IJCRR - 6(4), February, 2014
Pages: 01-06
Date of Publication: 20-Feb-2014
Print Article
Download XML Download PDF
ECOTYPIC VARIATIONS IN INDIAN POPULATIONS OF ERYNGIUM FOETIDUM L.
Author: Thara Saraswathi K.J., Chandrika R., Jayalakshmi N. R., Mythri B.
Category: General Sciences
Abstract:Aim: The ecotypic variations with respect to total phenolics, flavonoid, tannin and Vitamin-C content in three different populations of E. foetidum collected from Andaman, Darjeeling (West Bengal) and Hassan (Karnataka) have been studied. Methodology: The total phenolic and total tannin content was estimated by FC method against standard pyrogallol and gallic acid respectively. Vitamin C was extracted with 4% oxalic acid solution and estimated using 2, 4- dinitrophenyl hydrazine reagent alongside standard ascorbic acid. The total flavonoid content was determined by Aluminum chloride colorimetric method with standard Quercitin (1mg/ml). The values were expressed as mg/g equivalents of respective standards. Results: A significant difference (p= 0.05%) in the concentration of secondary metabolites among all the studied populations was observed. The total phenolic content was highest in the Darjeeling population, flavonoid and tannin content was maximum in Andaman population and Vitamin C content was predominantly high in the Karnataka population. Conclusion: The variations in phytochemical constituents could be attributed to the possible interaction of the plant populations with their geographical location and their subsequent adaptations to the same.
Keywords: E. foetidum, ecotypes, total phenols, flavonoid, tannin, vitamin C.
Full Text:
INTRODUCTION
Eryngium is a large and taxonomically complex genus of the family Apiaceae comprising nearly 250 species distributed in many parts of Asia, America, Africa and Australia (Worz et al, Calvino et al). Eryngium foetidum L. commonly named as “wild coriander” (Seaforth et al) is the most popular herb under this genus extensively used for seasoning of a variety of continental dishes like salads, soups, sauces, noodles, and ceviche owing to its unique, pungent corianderlike aroma in most of the tropical regions. The herb has a wide range of usage in traditional medicine for treating cold, cough, fever, seizures, ear ache, hypertension, worms, infertility complications, snake bites, arthritis and malaria (Mitchelle et al, Shavandi M A et al). The scientific validation for the medicinal properties of the plant has been established for analgesic and anti-inflammatory properties (Garcia et al, Saenz et al, Mekhora C et al ), anti-helminthic (Forbes WM,2009) and anti-clastogenic activity (Promkum C et al). The essential oil distilled from this plant has also been valued in perfumery industry for its aromatic properties in international and domestic markets (Wong et al, Pino et al, Ignacimuthu S et al., 2004). In recent years, much attention has been given to the intake of phenolic acids, tannins, flavonoids and vitamin C in the human diet and analyzing the possible health benefits due to their antioxidant nature. These secondary metabolites are believed to be responsible for the wide spectrum of pharmacological activities seen in many plants. Nowadays, these are receiving considerable attention because of their reported protective role against cancer, heart disease, health promoting effects like anti-inflammatory, anti-microbial, antioxidant, prevention of osteoporosis, as well as protection of plants from pests and diseases. Vitamin C (a water soluble vitamin) acts as a coenzyme for large number of metabolic activities in the living organisms and also possess antioxidant property (Amina Abd El – Hamid ALY 2010, Daniel Modniki, Maciej Balcerek, 2009; Daniel Modniki, Maciej Balcerek 2009, Min B.R et al., 2008). In India, occurrence of E. foetidum is rare, endemic and is localized to small pockets of Tamil Nadu, Kerala, Karnataka, North East India and Andaman Island (Chandrika R et al., 2011, Kar and Borthakur 2007). The restricted distribution of the plant is attributed to the long dormant period before seed germination and the dependence of vital phenological events on first showers of monsoon (Chandrika et al, 2013). Hence, the present study was conducted to observe the effect of ecological variations and its impact on the production of secondary metabolites (total phenolics, flavonoids, tannin and vitamin C) in various Indian populations.
