IJCRR - 6(9), May, 2014
Pages: 138-144
Print Article
Download XML Download PDF
ESTIMATION OF GLYCATED ALBUMIN LEVELS IN VARIOUS THYROID DISORDERS
Author: Suresh Babu Kondaveeti, Bhanu Prakash G., I. Anand Shaker
Category: Healthcare
Abstract:Background: Glycation is the linking of sugar molecule to various compounds such as glucose, lipid or protein. As albumin is one of major protein in our circulation, to which sugar has bonded to form Glycated albumin. Albumin is present not only in blood but also in major organs and body fluids and it serves to maintain cell shape and distribution of hormones. Objective: The present study was designed to compare the levels of GA in Various thyroid disorders group with Normal healthy control group. Materials and Methods:The present cross sectional study includes 200 cases, (n=50 in each group) newly diagnosed hyperthyroidism, hypothyroidism, subclinical hypothyroidism and normal controls between 30-55 years of both genders.serum samples obtained for the estimation of T3,T4,TSH and glycated albumin. Statistical analysis done by using SPSS, version16.0. One-way ANOVA was performed. All P-values which were ?0.05 were considered as stastically significant. Results: Glycated albumin values were found to be significantly correlated among the different groups (p=0.001). GA levels significantly increased in hypothyroidism, subclinical hypothyroidism and lowered in hyperthyroidism group compared to normal group. Significant correlation was found between T3, T4, TSH and GA levels. Conclusion: The present study showed increased levels of GA among experimental groups when compared with the normal control group. Hence It was concluded that estimation of serum GA will be useful as a noval parameterin thyroid disease cases.
Keywords: Glycated albumin, Hypothyroidism, subclinical hypothyroidism, Hyperthyroidism
Full Text:
INTRODUCTION\
Thyroid gland is a body organ located in front of the neck. It has two lobes, one on each side of trachea. The thyroid gland takes iodine from blood and synthesizes two active hormones called triiodothyronine (T3) and thyroxine (T4) and these hormonal production regulated by TSH[1]. Thyroid hormones control the body’s cell metabolism. When thyroid hormones are released into the bloodstream, cells increase the rate at which they convert oxygen and nutrients into energy and heat for the body’s use [2]. Abnormalities of thyroid gland function are more common among individuals with developmental disabilities than in the general population, But overt hypothyroidism and subclinical hypothyroidisms are become relatively common now a days in general populationas well [3,4]. When thyroid gland is overactive and produces too much thyroid hormone an individual develops hyperthyroidism and Subclinical Hypothyroidism is defined as a condition in which serum TSH concentration above the statistically defined upper limit of the reference range, when the serum free T4 concentration is within its reference range [5,6]. Primary hypothyroidism and thyrotoxicosis are very common in adult female genders with prevalence rate 3.5per thousand and 0.8 per thousand respectively. There is increased T3 and T4 levels with decreased or normal TSH levels in case of thyrotoxicosis especially in developed countries[7]. Glycated albumin formed by non enzymaticglycation in which reducing sugars are covalently attached to amino acid residues especially serine, lysine residues of protein[8]. The reducing carbonyl group of glucose can react with the amine groups of human serum albumin (HSA) to form glycated albumin (GA) (Schiff-base) which results in a 162 Da molecular weight increase for each glucose-induced glycation on HSA. This is followed by the Schiff-base reorganized itself to the more stable aminomethyl ketone by the Amadori rearrangement [9] In hypothyroid patients the Glycated protein levels have been increased and in case of hypothyroid cases its levels were found to be decreased [10]. Compared to normal subjects the GA levels higher in primary hypothyroidism cases and lower in Grave’s disease. As thyroid hormone promotes albumin metabolism the mean concentration of albumin and total protein in serum were lower in hyperthyroid patients than the hypothyroid patients[11]. It has been proved from the previous studies that GA can be used to monitor short term glycemic controlthan glycated hemoglobin[12]. We therefore hypothesized that GA levels do not reflect accurately glycemia in hypothyroidism.
