IJCRR - 6(11), June, 2014
Pages: 91-96
Date of Publication: 13-Jun-2014
Print Article
Download XML Download PDF
ROLE OF OXIDATIVE STRESS IN DIABETIC RETINOPATHY
Author: Sri Hari Atti, Angirekula Saseekala, Varun Siri A.
Category: Healthcare
Abstract:Objective: Diabetic retinopathy is the most commonest form of complications of diabetes mellitus which leads to vision loss if untreated. It has been hypothesized that oxidative damage may be involved in the pathogenesis of development of diabetic retinopathy. The aim of our present study is to evaluate the role of oxidative stress in diabetic retinopathy patients. Methods: We tried to assess the role of oxidative stress in patients by estimating the levels of lipid peroxidation assessing plasma Malondialdehyde(MDA) levels and antioxidant status by reduced glutathione(GSH), Vitamin-E ,Vitamin-C in blood. For this, we have taken 50 cases of diabetic retinopathy compared with 25 age matched controls . Results: There were significant increase in the levels of MDA and significant decreases in the levels of antioxidants like GSH, vitamin-E and Vitamin-C in patients when compared with controls. Conclusion: Our results indicate that oxidative stress is associated with the development of diabetic retinopathy which needs further studies.
Keywords: Diabetes, retinopathy, oxidative stress, antioxidants.
Full Text:
INTRODUCTION
Diabetes mellitus comprises a group of common metabolic disorders that share the phenotype of hyperglycemia. It is the most common endocrine metabolic disorder, affecting about 170 million people worldwide(1). The incidence of diabetes mellitus and its complications are increasing rapidly in India due to an aging population and the westernization of lifestyles. The ophthalmic complications of diabetes include corneal abnormalities, glaucoma, iris neovascularisation, cataracts and neuropathies. However, the most common and the potentially most blinding of these complications is diabetic retinopathy(2). Diabetic retinopathy is caused primarily by microvascular injury and the duration of diabetes, hyperglycemia, hypertension, and Hyperlipidemia have been shown to be risk factors for diabetic retinopathy(3). It is known that 20-50% of the long duration cases show proliferative diabetic retinopathy. In diabetes, the retina experiences increased oxidative stress(4), and reactive oxygen species are considered as a link between elevated glucose and the metabolic abnormalities important in the development of diabetic complications.(5).Oxidative stress increases in diabetes owing to an increase in the production of oxygen free radicals and a deficiency in the antioxidant defense mechanisms. The lipid peroxidation of the cellular structures, a consequence of the increased oxygen free radicals is thought to play an important role in the atherosclerosis and the microvascular complications(6).Elevated glucose causes a hypoxia like imbalance by increasing the NADH to NAD ratio. This altered ratio has been hypothesized to be a mechanism for ischemic retinopathy(7) and a cause of increased production of the superoxide ion(8). Greater ischemia manifested by capillary non perfusion of the retina leads to greater risk of extraretinal neovascularization and proliferative diabetic retinopathy, which is partly in response to up regulation and release of VEGF(9). Increased superoxide ion increases the oxidative load with greater reactive oxidative intermediates and advanced glycation end products, which also lead to increased release of VEGF(10) and the risk of neovascularization(11). Both ischemia and increased oxidation can lead to an increased production of lipid peroxides, which are themselves angiogenic(11). Taking the above facts into consideration, the present study was planned to evaluate the possible role of oxidative stress and antioxidant status in the pathophysiology of diabetic retinopathy patients.