MATERIALS AND METHODS
Plant collection
The populations of E. foetidum were collected from Hassan, Karnataka ( 12°. 967N and 75°.783 E), Andaman Islands (16° to 14° N and 92° to 94° E) and West Bengal, Darjeeling (27° 01´ 59´´ N and 88° 16´ E) during August-September 2012. The climatic conditions of all the locations were recorded for purpose of interpretation of the present experimental findings (Table 1). The plants were identified and a voucher specimen of each population was deposited at the Regional Research Institute, Bangalore. Preparation of plant extract: About 20g of dried plant material was extracted with 200 ml of methanol with occasional agitation at RT for 24 h. The extract was filtered, centrifuged at 8000g for 10 min, supernatant collected, rotary evaporated at 250 C and stored in air tight bottles at 40 C till further use. Determination of total phenolic, total tannin and total flavonoid content: The total phenolic and total tannin content was estimated by F-C method as according to Singleton VL and Rossi JA (1965) N and Tamilselvi et al., (2012) with 1mg/ml of pyrogallol and gallic acid as a standard. The unknown extract values are expressed as mg/g of pyrogallol and gallic acid equivalents. The total flavonoid content was determined by Aluminum chloride colorimetric method (Chang C. et al). Quercitin (1mg/ml) was used as a standard and the values are expressed as mg/g of Quercitin equivalents. Extraction and estimation of total Vitamin C content: Vitamin C was extracted with 4% oxalic acid solution and estimated by using 2, 4- dinitrophenyl hydrazine reagent with A540 nm (S Sadashivam and A Manickam, 1997). Standard ascorbic acid at 5mg/ml was used to derive a calibration curve and the results are expressed as mg/g of ascorbic acid equivalents. Statistical analysis The variation in secondary metabolite content was statistically analyzed by applying ANOVA followed by Tukey HSD test and results are expressed as mean ± S.E (n=10) with p=0.05 as a limit of significance.
RESULTS
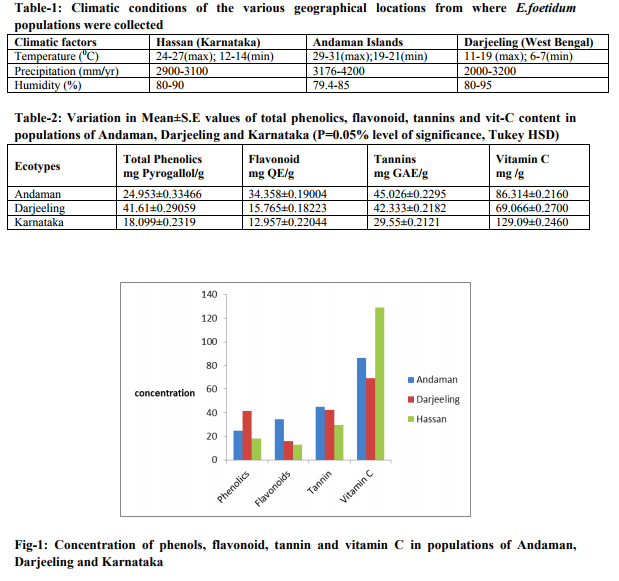
The mean variations in secondary metabolites from different populations are represented in Table 2 and graphically in Fig. 1 with Tukey HSD analysis at 0.05% level of significance. The total phenolic content in the dried plant extract was significantly high in Darjeeling population followed by Andaman and least in Karnataka population. Likewise the total flavonoid and tannin content was elevated in Andaman, followed by Darjeeling and lowered in Karnataka. Finally, the total vitamin C content was notably high in Karnataka population followed by Andaman and least in Darjeeling. In comparison with other secondary metabolites, Vitamin-C content was found to be quantitatively high in all the populations studied.
DISCUSSION
The variations among the ecotypes for Vitamin-C and secondary metabolite content are due to the biological diversity of E. foetidum populations in India. In the present study, all the ecotypes of E. foetidum showed very high concentration of Vitamin-C when compared to other secondary metabolites (total phenolics, flavonoid and tannins). Vitamin C is found to be the major constituent distributed in the plant leaves of E. foetidum (Campos et al. 2007; Promkum et al. 2012). Vitamin C was higher in the Karnataka population with significant difference (P < 0.05) compared with other two ecotypes. The Darjeeling population showed significantly lower Vitamin-C content than the other ecotypes. Variations in the content of Vitamin-C have been reported by Koziol (1992) while studying Chenopodium quinoa Willd ecotypes. According to Jimenez et al. (2009) the variations can be attributed to genetic or environmental growth conditions. Lee and Kader (2000) reported more Vitamin- C production when grown under cool temperatures rather than extreme hot and cold temperatures. The variation in Vitamin C content has also been observed with extended storage, higher temperature, low relative humidity, physical damage, chilling injury, large genotypic variations and climatic conditions (Lee and Kader, 2000; Dumas et al., 2003; Xu et al., 2008). According to Wall (2006), the levels of Vitamin-C in plants is directly proportional to the availability of light to the plant. In contrast, an excess of soil Nitrogen or Phosphorus tend to decrease ascorbic acid content. Hence it is possible to infer that the Karnataka population growing in a favorable moderate climatic condition contained high Vitamin-C content. Whereas, Darjeeling region with extreme cold conditions and limited supply of sun light yielded considerably low yield of Vitamin- C. The climate of Andaman Islands being hotter with high precipitation and humidity levels would have favored relatively higher production of Vitamin- C content in the populations of that region. The high Vitamin- C content reported among the populations in this investigation was similar to the data reported by Campos et al. (2007) in E. foetidum, Ruales and Nair (1993) and Dini et al. (2010) in Chenopodium quinoa Willd. All the ecotypes of E. foetidum presently studied showed that the variation in secondary metabolite production. Previous studies by Shui et al. showed that variation in secondary metabolite was related to the balance between carbohydrate source and sink. The greater the source-sink ratio, higher the production of secondary metabolites, which is directly linked to the plant growth in a particular ecotype. The presence of higher primary nutrients in the soil like nitrogen, phosphorous and potassium increases the secondary metabolite production (Tucker, 1999) and an even minor variation significantly varies the concentration of these metabolites (Jaleel et al. 2008).This implies that the increased nutrient content in the soil could have enhanced the production of total non structural carbohydrates which in turn would have contributed towards higher production of secondary metabolites (Ibrahim MH, 2010) in E. foetidum populations from Darjeeling and Andaman populations.
