates are refined form of protein containing the greater amount of protein with greater digestibility. Nowadays it's the major source of cheap proteins especially for athletes, bodybuilders, vegetarians, and has gained wide application in various beverages and dairy industries, and infant foods due to its different functional properties. It is widely produced from deoiled cake of legumes such as peanuts, cowpeas, soybeans, etc and animal sources like fish and milk by a combination of isoelectric and alkaline precipitation using H2SO4 and NaOH, followed by centrifugation to obtain the pure isolates which can further be dried to powdered form of about 90% protein. The problem of antinutritional factors in legumes can be successfully reduced by these processing methods.
Keywords: Protein Isolates, isoelectric precipitation, Ultrafiltration, antinutritional factor
Full Text:
INTRODUCTION
Proteins are nitrogen-containing compounds made of up amino acids unit. They are the major structural component of muscles and other tissues in the body. They are also component of hormones, enzymes and haemoglobin (Jay et al. 2004). Proteins composed of twenty different amino acids linked together by a peptide bond and the resulting chain is called polypeptide. They have similar basic structure but differ in their side chains. This difference in side chains gives the proteins their specificity and functionality. These amino acids are classified as essential (those that cannot be synthesis by body but rather taken as supplement from diet), and nonessential (that can be synthesis by the body) (Clark 2003). Demand for the relatively cheap sources of protein that can be incorporated to value –added food products is increasing worldwide, and numerous researches are still going on various sources of plant proteins that may help in improving the nutritional value of food products at low cost (Gurpreet et al. 2006). Proteins are available in different varieties of dietary sources including animals, plants origin, and from highly marketed spot supplement industry. Typically, all dietary animal proteins (e.g. eggs, milk, meat, fish and poultry) are considered complete protein because they contain all essential amino acids. Proteins from vegetable sources (such as legumes, nuts and soy) are incomplete proteins since they are lacking one or two essential amino acids (Hoffman and falvo, 2004). Assessing the quality of protein is important when considering the nutritional benefit that it can provide. Protein quality of food is the ability of the food to meet the nutritional requirement of an individual species. It is indicated by how well the protein is digested, absorbed, and utilised for the growth and sustenance of the body (Wardlaw et al. 1996). The quality of protein can be determined by assessing its essential amino acid profile. Numerous methods to determine the quality of proteins include : ( 1) Protein efficiency ratio (PER) which is achieved by measuring the animal growth after feeding with the test protein. The weight gained in grams per gram of protein consumed is calculated and then compared with the standard value of 2.7 (standard value of casein protein). If the value is greater than 2.7, the food is regarded as excellent source of protein. However, the result mostly provides a measure in rat but does not provide a strong correlation to human growth needs. (2)Biological value is calculated by dividing the nitrogen used for tissue formation and the nitrogen absorbed from the food and then multiplied by 100 and express as percentage of nitrogen utilised. This gives an idea of how efficient the body utilises the protein consumed in the diet. Higher value indicates that food supplies high essential amino acids. (3) Net protein utilisation (NPU) is almost identical to BV except that direct measure of retention of absorbed nitrogen is involved. However, the BV is calculated from nitrogen absorbed while NPU is calculated from nitrogen ingested (Jay et al. 2004). (4) Protein digestibility Corrected Amino acids Score, this, according to joint (FAO/WHO, 1990) could be determined by expressing the content of the first limiting essential amino acid of the test protein as a percentage of the content of the same amino acid content in a reference pattern of essential amino acid. The reference values used were based on essential amino acid requirement of preschool-age children, which was corrected for true faecal digestibility of the test protein. The value obtain was known as protein digestibility corrected amino acid score (PDCAAS).
