IJCRR - 6(9), May, 2014
Pages: 18-29
Date of Publication: 12-May-2014
Print Article
Download XML Download PDF
VALIDATION OF NEWLY FORMULATED LAPORTEA ARISHTA BY USING DIFFERENT ANALYTICAL METHODS
Author: Deepa Deepa Deepa Deepa Deepa Deepa P., Seena
Category: General Sciences
Abstract:PurposPurposPurposPurposPurposPurpose: Traditional medicines have nurtured the knowledge of natural remedies against diseases since ages. Growing awareness about harmful side effects of modern medicine has led to interest in Ayurveda. Method: Laporteainterrupta (L) Chew of family Urticaceae having common name Hawaii woodnettle is an herb having many traditional uses. physicochemical analysis of crude drug, Laporteainterrupta-leaves were performed,Laporteainterrupta leaves extracted in soxhlet apparatus using ethyl acetate, petroleum ether, chloroform, methanol and water for identification of constituents by qualitative phytochemical analysis (tests for protein, carbohydrates, phenols and tannins, flavanoids, saponins, glycosides, steroids, terpenoids and alkaloids) and quantitative phytochemical analysis (total phenol content and total flavanoid content). The collected leaves of Laporteainterrupta was used to prepare arishta. After preparing the arishta the organoleptic characteristics (color, odor, taste and appearance), the physicochemical analysis ( pH, acid value, alcohol content, total solid content, viscosity and refractive index), quantitative analysis (alcohol content by spectrophotometry by dichromate method, total reducing sugar, total phenol content and total flavanoid content), anti-oxidant activity determination ( Ferric Thiocyanate method, Thiobarbituric acid method and Total Antioxidant Activity by FRAP method) were performed. LCMS was performed for determining various constituents (Qualitative analysis) and standardization of formulation was done by using UV and HPLC. The above formulated LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical LAPORTEA ARISHTA was validated by using different analytical equipmentequipmentequipmentequipmentequipmentequipmentequipmentequipmentequipment's as per ICH guidelines.guidelines.guidelines.guidelines.guidelines.guidelines.guidelines.guidelines.guidelines.guidelines.guidelines. Results: Results: All results are coming between standard ranges. results are coming between standard ranges. results are coming between standard ranges. results are coming between standard ranges. results are coming between standard ranges. results are coming between standard ranges. results are coming between standard ranges. results are coming between standard ranges. Conclusion: Conclusion: Conclusion: Laporteainterrupta (L) Chew of family Urticaceae found to have number of active constituents from LCMS reports, which may can take as a proof for its traditional uses. Laporteainterruptaleaves have been successfully formulated and determined its anti-oxidant activity and validated using HPLC.
Keywords: Arishta, Physicochemical analysis, organoleptic characteristics
Full Text:
INTRODUCTION
Herbal medicine (or "Herbalism") is the study and use of medicinal properties of plants. They have the ability to synthesize a wide variety of chemical compounds that are used to perform important biological functions, and to defend against attack from predators such as insects, fungi and herbivorous mammals. Ayurveda - The Natural Way of Treating Disease Ayurveda is a form of alternative medicine that uses different parts of herbs, plants and parts of animals to cure diseases and ailments. Arishta are medicinal preparations made by soaking the drugs in coarse powder form or in the form of decoction (Kashaya), in a solution of sugar or jaggery, as the case may be, for a specified period of time, during which it undergoes a process of fermentation generating alcohol, thus facilitating the extraction of the active principles contained in the drugs. Laportea interrupta (L) Chew of family Urticaceae having common name Hawaii woodnettle is an herb having many traditional uses like Allergies, Alopecia, Amenorrhea, Arthritis, Asthma, Bedwetting/incontinence, Female Hormones, Fibromyalgia, Libido, Longevity/tonics, Menorrhagia, Nutrition, Osteoporosis, PMS, Prostate, Rheumatoid arthritis. Flavanoids have been reported to exert wide range of biological activities like anti-inflammatory, antibacterial, antiviral, anti-allergic, cytotoxic antitumor, treatment of neurodegenerative diseases, vasodilatory action. Standardization is the development and implementation of concepts, doctrines, procedures and designs to achieve and maintain the required levels of compatibility, interchangeability or commonality in the operational, procedural, material, technical and administrative fields to attain interoperability. Validation is documenting that a process meets its pre-determined specifications and quality attributes the objective of validation of an analytical procedure is to demonstrate that it is suitable for its intended purpose. Typical validation characteristics which should be considered are accuracy, precision, repeatability, intermediate precision, specificity, detection limit, quantisation limit, linearity, range.