MATERIALS AND METHODS
The present cross-sectional study was conducted from December 2010 to January 2012 in MAPIMS&R hospitalMelmaruvathur, Tamil Nadu respectively. A total of 150 cases were selected (n=25 in each group) newly diagnosed hyperthyroidism, subclinical hypo thyroidism, hypothyroidism and normal control between the ages of 30-58 years of both sexes (M:F ratio 65:85) were selected for this cross sectional study from the outpatient department of endocrinology in MAPIMS&R and were compared with 50 (M: F 20:30) control subjects. After the full explanation of the study, written informed consent was obtained from each study subject. The study was approved by the Ethics committee of the institution prior (EC NoMAPIMS/RC/NOV/2010/24) the informed consent was obtained from all the subjects.Demographic and anthropometric details like age, weight, height, duration of disease, duration of treatment were recorded for all the study subjects. Family history of Thyroid disease and hypertension, smoking and alcohol consumption habits were obtained from the medical records of the study subjects.Patient on treatment for any disorder, lipid lowering drugs, diabetes, malignancy and pregnant women were excluded.Blood samples were collected for the biochemical estimations such as T3, T4, and TSH using Hitachi auto analyzer by Electro Chemiluminescence immune assay (ECLIA) method.Plasma glycated albumin (GA) levels were measured by an enzymatic method using albumin specific protease, ketoamine oxidase and albumin assay reagent on the Hitachi autoanalyser 912 (Lucica GA-L, Asahi Kasei Pharma Corp, Tokyo, Japan) [13,14] GA was hydrolyzed to amino acids by albumin specific protease and then oxidized by ketoamine oxidase to produce hydrogen peroxide, which was measured quantitatively. The GA value was calculated as the percentage of GA relative to total albumin, which was measured with bromocresol purple method. The measured values of GA was not influenced by the substances such as bilirubin F up to 14.6 mg/dl, bilirubin C up to 15.2 mg/dl, glucose up to 1000 mg/dl, ascorbic acid up to 100 mg/dl. The statistical analysis was done by using SPSS version 16.0. One Way ANOVA method was applied to observe association of microalbuminuriawith GA and duration ofdiabetes. P value < 0.0001 was considered as statistically significant.
RESULTS
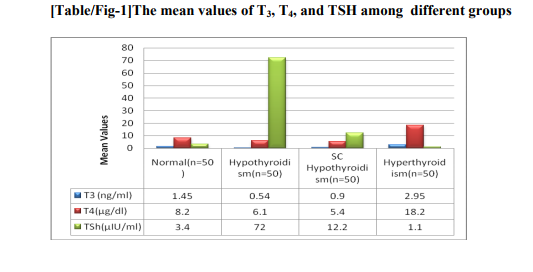
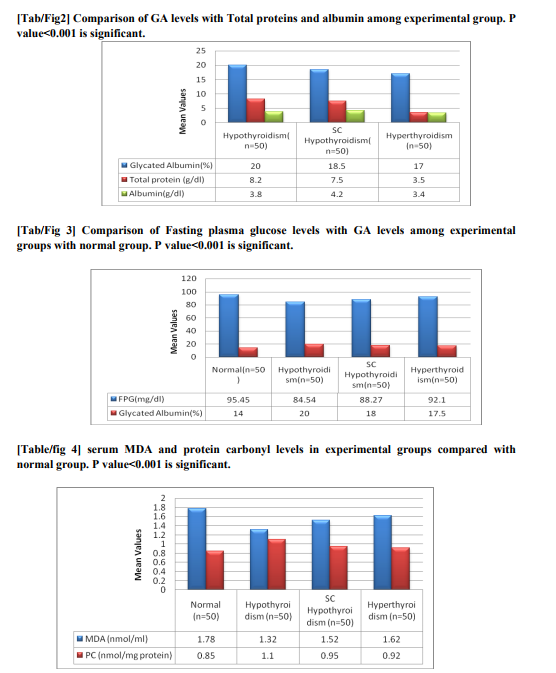
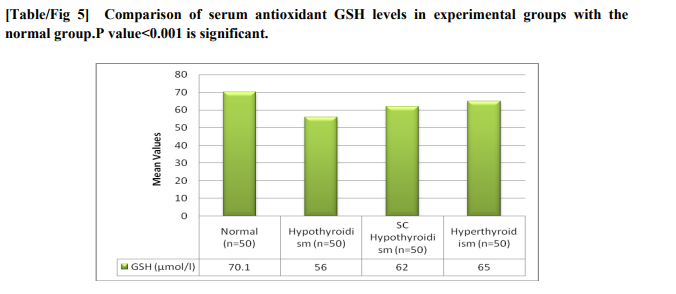
The mean values of thyroid hormones among experimental group along with the normal group has shown in fig 1.GA values were much more elevated in hypothyroidism group compared to subclinical hypothyroidism group shown in figure 2 and the values were lower than the normal level in hyperthyroidism group and higher than normal in hypothyroidism group which is statistically significant.Figure3 shows the GA values and FPG levels of different groups which were below normal in case of hyperthyroidism and above normal in subclinical hypothyroidism which is stastically significant. GA values found to be significant among the groups (p=0.001). In hypothyroidism specificity and the sensitivity was found to be 100% and 98.4%and in hyperthyroidism it is 94.8% and 72%. Where as in Subclinical hypothyroidism it is around 95.5% and 78%. Compared to hypothyroidism group the albumin values in hyperthyroidism group found to be decreased which is statistically correlated negatively with hypothyroidism but no statistical significant correlation with total proteins. While comparing with the normal GA levels as 12-16 mg % the sensitivity, specificity, positive predictive value(PPV) of GA for hyperthyroidism, hypothyroidism, and subclinical hypothyroidism.fig 4 and fig 5 showing the serum MDA and protein carbonylation levels in the experimental groups which are elevated in hypothyroidism group as well as in subclinical hypothyroid group. At the same time serum antioxidants such as GSH levels decreased in respect to increase of oxidants in all the groups except normal group