Protein isolates
Isolate are the most refined form of protein products containing the greatest concentration of protein but unlike flour and concentrates contains no dietary fibre. Isolates originated from United State around 1950s (Jay and Michael 2004). They are very digestible and easily incorporated into different food products. Protein isolates are nowadays believed to have played a major role in the development of new class of formulated foods. It is high concentration of protein with the advantage of colour, flavour and functional properties make it an ideal raw ingredient for used in beverages, infant foods and children milk food, textured protein products and certain types of specialty foods (Olaofe 1998). Protein isolates have been developed from a variety of legumes among which are soy bean, peanut, canola, cashew nut, almonds, sesame, pinto and navy beans (Seyam et al. 1983). Extraction Methods Isoelectric precipitation: - - The isoelectric point (pI) of a protein is the pH where the net charge on the protein is zero. At this point, there exist no electrostatic repulsion keeping them apart, thus protein aggregate and precipitate. Proteins have different isoelectric point because of their different amino acid sequence, and therefore, they can be separated by adjusting the pH of a solution. When the pH is adjusted to the isoelectric point of a particular protein it precipitates leaving the other protein in the solution (McClements 2013). A more recent study on isoelectric precipitation of soya protein confirmed that both extraction condition and the type of acid used for isoelectric precipitation had little effect. However, it was found that an isoelectric point of around pH 4.2 leads to high proportion of protein precipitated out (Smith et al. 1939). Alkaline extraction: It was found that alkaline reagents were more effective in extraction of protein from food legumes. However, several changes such as destruction of lysine, formation of lysine-alanine, racemisation may occur during alkali extraction and reduce the protein quality (Tobin et al. 1978). Hence, a better technology to produce protein isolate and concentrate with maximum yield and minimum or no antinutritional factors need to be developed. Ultrafiltration method: - This is a potential membrane processing for extraction of protein, a solution of protein is placed in a cell containing a semi-permeable membrane, and pressure is applied. Small molecules pass through the membrane, whereas the larger ones remain in the solution. The semi-permeable membranes with cut-off points ranging between 500 and 300,000 are mostly available (McClements 2013). The extraction of soy protein isolate by this method was first reported over twenty years ago. The Ultrafilteration system provides a commercially feasible alternative to the existing isoelectric precipitation method of soya beans
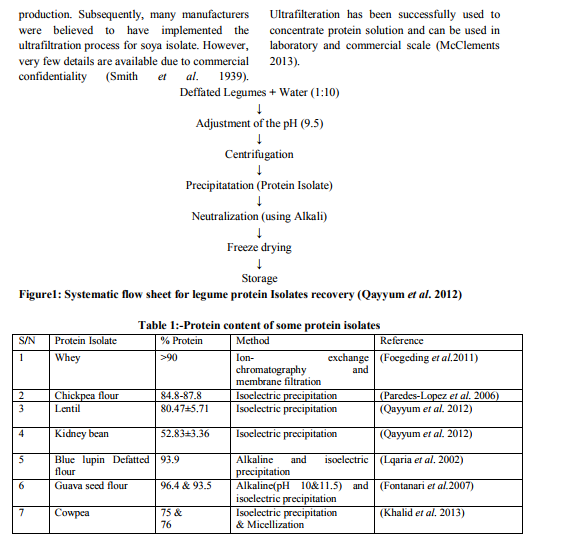
Protein isolate from different plant and animal sources
animal sources Proteins that are utilised in food processing are of various origins, and can roughly be classified into animal proteins (gelatins), vegetable proteins (e.g. peanut protein, soy protein, wheat proteins, Almond protein, canola meal protein etc.), and animal derived protein (e.g. milk proteins). However, many vegetable proteins require processing to provide food material having acceptable functional properties such as emulsification, oil and water absorption, texture modifications, colour control and whipping properties, which are primarily attributed to the protein characteristics. Many plants have attracted a deal of interest as a source of lowcost protein to supplement human diet, this include among others soybeans and peanut (Ogunwolu et al. 2009).