CONCLUSION
In the present study the variation in the polyphenol and vitamin C contents in E. foetidum populations from India were determined. This finding helps us to know the importance of secondary compounds showing significant variation among different ecotypes. The significance involved in relating external and edaphic factors to trace out the performance of secondary metabolite production in the ecotypes needs further emphasis.
ACKNOWLEDGEMENT
Dr. K.J. Thara Saraswathi is thankful for the financial support under UGC grants. The authors wish to thank the Post Graduate Department of Studies in Microbiology and Biotechnology, Jnana Bharathi Campus, Bangalore University, Bangalore for providing the necessary facilities.
References:
1. Amina Abd Ei, Hamid ALY (2010). Biosynthesis of phenolic compound and water soluble vitamins in cilantro (Eryngium foetidum L.) plantlets as affected by low doses of gamma irradiation. Analele Universitaii din Oradea – Fascicula Biologie., 17(3): 356-361.
2. Calvino, Carolina, I.; Martinez, Susana, G.; Downie, Stephen, R., 2008: The evolutionary history of Eryngium Apiaceae, Saniculoideae Rapid radiations, long distance dispersals, and hybridizations. Molecular Phylogenetics and Evolution. 46(3): 1129-1150.
3. Chandrika R, Thara Saraswathi KJ, Shivakameshwari MN (2013). Phenological events of Eryngium foetidum L. from Karnataka, India. International Journal of Plant Reproductive Biology., 5(1) : 89-91.
4. Chandrika R, Vyshali P, Saraswathi KJT, Kaliwal BB (2011). Rapid multiplication of mature flowering plant of Eryngium foetidum L. by in vitro technique. International Journal of Biotechnology Applications. 3(4):114-117.
5. Chang, C.C., Yang, M.H., Wen, H.M, Chern, J.C., (2002): Estimation of total flavonoid content in propolis by two complementary colorimetric methods. Journal of Food and Drug Analysis, 10: 178-182.
6. Daniel Modnicki, Maciej Balcerek (2009). Estimation of total polyphenols contents in Ocimum basilicum L., Orgianum vulgare L. and Thymus vulgaris L. commercial samples. Herba Polinica. 55(1): 35-42.
7. Dini, I., G.C. Tenore, and A. Dini. 2005. Nutritional and antinutritional composition of Kancolla seeds: an interesting and underexploited Andine food plant. Food Chemistry 92:125-132.
8. Dini, I., G.C. Tenore, and A. Dini. 2010. Antioxidant compound contents and antioxidant activity before and after cooking in sweet and bitter Chenopodium quinoa seeds. LWT- Food Science and Technology 43:447-451.
9. Dumas, Y., M. Dadomo, G. Di Lucca, and P. Grolier. 2003. Review: Effects of environmental factors and agricultural techniques on antioxidant content of tomatoes. Journal of the Science of Food and Agriculture 83:369-382.
10. Forbes WM and Steglich C 2009 Methods of treating infectious diseases, Patent Application Number-20090047342.
11. Ibrahim MH, Jaafar HZ, Rahmat A, Rahman ZA.Molecules. 2010 .The relationship between phenolics and flavonoids production with total non structural carbohydrate and photosynthetic rate in Labisia pumila Benth. under high CO2 and nitrogen fertilization. 29; 16(1):162-74.