MATERIALS AND METHOD
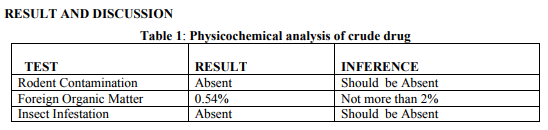
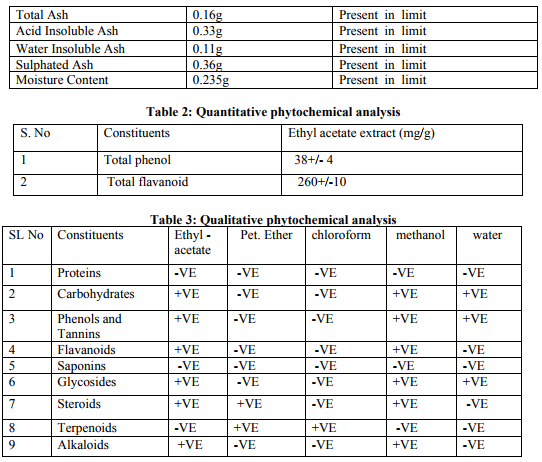
Collection of plant material: The leaves of Laportea interrupta were collected and washed well then dried under shade and coarsely powdered. Physicochemical analysis of crude drug: Total ash value, Acid insoluble ash, Water insoluble ash, Sulphated ash, Moisture content. Extraction of plant material: The plant material was dried and coarsely powdered and then extracted in Soxhlet Apparatus using 250ml of ethyl acetate (70-80°c), 250ml of Petroleum Ether (60-80°c), 250ml of Chloroform (50.5-61.5°c ), 250ml of methanol (60-70°c) and 250ml of Distilled water (0.25%) by continuous hot percolation according to their polarity. (Harborne, J.B., 2007). Qualitative phytochemical analysis: The extracts were subjected to qualitative tests for identification of phytochemical constituents such as proteins ( Million’s test and Ninhydrin test), carbohydrates ( Fehling’s test, Benedict’s test, Molisch’s test and Iodine test), phenols and tannins (Ferric Chloride test and Lead Acetate test), flavanoids ( Shinoda test and Alkaline Reagent test), saponins, glycosides (Liebermann’s test, Salkowski’s test and Keller-Kilani), steroids, terpenoids, alkaloids ( Mayer’s test and Wagner’s test) present in it. (RNS Yadev et al. 2011) . Quantitative phytochemical analysis Total Phenol Content: The amount of phenol in the ethyl acetate extract was determined by Folinciocalteu reagent method with some modification and then absorbance were measured at 765nm. Total Flavanoid Content: The amount of phenol in the ethyl acetate extract was determined by Aluminium chloride colorimetric method with some modification and then absorbance were measured at 420nm.