Whey protein isolates (WPI)
Whey is the liquid by-product of cheese which can further be processed into a spray dried products like instance whey protein concentrates (WPC), whey protein isolate (WPI) or whey protein hydrolysate (WPH) (Brucic et al. 2009).By definition from the code of Federal Regulation, is the liquid substances obtained by separating the coagulum from milk or cream in the cheese making. During cheese making, the whey protein remains in the serum phase which represents about 20 percent of the milk protein (Kimberlee 2012). In the production of whey protein isolates, significant amount of fat and lactose get removed, as a result of which individual with lactose-intolerant can safety consume these products (Akintayo et al 1999).Whey proteins are widely used as ingredients in different foods (dairy, meat and bakery products) due to their unique functional and nutritional properties (Brucic et al. 2009). The heat sensitivity of whey protein may present a major challenge in some formulations. Consequently, the Dairy Research Institute, established under the leadership of America’s dairy farmers through the dairy check off program, has supported a variety of researches that aims to improve whey protein’s performance in higher heat processing (Kimberlee 2012). Beta-lactoglubin and α- lactalbumin are the major whey protein responsible for heat stability characteristics of ingredients such as WPC and WPI (Vardhanabhuti and Foegeding 2008).The relationship between sugar addition and heat stability of whey protein isolate have been studied by many researchers. Sucrose addition was found to increase gelation temperature and gel strength of WPI and bovine serum albumin (Rich and Foegeding 2000). Adding glycerol improved heat stability of WPI and decreases turbidity and protein gelation (Kulmyrzaev et al. 2000).Thermal denaturation temperature of WPI was also found to increased by addition of sorbitol more effectively than glycerol (ParedesLopez 2006).Many food application contains sugars or sugar alcohols, and their presence can help to improve the heat stability of whey protein ingredients by preventing the formation of large aggregates and providing better clarity in application such as beverages (Kimberlee 2012). Fish Protein isolates (FPI) To solve the problem of utilisation of unconventional raw material (dark muscle fish, fatty fish) and also fish by-products (fish trims, fish frames etc.) a process was developed to economically develop a functional protein isolates from these kind of raw materials. This technology uses the pH dependant’s solubility properties of fish muscle proteins for their separation and recovery from other components of muscle that are not desirable in a final product. Fish protein isolate is a protein concentrate which is prepared from fish muscle without retaining the original shape of the muscle. It is not generally consumed directly, but used as raw material for production of other value added products. Fish protein isolate does not retain the original shape of muscle, and is normally utilized as ingredient for the production of value added products. It is still a good source of protein for the production of ready to eat fish products. The overall processes involved are simple. The proteins of the muscle tissue are first solubilised. The solubilisation can be accomplished by addition water with alkali added to approximately pH 10.5 or higher, or with acid added to about pH 3.5 or lower. It is usually necessary to choose the pH at which the consistency of the solution decreases to a value that allows the removal of undesirable material. The mixture is then centrifuged, and due to density differences the oil rises to the top and can then be removed. Other insoluble impurities such as bone or skin are also sedimented at this stage. The muscle protein are then precipitated and collected by a process such as centrifugation (Shaviklo 2006)
Peanut protein isolates (PPIs)
Peanut contains 26-29% protein with good nutritional quality. Peanut proteins are used for their functional properties (emulsification, forming) or for their nutritional properties in different food products. They are also used for human nutrition in developing countries to supplement cereals, beverages and skim milk. Peanut protein isolate can be prepared from the defatted peanut cake or powdered by macerating with high salt phosphate buffer (20 mM Na2HPO4, 2 mM KH2PO4, 5.4 mM KCl, 1M NaCl, pH 7.4), followed by centrifugation and supplementing the supernatant with (NH4)2SO4 to 90% saturation. After centrifugation, the pellet can be dialyse against distilled water overnight at 4oC and then freeze-dried (Mouecoucou 2004). Soy protein isolates (SPIs) Soy protein isolate is a common isolate. It has high protein content of about 90%. It is made out of defatted soy meal by removing most of the fat and carbohydrates (Seyam 1983). Soybean is crushed into oil and defatted meal. The meal is usually used as animal feed, while smaller amount is further processed into food ingredients including soy flour, protein concentrate, protein isolates and textured protein (Kinsella 1976). Soy protein isolate is usually combined with other food ingredients such as vitamins, minerals and flavour in preparation of soy protein shake powder (Seyam 1983). Advance in food technology resulted in the development of a variety of soy product such as concentrates, isolate and extruded-expanded products, this consequently leads to increased utilisation by technically developed regions of the world (Young 1979). The production of soy protein isolate involve solubilising the protein and carbohydrate at neutral or alkaline pH and the recovery of the solubilised protein, by separation and optionally washing and neutralization before drying (Moure et al. 2006). Three steps involved in the processing of soy protein isolates (SPI) are (1)The soy flakes are slurried with water under alkaline conditions (pH 6.8-10 at 27-66 oC using sodium hydroxide and other alkaline substances approved for food used) so that the protein and the oligosaccharides can dissolve into the solution. The protein solution is then separated from the insoluble residue by centrifugation, (2) the supernatant containing the protein and sugars is then acidified to isoelectric pH 4.5 ( where the solubility of proteins is minimal), using hydrochloric acid (HCl). This leads to the precipitation of protein as curd, (3) the solubility of the precipitated protein is restored by neutralizing to alkaline pH of 6.5-7.0 after re
diluting with fresh water or spray dried in its acidic form and packed in multilayer paper bags (Lusas et al. 1995, and Anon 2008). Canola protein isolates (CPIs) According to USDA 2010, Canola meal has been the second largest feed meal after soybean meal. It has a good amino acid profile with a well balance amino acid composition although it has found only marginal used in the food industry, due to the presence of anti-nutritional factors. The vast majority of canola protein isolates are prepared by alkaline extraction method fallowed by isoelectric precipitation. Although, this extraction method generates high yield of nitrogen, but the isolate produced by this method have been found to have poor solubility and digestivity, this is most probably due to the nature of proteins constituent of canola meal which consist of alkaline-soluble fraction that can be easily denatured during the extraction process. CPI thus possesses generally an unacceptable food-functional properties including poor water holding, gelling and oilbinding, foaming and emulsification properties. Many studies have been carried out to modify the properties of this isolate e.g. by succinylation, acylation and enzymatic hydrolyses. However, enzymatic hydrolyses has been prepared because it’s less effect than chemical method (Alashi 2011).