DISCUSSION
Thyroid hormone exert profound effects in the regulation of glucose homeostasis, including modification of circulating insulin levels and other counter regulating hormones[15].thyroid disorders has got a major impact on glucose control. When thyroid dysfunction the glucose homeostatic balance is broken and it is mainly associatedwith increased hepatic gluconeogenesis whichis a characteristic of excess of thyroid hormones i.e Hyperthyroidism [16]. In hypothyroidism glucose homeostasis also get affected although it’s clinical impactless it’s because of less disposal of glucose [17]. At the same time insulin resistance has been reported in subclinical hypothyroidism with altered glucose levels [18]. As deficiency or over production of thyroid hormones plays major role in glucose homeostasis, so we tried to assess the glycated products such as glycated albumin levels in various thyroid disorders. In our study GA levels significantly correlated (p=0.001) when inter comparison was done among different groups and we found GA levels are above normal in case of hypothyroidism, subclinical hypothyroidism and lower the normal values in case of hyperthyroidism. The albumin values were found to be decreased in hyperthyroidism group compared to hypothyroidism which was statistically significant. The increased GA levels in hypothyroid patients along with non clinical hyperglycemia because of deterioration of protein metabolism which further decreases turnover of proteins and increase of half life of proteins. And the increase in glycation of proteins via auto oxidative glycation incase of increased oxidative stress[19]. There is marked increase in protein carbonylation, serum Malondialdehyde and decreased glutathione levels in hypothyroid cases explaining increased peroxidation of lipids might be a contributing factor for increased protein glycation[20] . Low grade inflammation and free radical formation causes increase in higher immunoglobulin production explains glycation of these immunoglobulins. Disturbance in glucose homeostasis with decreased glucose absorption and utilization leads to insulin resistance this further causes glycation of proteins. In case ofsubclinical hypothyroidism the GA levels were not that higher compared with hypothyroid group. The GA levels were lower in hyperthyroid in contrast to hypothyroid and subclinical hypothyroid groups but with higher plasma glucose levels[21,22,23]. Previous studies showed that there is increase in glycated HbA1c which corresponds to raise in glucose concentration but with the low levels of Glycated albumin which is very contradictive[24].
CONCLUSION
There are only studies reported about HbA1C levels in thyroid disorders so we tried to estimate glycated products such as GA other than HbA1C, and present study suggests an increase GA levels in all the thyroid disorders with which we conclude, that we can consider GA as a diagnostic criterion in thyroid disorders.
ACKNOWLEDGEMENT
The authors are thankful to the Management, MAPIMS&R and M.Sachithanandam, Vishnu Diagnostics Center, No. 1 Ennakara street, Kancheepuram 631501 Tamilnadu for providing the necessary facilities and permitting to carry out this research work. The authors are very much thankful to all the Physicians who had referred the cases to this Diagnostic Center. Authors also acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
Source of support: - Nil
Conflict of Interest: - None declared
References:
REFERENCES
1. Pierce JC: Pituitary Thyrotropin: Chemistry in The Thyroid. 1998.fourthedition.
2. Muller MJ, Acheson KJ, Jequier E, Burger AG. Effect of thyroid hormones on oxidative and nonoxidative glucose metabolism in humans. Am J Physiol.1988;255:E146-52.
3. Canaris G,J, Manowitz NR, Mayor G, and Ridgway C: The Colorado Thyroid Disease Prevalance study. Arch Intern Med. 2000.160;526-534.