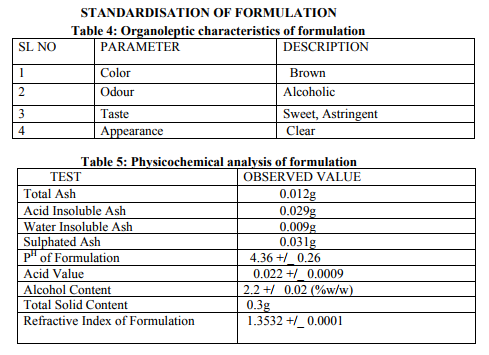
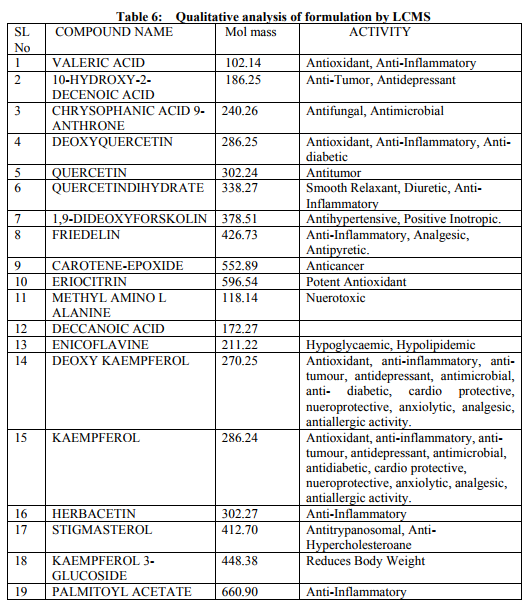
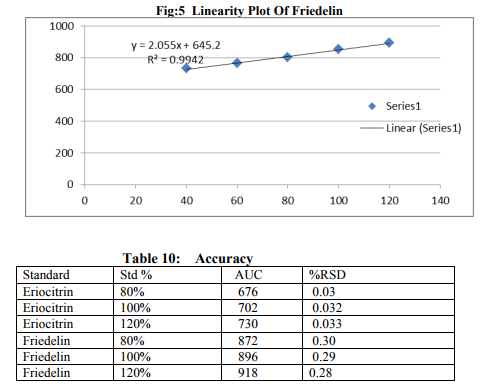
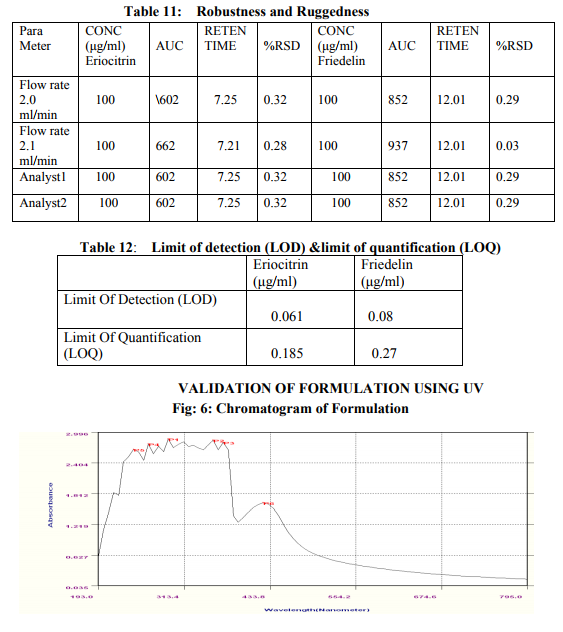
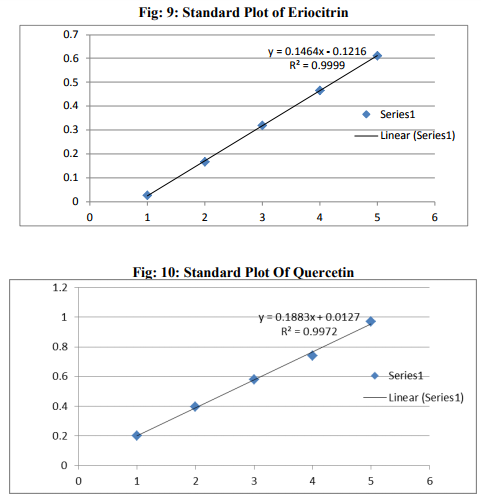
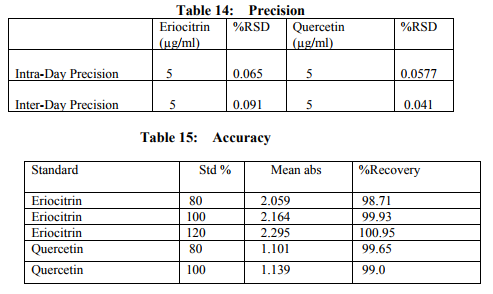
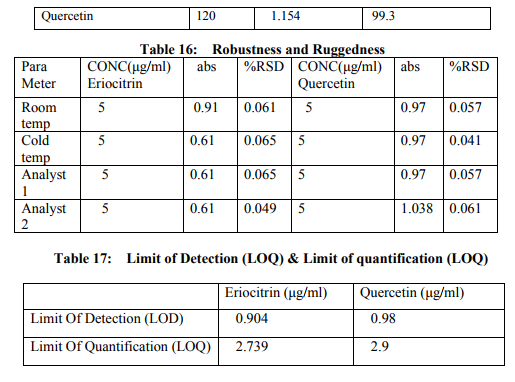
Preparation of formulation: An earthen pot sufficiently large and strong is chosen. The proportion of the different ingredients are 32 seers of distilled water, boiled and added 1.25 seers dried plant leaf powder jaggery 12.5 seers, honey 6.25 seers and sugar 6.25 seers, mixed well to form a uniform solution and pour into it . Pot is buried in a pit made in the soil for 30 days. Physicocemical analysis of formulation: Determination of PH of formulation, Determination of acid value, Determination of alcohol content, Total solid content, Determination of viscosity of formulation, Determination of refractive index. Quantitative analysis of formulation Alcohol content by spectrophotometry (Dichromate Method): Alcohol was estimated by caputi et al (1968) and measured the absorbance at 620nm. Total reducing sugar: Total phenol content: The amount of phenol in the formulation was determined by Folin-ciocalteu reagent method with some modification and then absorbance were measured at 765nm and the results were determined from the standard curve and expressed in gallic acid equivalents (mg/g of extracted compound).. Total flavanoid content: The amount of phenol in the formulation was determined by Aluminium chloride colorimetric method with some modification and then