Chick pea protein Isolate
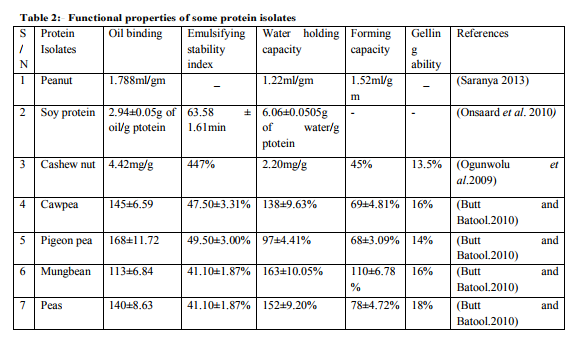
Chick pea protein is the world’s third largest pulse crop in term of area, grown mostly in West Asia and Mediterranean region. It is one of the major vegetable proteins. Many functional properties of this protein isolate has been studied whereas, information on gelation properties of chick pea protein isolate was scares. The chick pea protein isolate dispersed with sodium and calcium salts showed different rheological behaviour at different ionic strength and pH. Increasing the ionic strength of dispersion could strengthen the gelation properties of CPI under acidic conditions, however reduced the elastic parameters of CPI at pH of 7.0 (Zhang 2007).Cashew nut protein isolate Cashew has considerable economic importance because its components have numerous economic uses. The kernel has high food value with about 40-57% oil and 21% protein content. A cashew kernel meal contains about 42% crude protein, a low crude fibre and 0.5% and 0.2% calcium and phosphorous, respectively, which is comparable to that of peanut composition, which has been used for peanut protein isolate and concentrate. Protein isolates and concentrates can be obtained from defatted cashew nut powder by both alkaline extraction-isoelectric precipitation (IP) and alkaline extractionmethanol precipitation (MP). Cashew nut protein isolates has water and oil absorption capacities of 2.20ml/g and 4.42ml/g respectably, emulsifying stability index (447%), foam capacity and stability (45% and 55%, respectively ), and low gelation capacity of (13.5%) (Ogunwolu 2009).