MATERIALS AND METHODS
The present study was conducted at the Department of Opthalmology and the department of biochemistry at S.V.Medical college, Tirupati. A total of 50 known cases of diabetes mellitus patients of age group 45-75 were selected for this study. Age and sex matched 25 normal healthy adults were selected as controls. Informed consent from all cases and controls were obtained. Ethical clearance was obtained for this study. A detailed medical history was obtained to identify those with known or suspected systemic hypertension, peripheral or coronary artery disease, venous thrombotic events, cerebrovascular disease, alcohol abuse, smoking and current use of cholesterol lowering drugs or vitamin supplements. All subjects underwent a complete opthalmoscopic examination including visual acuity, slit lamp examination, intraocular pressure measurement using tonometry, gonioscopy, and dilated fundoscopic examination and systemic examination. Based on ophthalmic and systemic examinations, the study had two groups. Group 1 included 25 age matched healthy adults as controls and Group 2 included 50 patients of type-2 diabetes with retinopathy out of which 23 had non proliferative diabetic retinopathy and 27 had proliferative diabetic retinopathy. 10 ml of fasting blood samples were collected by venipuncture and for the separation of sera, 5ml of blood was centrifuged at 3000rpm for 5min and the remaining 5ml of blood was taken into a plain vial containing EDTA and was centrifuged at 3000rpm for 10min for the separation of plasma. The plasma MDA levels were estimated by using thiobarbituric acid reacting substances(TBARS) by the method of Yagi(12) and Sinnhuber et al(13). Reduced glutathione was determined by the method of Beutler et al(14). Serum Vit-E was measured by the method of Baker on the basis of reduction of ferric ions to ferrous ions by Vit-E and the formation of red colored complex with 2-2’ dipyridyl at 520nm(15). The activity of Ascorbic acid was determined by the method of Teitz(16). All the results were expressed as mean±SD and statistical comparisons were done using student t-test using the SPSS package and ANOVA correlation study.
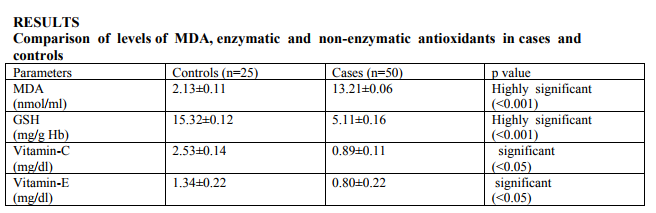
Evaluation of oxidative stress is done based on the levels of MDA and statistically significant increase in the levels of MDA was observed in diabetics with retinopathy when compared to controls. Statistically significant decreases were observed in the levels of enzymatic antioxidants like GSH and non enzymatic antioxidants like vitaminE and C in cases when compared to controls.
DISCUSSION
Diabetic retinopathy is one of the most common microvascular complications of diabetes and ranks as a common cause of blindness worldwide(17). Diabetic retinopathy could become a major threat to public health in the future due to the global prevalence of diabetes of diabetes, which is projected to affect 438 million people by 2030(18). Both the duration of diabetes and its metabolic control have been identified as the risk factors most strongly associated with the development of diabetic retinopathy(19). Diabetic retinopathy occurs in 70% of all persons having diabetes for more than 15 years. The prevalence of diabetic retinopathy among urban subjects with diabetes in India was reported to be about 17%(20), whereas in a clinical study it was found to be 34% among patients with type 2 diabetes(21). The prevalence of diabetic retinopathy was 0.5% in the general rural populations of south India and 10.5% among patients with diabetes(22). Diabetic retinopathy is characterized by the appearance of vascular lesions of increasing severity, culminating in the growth of new vessels. Several hypotheses have been tested on the cause and pathogenesis of diabetic retinopathy. In non proliferative retinopathy, prolonged hyperglycemia may produce microaneurysms because of the selective decrease of cells in the vicinity of the retinal microvasculature and may also induce thickening of the vascular fundus membrane, endothelial proliferation, microvascular dilatation due to decreased endothelin-1 synthesis, increased vascular permeability and vascular occlusion in microvascular endothelial cells. In addition, prolonged hyperglycemia may increase blood flow and viscosity, resulting in increase of shear stress against the vascular wall, as well as injury of the vascular wall, in severe cases, it is known to induce macular edema(23). However, the main pathogenesis of proliferative retinopathy is known to be neovascularization caused by several factors