absorbance were measured at 420nm and the results were determined from the standard curve and expressed in Quercetin equivalents (mg/g of extracted compound) Anti-oxidant activity determination of formulation: Ferric Thiocyanate (FTC) method, Thiobarbituric Acid (TBA) method, Total Antioxidant activity by FRAP method. Qualitative analysis of formulation by LCMS: Specifications of LCMS are LC Column: REVERSE PHASE C-18, Pump: SPD 10 AVP, Mobile Phase: 2% OPA IN WATER: METHANOL (50:50), Ionization Mode: ELECTRONIC SPRAY IONIZATION, Mode: BOTH POSITIVE, Injection Volume: 10 µL, Flow Rate: 2 ML/MIN, Column Temperature: 250°C, Column: PHENOMENEX RP 18, Column Dimension: 25CM 2.5 MM,LC Detection: 254NM,m/z range: 50- 1000,Soft Ware: CLASS V P INTEGRATED AND Library: METWIN 2.0. Standardisation of formulation using UV: Specifications of UV are Model: ELICO SL 164, Instrument: UV-Visible Double Beam Spectrophotometer, Wavelength Range: 190-999.9 nm, Accuracy: +/- 0.5nm, Repeatability: +/- 0.2nm, Resolution: 0.1nm, Bandwidth: 0.5-6.0 nm (Variable at an Interval of 0.1nm), Photometric range: -2.5 TO +2.5 Abs, Accuracy: +/- 0.005 Abs at 0.1Abs from 200-850nm, Repeatability: +/- 0.002 Abs at 1.0 Abs from 200-850nm, Stray light: less than 0.05% at 220-340nm,Base line correction: Automatic Base Line Correction, Scan speed: Slow, Medium and Fast, Data interval: Depend Upon Wavelength, Sample holder: 5 Position, Automatic positioning far 10mm and Sample Cuvette and Fixed position for Reference Cuvette, Source: Tungsten- Halogen lamp (310-999.9nm) and Deuterium