Functional Properties of Protein Isolate Bulk density:-
- Bulk density is an important parameter that determines the packaging requirement of a product. It signifies the behaviour of a product in dry mixes and can varied with fineness of particles (Butt and Batool 2010).The bulk density of protein isolate of sources can be determined by taking 10 grams of protein isolate into 100 ml graduated cylinder and taping several time on a laboratory bench until the isolate settled. The values recorded can be express as g/cm (Olaofe 1998, and Okaka and Potter 1977).Protein solubility :- Protein solubility is a useful indicator for the performance of protein isolates incorporated in the food system and also the extent of protein denaturation because of heat and chemical treatment at different pH (Horax 2004).To determine the protein solubility, the protein isolates (250 mg) should be homogenized in 20 ml of 0.1 M NaCl at pH 0f 7.0 for 1h fallowed by centrifugation at 10,000×g for 30 min. Nitrogen content should then be determined in the soluble fraction and the solubility can be expressed as the percentage total nitrogen of the original sample to that of soluble fraction (Morr 1985). Foaming Capacity and stability: - The foaming properties are used as an index of the whipping characteristics of the protein isolate (Mwasaru 1999). The capacity and stability of foams can be determined by dispersing 50ml of 3% (w/v) of protein isolate sample in distilled water and transfer immediately into a graduated cylinder, the volume should be recorded before and after whipping. The foaming capacity can be expressed as the percentage volume induced by whipping. The change in volume of foam after 60 min of standing at room temperature is recorded as foam stability (Lin 1974). Water/oil absorption: Proteins has both hydrophilic and hydrophobic properties therefore, can interact with water and oil in foods (Butt and Batool 2010). The lower water absorption capacity can be due to less availability of polar amino acids (Kuntz 1971), and the low fat absorption may be due to the presence of large proportion of hydrophilic and polar amino acids on the surface of the protein molecules (Sathe et al.1982). Water absorption is determine by mixing approximately 3g of protein isolate with 25ml distilled water in a pre-weighed centrifuge tubes, the tubes are stirred and centrifuge for 25 min at 3000×g for 30 min interval. The supernatant is carefully decanted and the centrifuge tubes containing the protein isolate are re-weighed. Water absorption is express as the number of grams of water absorbed per gram of sample (Sosulski et al. 1976). High water absorption of protein isolates help to reduce moisture loss in package bakery products (Lin 1974). Oil absorption can also be estimated by mixing 0.5g of protein isolates with 6ml corn oil in preweighed centrifuge tubes. The tubes are stirred for like one minutes to get complete dispersion of the sample in oil. After 30 min holding time, samples are centrifuge at 3000×g for 25 min, the supernatant be decanted and the tubes inverted for 25 min to drain the oil before re-weighing. The oil absorption is express as gram of oil absorbed per gram of protein isolate (Sosulski 1976). The high oil absorption is essential in the formulation of food systems like sausages, cakesbatters, and mayonnaise and salad dressing (Butt and Batool 2010).
Emulsifying activity and stability:
Proteins are surface active agents that can form and stabilize the emulsion by creating electrostatic repulsion on oil droplet surface (Makri et al. 2005). To determine the emulsifying activity, Protein isolate (3.5g) is homogenised for 30 seconds in 50ml water using homogenizer at approximately 10,000rpm. Corn oil (25ml) should is then be added to the mixture and homogenised for 30 sec. The emulsion is divided into two equal volume aliquots and centrifuged at 1100×g for 5 min, the aliquots is heated for 15 min at 85oC. The ratio of the height of emulsion to the high of liquid layer is noted to calculate emulsion activity. The emulsion stability remains after heating (Naczk et al. 1985). Least gelation concentration: - This is a qualitative parameter that expresses the minimum protein concentration at which the gel does not slide along the test tube walls in inverted position (Young and Scrimshaw 2006).The lower the gelation concentration, the better is the gelling ability of proteins, because protein gels are aggregates of denatured molecules (Akintayo 1999).
Antinutritional factor in protein isolate
Several studies have reported the preparation and functional properties of protein isolates and concentrate (Sathe and Salunkhe 1981). Legumes contain some anti-nutritional factors such as lectins, saponin, haemagglutinin, protease inhibitor, oxalate, goitrogen, phytates, trypsin inhibitor and tannin (Amarowicz and Pegg 2008). These compounds reduce protein digestibility and availability. Some antinutritional in legumes have been reported to have health benefits. Tannin, a polyphenolic compound is reported to possess antioxidative property (Apata and Ologhobo 1997).Very extensive work has been carried out on optimising the conditions for trypsin inhibitor destruction during processing of soya protein isolate (Smith and Circle 1977). Studies had also shown that trypsin and chymotrypsin inhibitory activities were reduced to 75.3% and 84.8% in protein concentrates, 98.0% and 85.4% in protein isolates of Great Northern beans respectively. However, these protein fractions did