4. Boelaert K, Franklyn JA. Thyroid hormone in health and disease.J Endocrinol2005;187:1-15.
5. Wiersinga WM. Subclinical hypothyroidism and hyperthyroidism.I. Prevalence and clinical relevance. Neth J Med 1995;46:197-204.
6. Wilson GR: Subclinical Thyroid Disease: scientific review and guidelines for diagnosis and management. JAMA, 2008. 291:228-38.
7. Carl A. Burtis, Edward R. Ash wood, David R. Bruns. Tietz text book of Clinical Chemistry and Molecular Diagnostics. 4th ed. Philadelphia: W.B. Saunders Company publishers: 2008.
8. Rondeau, P.; Bourdon, E. The glycation of albumin: Structural and functional impacts. Biochimie. 2011, 93, 645–658.
9. Day, J.F.; Thorpe, S.R.; Baynes, J.W. Nonenzymaticallyglucosylated albumin. In vitropreparation and isolation from normal human serum. J. Biol. Chem. 1979, 254, 595– 597.
10. CirilloR,Balzano S, Cossu E, BartelenaL,Solinas MP, Falcone M, Balestrieri A, Martino E. The effect of altered thyroid function on serum fructosamine concentrations. Clin Biochem.1988 Jun; 21(3):179-81.
11. Larsen P, Davies T. Hypothyroidism andthyroiditis. In Williams Textbook of Endocrionology. 10th ed. Larsen PR, Kronenberg HM, Melmed S, Polonsky KS, Eds. Maryland Heights, Missouri, Saunders Elsevier, 2002, p. 423–455
12. Masaaki Inaba, SenjiOkunoet al. Glycated Albumin Is a Better Glycemic Indicator thanGlycated Hemoglobin Values in Hemodialysis Patients withDiabetes: Effect of Anemia and Erythropoietin Injection J Am SocNephrol 18: 896 –903, 2007. doi: 10.1681/ASN.2006070772
13. Kouzuma T, Usami T, Yamakoshi M, et al. An enzymatic method for the measurement of glycated albumin in biological samples. ClinChimActa 2004; 324: 61-71.
14. Kouzuma T. Study of glycated amino acid elimination for an improved enzymatic glycated albumin measurement method. ClinChimActa 2004; 346: 135-143
15. Raboudi N, Arem R, Jones RH et al. Fasting and postabsorptive hepaticglucose and insulin metabolism in hyperthyroidism. Am J Physiol1989;256:E159-66.
16. Weinstein SP, O’Boyle E, Fisher M, Haber RS. Regulation of GLUT2 glucose transporter expression in liver by thyroid hormone: evidencefor hormonal regulation of the hepatic glucose transport system.Endocrinology 1994:135:649-54.
17. R ochon C, Tauveron I, Dejax C et al. Response of glucose disposal to hyperinsulinemia in human hypothyroidism and hyperthyroidism. ClinSci2003;104:7-15.
18. Maratou E, Hadjidakis DJ, KolliasAet al. Studies of insulin resistance in patients with clinical and subclinical hypothyroidism. Eur J Endocrinol2009;160:785-90.
19. Meller J, Zappel H, Conrad M, Roth C, Emrich D, Becker W. Diagnostic value Q of 123 iodine scintigraphy and perchlorate discharge test in the diagnosis of congenital hypothyroidism. ExpClinEndocrinol Diabetes. 1997;105:24-27.
20. Nanda N, Bobby Z, Hamide A. Oxidative stress and protein glycation in primary hypothyroidism.Male/female difference.ClinExp Med 2008;8:101-108.
21. Weijers RN, Slaats EH, Kruijswijk H, Fructos amine values in hyperthyroidism, hypothyroidism and gammopathy WeinKlinWochenschr 1990; 18 0:Suppl21-24.
22. Ford HC, Lim WC, Crooke MJ, HemoglobinA 1c and serum fructosamine levels in hyperthyroidism ClinChemActa 1987; 166:31 7-21.
23. Mohan Kumar, Bobby Z, Selvaraj N, Kumard as A, Chandra KonerB, Sen SK, Possible link between glycated hemoglobin and lipid peroxidation in hyperthyroidism ClinChemActa. 2004; 342:1 87-92.
24. Kim HB, Han KH, Lee BW, Kim H, Lee MH, Chung ES, HbA1c and serum fructosamine levels in hyperthyroidism J KorSocEndocrinol 1992; 7:46-51
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License