lamb with Quartz window (190-340nm) and Detectors: Photomultiplier Tube (PMT).Validation experiments were performed to demonstrate linearity, precision, accuracy, robustness, ruggedness, LOD and LOQ as per ICH guidelines. Standardisation of formulation using HPLC: Specification of HPLC is Instrument: Schimadzu, LC Column: Reverse Phase C-18 Phenomenex C18, Pump: SPD 10 AVP, Mobilephase: Acetonitrile: Methanol (40:60), Injection volume: 10 µl, Flow Rate: 2.0 ml/min, Column Temperature: 25°C, Column: Phenomenex RP 18, Column Dimension: 5cm 1.5 cm,LC detection: 254nm and Soft Ware: Class V P Integrated.Validation experiments were performed to demonstrate system suitability, linearity, precision, accuracy study and robustness as per ICH guidelines.
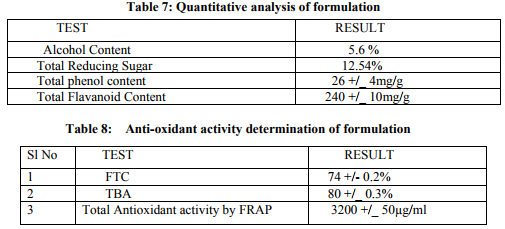
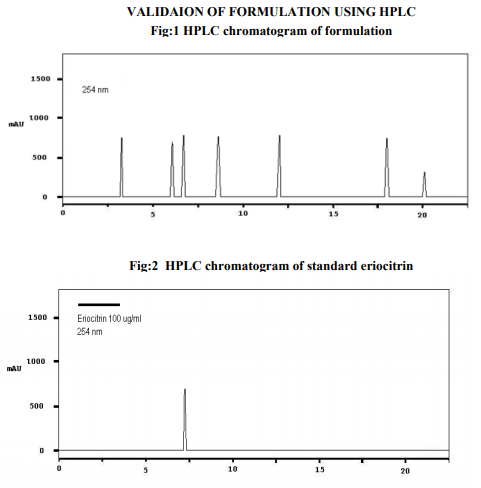
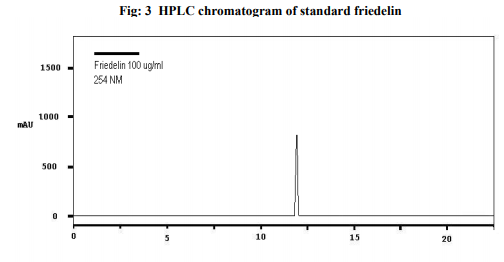
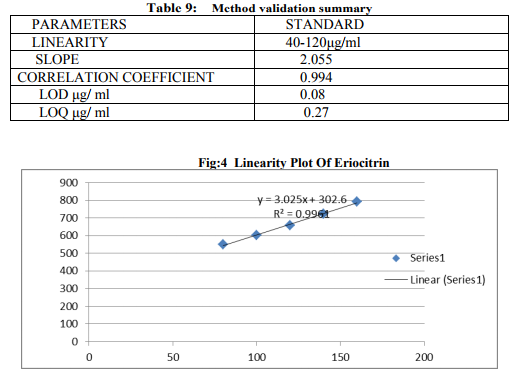
CONCLUSION
Laportea interrupta (L) Chew of family Urticaceae found to have number of active constituents from LCMS reports, which may can take as a proof for its traditional uses. Laportea interrupta leaves have been successfully formulated and determined its anti-oxidant activity and validated using UV and HPL
References:
1. RNS Yadav , Munin Agarwala. Phytochemical Analysis of Some Medicinal Plants, Journal of Phytology .2011; 3(12):10-14.
2. N. Savithramma, M. Linga Rao and D. Suhrulatha. Screening of Medicinal Plants for Secondary Metabolites, leucoanthocyanins and emodins. Middle-East Journal of Scientific Research, 2011; 8 (3):579-584.