not contain oligosaccharides of the raffinose family (such as raffinose, stachyose, and verbascose) and hemagglutinating activities. Therefore, most of the antinutritional factors can possibly be eliminated by preparation of protein isolates and concentrates (Sathe and Salunkhe 1981).Specific studies were carried out on isoelectric process for protein isolate, where trypsin inhibitors levels were found to be dependent on the degree of heat treatment to which the raw materials had been subjected and on the pH used for isoelectric precipitation. Trypsin inhibitor levels in isolate produced by ultrafiltration process have also been studied and found to be fairly high; this might possibly be due to complexing of the relatively low weight trypsin inhibiting protein fractions with higher molecular weight proteins (Honig and Wolf 1987). Application of protein isolate in food systems. Protein isolates are the acceptable ingredients for dairy application due to their fine particle size and dispersibility, Emulsification, emulsion stability, colour and flavour are critical in dairy application. Isolates (especially soy proteins) are being used to fortify all type of pasta products such as macaroni, spaghetti, to improve the nutritional value etc (Sipos 2013). Protein isolates are important sources of protein with high lysine content. Isolates from different legumes varied slightly in physiochemical and thermal properties. They are used as proteinaceous ingredients in many food products such as salad dressing, meat products and dessert. Whey proteins are mainly used in beverage applications, due to their health benefits (Kudre 2013). The problem of poor flavour, mouth feel, texture, dryness and flavour associated with the use of soy flour and soy concentrate above 10% has been resolved by using soy isolate in meat loaves, sausage-type products for their emulsion-stabilizing effect, gelation, and moisture retention and improved effects on texture (Kinsella 1976). Soy protein is regarded as textured protein products use both in meat and vegetarian meat analogue industry and thus, has good water holding capability (Riaz 2006). It is often used as meat extenders in comminuted meat products such as patties, fillings, meat sauces, meat balls, etc (Berk 1992). Many soy protein isolates have been developed for providing different functional or physical properties to meet the requirement of various food systems. Soy protein isolates form firm, hard, resilient gels, unlike soy flour and concentrates that form soft and fragile gels (Riaz 2006). The firm-forming ability of soy protein isolates is important in meat products. When heat and pressure are applied the protein films fused together to form a firm, continuous, textured mass that can be sliced and used as meat substitutes. They also vary in their ability to form gels. Some are designed to form gel while others will not form gel at 14% solid content. Protein isolates are utilised in meat and baking application due to their water and fat absorption properties, the value for these properties for protein isolates ranges from 150 to 400% (Sipos 2013). Neutralised protein isolates are often highly dispersible and form gel under appropriate aqueous conditions. They have both emulsifying and emulsion stabilising properties, and are excellent fat and water binders, For this reason, they are widely used in processed meat products, as well as both coarse and fine emulsions (e.g., patties, loaves and sausages) (Sipos 2013). Different protein products such as whey protein, soy protein isolates, wheat gluten, rice bran protein, peanut protein, and cottonseed proteins where investigated for film development (Rhim 1998).
CONCLUSION
Protein isolates shows a lot of potential to combat the problem of malnutrition. The underutilised plant and animal sources can be exploited in order to extract the proteins and make them available for used as food supplements. Research is needed compared the benefit of different protein isolates and their benefit as food supplements.
References:
REFERENCES
1. Akintayo ET, Oshodi AA, Esuoso KO. Effect of ionic strength and pH on the foaming and gelation of pigeon pea (Cajanus cajan) protein concentrates, Food Chem. J 1999; 66: 51-56.
2. Alashi A, Blanchard C, Mailer R, Agboola S. Improving the emulsifying properties of canola meal protein isolate by enzymatic modification. 17th Australian Research Assembly on Brassicas (ARAB), 2011.
3. Amarowicz R, Pegg RB. Legumes as a source of natural antioxidants. Euro J. Lipid Sci. Tech 2008; 110:865-878.
4. Anon, 2008. Soy processing Available at http://dspace.dial.pipex.com/town/park/gfm 11/soya.shtml Assessed 17/09/2010.
5. Apata DF. Ologhobo AD. Trypsin inhibitor and the other anti-nutritional factors in tropical legume seeds. Tropical Sci. 1997; 37:52-59.
6. Berk, Z. Technology of production of edible flours and protein products from soybeans. FAO Agricultural Services Bulletin No. 97, FAO UN, Rome. 1992.
7. Brucic SR, Lelas V, Brucic M, Bosiljkov T, Jezek D, Badanjak M. Thermal gelation of whey protein at different pH values. Proceedings of the 9th International Conference on Chemical and Process Engineering, in May in Rim, Italia: AIDIC, 2009; 17:831-836.
8. Butt MS, Batool R. Nutritional and Functional Properties of Some Promising Legumes Protein Isolates, Pak J Nutri. 2010; 9 (4): 373-379.
9. Clar DR. Improving in vitro protein digestibility and determining protein nutritional quality by modelling [Dessertation. Athens: Georgia Uni. US. , 2003.