12. Ignacimuthu S, Arockiasamy S, Antonysmay M, Ravichandran P (1999). Plant regeneration through somatic embryogenesis from mature leaf explants of Eryngium foetidum, a condiment. Journal of Plant Cell, Tissue and Organ Culture., 56:131-137.
13. Jaleel C. A., Sankar B., Sridhara R. and Panneerselvam N. R. 2008. Soil alinity alters growth, chlorophyll content, and secondary metabolite accumulation in Catharanthus roseus. Turk J Biol; 32 : 79-83
14. Jiménez, M.E., A.M. Rossi, and N.C. Sammán. 2009. Phenotypic, agronomic and nutritional characteristics of seven varieties of Andean potatoes. Journal of Food Composition and Analysis 22:613-616.
15. Kar A and Borthakur SK 2007 Wild vegetables sold in local markets of Karbi Anglong, Assam, Indian J. Traditional Knowledge 6(1) 169-172.
16. Koziol, M.J. 1992. Chemical composition and nutritional evaluation of quinoa (Chenopodium quinoa Willd.) Journal of Food Composition and Analysis 5:35-68.
17. Lee, S.K., and A.A. Kader. 2000. Preharvest and postharvest factors influencing vitamin C content of horticultural crops. Postharvest Biology and Technology 20:207-220.
18. Min BR, Pinchak WE, Merkel R, Walker S, Tomita G, Anderson RC (2008). Comparative antimicrobial activity of tannin extract from perennial plants on mastitis pathogens. Scientific Research and Essay. 3(2): 066-073.
19. Mitchell SA, Ahmad MH (2006). A review of medicinal plant research at the University of the West Indies, Jamica, 1948- 2001. West Indian Med J, 55, 243-69.
20. Mohammad Amin Shavandi, Zahra Haddadian, Mohd Halim Shah Ismail (2012). Eryngium foetidum L. Coriandrum sativum and Persicaria ordorata L : A Review. Journal of Asian Scientific Research. 2(8): 410-426.
21. Pino J A, Rosado A and Fuentes 1997 Composition of leaf essential oil of Eryngium foetidum L. from Cuba J. Essential oil Res. 9 467-468.
22. Promkum C, Butryee C, Tuntipopipat S, Kupradinun P. 2012. Anticlastogenic effect of Eryngium foetidum L. assessed by erythrocyte micronucleus assay. Asian Pac J Cancer Prev.; 13(7):3343-7.
23. Ruales, J., and B.M. Nair. 1993. Contents of fat, vitamins and minerals in quinoa (Chenopodium quinoa Willd.) seeds. Food Chemistry 48:131-137.
24. Sadashivam S,Manickam A (1997). Vitamins – Ascorbic acid, Colorimetric analysis. Biochemical methods. New age International Publishers., pp.184-186.
25. Saenz MT, Fernandez MA and Garcia MD 1997 Anti-inflammatory and analgesic properties from leaves of Eryngium foetidum L. (Apiaceae), Phytotherapy Res. 11(5) 380-383.
26. Seaforth C, Tikasingh T (2005). Final ReportA study for the development of a handbook of selected Caribbean herbs for industry. Trinidad: CHBA/JICA, p. 44-7.
27. Shui, Y.C.; Feng, X.; Yan, W. 2009. Advances in the study of flavonoids in Gingko biloba leaves. J. Med. Plant Res., 3, 1248–1252.
28. Singleton VL, Rossi JA. Colorimetry of total phenolics with phosphormolybdic – phosphotungstic acid regents. Am J Enol Viticul., 1965; 16 : 144-158.
29. Tamil Selvi N, Krishnamoorthy P, Dhamotharan R, Arumugam P, Sagadevan E . Analysis of total phenols, total tannins and screening of phytocomponents in Indigofera aspalathoides (Shivanar vembu) Vahl EXDC. J Chem Pharma Research. 2012; 4(6): 3259- 3262.
30. Wall, MM. Ascorbic acid and mineral composition of longan (Dimocarpus longan), lychee (Litchi chinensis) and rambutan (Nephelium lappaceum) ecotypes grown in Hawaii. J Food Comp Ana 2006.19:655-663.
31. Wong KC, Feng MC, Sam TW and Tan GL. Composition of the leaf oil of Eryngium foetidum L. from Cuba. J. Essential oil Res. 1994; 6 :369-374.
32. Wörz, A. On the distribution and relationships of the South-West Asian species of Eryngium L. (Apiaceae-Saniculoideae). Turk. J.Bot., 2004, 28, 85-92.
33. Xu, S., J. Hu, H. Tan, and S. Zhang.. Effects of genotype and environment on vitamin C content and its heterosis in towel gourd fruits. J Sci of Food and Agr 2008 ; 88:293-293.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License