3. S.K Sharma, Compliance of Pharmacopoeial Quality Standards of Ayurvedic Medicine, July-sep 2009;30:221-224.
4. S.Sekar. Traditionally Fermented Biomedicines, Arishtas and Asavas from Ayurveda, Oct 2008;7(4): 548-556.
5. Richa Kushwahaet al, Standardization of Aswagandharishta Formulation by TLC Method, International Journal of Chem Tech Research.2011; 3: 1033-1036.
6. S.F.Sayyad, Preparation and Evaluation of Fermented Ayurvedic Formulation: Arjunarishta, Journal of Applied Pharmaceutical Science, 2012 ;2(5), 122-124.
7. Shitalgiramkaret al,Effect of Pre-Sterilization on Physicochemical Parameters and in Vitro Free Radicle Scavenging Potential of Saraswatarishta, Journal of Pharmacy Research, 2012, Vol 5(5), pp: 2657-2663.
8. Giuseppagattusoet al, Flavanoid Composition of Citrus Juices, 2007,Vol: 12, pp: 1641-1673.
9. Luis adriano.S.do nascimentoet al,Biflavones and Triterpenoids isolated Fromouratea castaneifolia (dc.)Engl. Ochnaceae, RevistaBrasileira de Farmacognosia, oct-dec 2009,Vol: 19(4), pp: 823-827..
10. Rezaeizadeh A, Determination of Antioxidant activity in Methanolic and Chloroformic extracts of Mamordica charantia, African Journal of Biotechnology, (2011); 10(24):4932-4940.
11. N.P. Damodaran, Standardisation of Ayurvedic Medicines-Dasamulam Kasayam, Anc Sci Life, 1989; 9(2): 54–60.
12. Kunle et al, Standardization of Herbal Medicines - A Review, International Journal of Biodiversity and Conservation, March 2012; 4(3): 101-112.
13. Ajay KR Meena , Standardisation of Ayurvedic Polyherbal Formulation-Pancasama Churna, International Journal of Pharmacognosy and Phytochemical Research,2010; 2(1): 11-14.
14. Maithani Jyoti , Preparation and Standardization of a Polyherbal Formulation, Journal of Advanced Scientific Research,2012;3(2): 84-85.
15. Pravin Het , Future Trends in Standardization of Herbal Drugs, Journal of Applied Pharmaceutical Science,2012;02 (06): 38-44.
16. Neeli Rose Ekka. Kamta Prasad Namdeo , Pradeep Kumar Samal ,Standardization Strategies for Herbal Drugs- An Overview, Research J. Pharm and Tech, 2008; 1(4):310-312
17. ManishaK.Gharate, Development and Validation of RP-HPLC Method for Determination of Marker in Polyherbal Marketed Kankasava Formulation, Scholars Research Library, 2011;3(5) : 28-33.
18. L. Haber , Validation of HPLC method, Biopharm, 1999; 12: 64-66.
19. Himanshu Kumar, Prashant Kumar Pandey, V. V. Doiphode, Sanjay Vir, K. K. Bhutani, M. S. Patole, Y. S. Shouche. HPLC Analysis And Standardisation Of Arjunarishta- An Ayurvedic Cardio Protective Formulation, Scientia Pharmaceutica. 2013; 53(1):11-17
20. N. Vador ,B. Vador, Rupali Hole. Simple Spectrophotometric Methods Of Standardizing Ayurvedic Formulation, Indian J Pharm Sci, 2012,74(2): 161–163.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License