10. Foegeding PJ, Luck B, Vardhanabhuti. Whey protein products. In: Encyclopaedia of Dairy Sciences. John W. Fuquay. 2nd end. Elsevier Ltd; 2011.p. 960.
11. Fontanari GG, Jacon MC, Pastre IA, Fertonani FL, Neves VA. Batistuti JP. Protein isolate of guava seed (Psidium guajava): functional properties of characterization. Cienc. Technol. Aliment 2007; 27:1.
12. Gurpreet K, Chandi GK. Sogi DS. Functional properties of rice bran protein concentrate. J Food Engr 2006; 79: 592– 597.
13. Honig DH, Wolf WJ. Mineral and Phytate Content and Solubility of Soy- bean Protein Isolates. J Agric Food Chem 1987; 35: 583 – 588.
14. Horax R, Hettiarachchy NS, Chen P, Jalaluddin M. Functional properties of protein isolate from cowpea (Vigna unguiculata L. Walp). Food Sci. J 2004; 69: 119-121.
15. Hoffman JR, Flavo MJ. Macronutrient Utilization During Exercise: Implications for performance and supplementation. Sport Sci. and Med J 2004; 3: 118-130
16. Khalid II, Elhardallou SB. Elkhalifa EAComposition and functional properties of cowpea (Vigna ungiculata L.Walp) flour and protein isolates. American J Food Tech. 2012; 7(3):113-122.
17. Kimberlee KJ. Whey Protein Heat Stability, U.S. Dairy Export Council. 2012.p. 1-8
18. Kinsella JE. Functional properties of food proteins: a review. Crit Rev in Food Sci. and Nutri 1976; 7:219-280.
19. Kudre TG, Benjakul S, Kishimura H. Comparative study on chemical compositions and properties of protein isolates from mung bean, black bean and bambara groundnut. J Sci Food Agr 2013; 93(10):2429-36
20. Kulmyrzaev A, Bryant C, McClements DJ. Influence of sucrose on the thermal denaturation, gelation, and emulsion stabilization of whey proteins. J Agr Food Chem 2000; 48:1593-1597.
21. Kunt ID. Hydration of macromolecules III. Hydration of polypeptides. J Amer Chem Soc 1971; 3:514-515.
22. Lin MJY, Humbert ES, Sosulski FW. Certain functional properties of sunflower seed proteins. J Food Sci 1974; 39: 368- 370.
23. Lqari H, Vioque J, Pedroche J, Millan F. Lupinus angustifolius protein isolates: chemical composition, functional properties and protein characterization. Food chem 2002; 76(3): 349-356.
24. Lusas EW. Riaz MN. Soy protein products: processing and use. J Nutri 1995; 125(3):573S-580S.
25. Makri E, Papalamprou E, Doxastakis G. Study of functional properties of seed storage proteins from indigenous European legume crops (lupin, peas, broad bean) in admixture within polysaccharides. Food Hydrocolloids 2005; 19: 583-594.
26. McClements DJ. Analysis of protein. Available on the internet: http://people.umass.edu/~mcclemen/581Prot eins.htm Assessed 31/06/2013.
27. Morr CV, German B, Kinsella JE, Regenstein JM, Van Buren JP, Kilara A, Lewis BA, Mangino ME. A collaborative study to develop a standardized food protein solubility procedure. J Food Sci 1985; 50: 1715-1718.
28. Mou_ecoucou, J, Villaume C, Sanchez C, M_ejean L. Effects of gum arabic, low methoxy pectin and xylan on in vitro digestibility of peanut protein. Food Research Inter 2004; 37:777–783
29. Moure A, Sineiro J, Domínguez, Parajó JC. Functionality of oilseed protein products. A review. Food Research Inter 2006; 39:945- 963.
30. Mwasaru MA, Muhammad K, Bakar J, Yaakob, Man C. Effects of isolation technique and conditions on the extractability, physicochemical and functional properties of pigeonpea (Cajanus cajan) and cowpea (Vigna unguiculata) protein isolates. I. Physicochemical properties. Food Chem 1999; 67: 435-444.
31. Naczk M, Diosady LL, Rubin. Functional properties of canola meals produced by a two- phase solvent extraction system. J Food Sci 1985; 50:1685-1689.
32. Ogunwolu SO, Henshaw FO, Mock H-P, Santros A. Functional properties of protein concentrates and isolates produced from cashew (Anacardium occidentale L.) nut. Food Chem 2009; 115: 852-858.
33. Okaka JC, Potter NN. Functional properties of cowpea-wheat flour blend in bread making. J Food Sci 1977; 42: 828-833.
34. Olaofe O, Arogundad LA, Adeyeye EI, Falusi OM. Composition and food of the variegated grasshopper. Trop Sci 1998; 38: 233-237.
35. Omosaiye O, Cheryan M. Ultrafiltration of Soy bean Water Extracts: Processing Characteristics and Yields. J Food Sci 1979; (44): 1027-1031.
36. Onsaard E, Pomsamud P, Audtum P. Functional properties of sesame protein concentrate from sesame meal. J Food Agric Ind 2010; 3(4): 420-443.
37. Paredes-Lopez O, Ordorica-Falomir C, Olivares-vazquez MR. Chickpea protein isolates: physicochemical, functional and nutritional characterization. J Food Sci 2006; 56(30):726-729.
38. Qayyum M, Butt MS, Anjum FM, Hawaz H. Composition analysis of some selected legumes for protein isolates recovery. J Ani Plant Sci 2012; 22(4):1156-1162.
39. Rhim JW, Gennadios A, Weller CL, Cezeirat C, Hanna MA. Soy Protein Isolatedialdehyde starch film. Ind Crp and Prdt 1998; 8:195-203
40. Riaz MN. Soy Applications in Foods. London: CRC, 2006. p. 39-226.
41. Rich LM, Foegeding EA. Effects of sugars on whey protein isolates gelation. J Agric and Food Chem 2000; 48(10):5046-5052.
42. Saranya G. Extraction of protein from commercially available peanut de-oiled cake. Training report, CIPHET Ludhiana. India 2013.
43. Sathe SK, Salunkhe DK. Functional properties of the Great Northan Beans (PaPhaseolus vulgaris L.) Proteins: Emulsion, Foaming, Viscosity, and Gelation Properties. J Food Sci 1981; 46:71.
44. Sathe SK, Salunkhe DK. Functional properties of the Great Northan Beans (PaPhaseolus vulgaris L.) Proteins: Emulsion, Foaming, Viscosity, and Gelation Properties). J of Food Sci 1981; 46:626.
45. Sathe SK, Deshpande SS, Salunkhe DK. Functional properties of winged bean (Psophocarpus tetragonolobus L. DC) proteins. J Food Sci 1982; 47:503-509.
46. Seyam AA, Banank OJ, Breen MD. Protein isolates from navy and pinto beans: their uses in macaroni products. J Agric Food and Chem 1983; 31:499-502.
47. Shaviklo GR. Quality assessment of fish protein isolates using surimi standard methods. UNU fisheries training program Iceland 2006; p.11.
48. Sipos ES. Edible uses of soybean protein. American soybean Assoc. Sipos and associates, Inc. Fort wayne IN 2013; p. 6-17 available on internet at: http://www.asaimeurope.org/backup/pdf/edibleuses.pdf Assessed 2/06/2013.
49. Smith AK, Circle CJ. Soy bean Protein Precipitation from Water and Alkaline Dispersions by Acids and by Electrodialysis. Ind Engr chem 1939; 31:1284-1288.
50. Smith AK, Circle JJ. Soy beans: Chemistry and Technology, Vol. 1.-Proteins. Westport: AVI Publishing Company 1977.
51. Sosulski FW, Humbert ES, Bui K, Jones JO. Functional properties of rapeseed flour concentrates and isolates. J Food Sci 1976; 41:1348-1354.
52. Tobin G, Carpenter KJ. The nutritional value of the dry bean (Phaseolus vulgaris): a literature review, Nutri Abstr 1978; 48:920.
53. Vardhanabhuti B, Foegeding EA. Effects of dextran sulfate, NaCl, and initial protein concentration on thermal stability of β- lactoglobulin and α-lactalbumin at neutral pH, Food Hydrocolloids 2008; 22(5):752- 762.
54. Wardlaw GM, Insel PM. Perspectives in Nutrition. Mosby-Year Books, New York 1996.
55. Young VR, Scrimshaw NS. Soybean protein in human nutrition: An overview. J American Oil Chem Soc 1979; 56:110-120.
56. Zhang T, Jiang B, Wang Z. Gelation properties of chickpea protein isolates. Food Hydrocolloids, 2007; 21:280–286
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License