including insulin like growth factor released from a retinal ischemic area(23). Recently, four biochemical pathways including enhanced glucose flux through the polyol pathway, increased intracellular formation of advanced glycation end-products, activation of protein kinase C isoforms, and stimulation of the hexosamine pathway have been suggested for eye disorders caused by hyperglycemia induced vascular injury in diabetes. These pathogenic mechanisms also appear to be associated with superoxide overproduction by the mitochondrial electron transport chain(24). In our study it was found that MDA levels of subjects with diabetic retinopathy were significantly higher than the levels of healthy controls. MDA level acted as markers of lipid peroxidation which represents oxidative stress and the reasons for increased lipid peroxidation products may be due to increased oxygen products as a result of auto-oxidation of glucose and glycosylated proteins, polyol pathways and decreased non enzymatic antioxidants. In addition, hyperglycemia increases the formation of triose phosphate, whose oxidation causes the formation of two free radicals alpha aldehyde and hydrogen peroxide(25). The increased levels of lipid peroxides can cause oxidative injury to blood cells, cross linking of membrane lipids and proteins, increasing of cell ageing, imbalance of prostacyclin/prostaglandin and vasoconstriction(26).GSH has a key role in enzymatic defense systems and helps in removing peroxides. In our study, there was statistically significant decrease in the levels of reduced glutathione when compared to healthy subjects. In the literature, different results have been reported in different organs and tissues regarding the levels of these two enzymes in diabetes mellitus. The low levels of antioxidant enzymes in diabetes mellitus may be because of the inactivation of the antioxidant enzymes by non enzymatic glycation due to persistent hyperglycemia or GSH could be overused implying a reduction of protection against reactive oxygen species, possibly due to a defective redox cycle or it may be due to defective intracellular synthesis. Statistically significant low levels were observed in the levels of vitamin- C and E in cases when compared to controls. Vitamin-C is water soluble vitamin and vitamin-E is fat soluble and they are the most important antioxidant vitamins, inactivates free radicals found in the cytosol, plasma and extracellular environment. The low levels of these vitamins suggest the overusage which again increases the oxidative stress. Thus, this study concluded that the increased oxidative stress and the decreased antioxidant status can predict the micro-vascular complications in diabetes mellitus. The raised MDA levels indicate the oxidative stress and the decreased GSH, Vitamin-C and E levels indicate the reduced anti oxidative status in diabetic retinopathy.
CONCLUSION
Hence, for the early detection and prevention of diabetic retinopathy, it is advisable to estimate the oxidative stress markers. It is also advised that vitamin supplements should also be taken to allow the non enzymatic and enzymatic antioxidant systems to respond to oxidative stress.
ACKNOWLEDGEMENTS
Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors/editors/publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
References:
1. Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes. Estimates for the year 2000 and projections for 2030. Diabetes Care, 2004;27:1047-1053.
2. Gupta.M.M, Chari.S. Lipid peroxidation and the antioxidant status in patients with diabetic retinopathy. Indian J Physiol Pharmacol, 2005;49(2):187-192.
3. Nguyen TT, Alibrahim E, Islam FM et al. Inflammatory, hemostatic and other novel biomarkers for diabetic retinopathy: the multi-ethnic study of atherosclerosis. Diabetes Care 2009;32:1704-9.
4. Kowluru RA, Atasi L, Ho YS. Role of mitochondrial superoxide dismutase in the development of diabetic retinopathy. Invest Ophthal Vis Sci, 2006; 47(4):1594- 1599.
5. Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature, 2001;414(6865):813-820.
6. Soliman GZA. Blood lipid peroxidation(superoxide dismutase,Malondialdehyde,glutathione) levels in Egyptian type 2 diabetic patients. Singapore Med J, 2008;49(2):129- 136.
7. Van den Enden MK, Nyengaard JR, Ostro WE, Burgan JH, Williamson JR. Elevated glucose levels increase retinal glycolysis and sorbitol pathway metabolism: implications for diabetic retinopathy. Invest Ophthalmol Vis Sci, 1995;36:1675- 1685.
8. Williamson JR, Chang K, Frangos M et al. Hyperglycemic pseudohypoxia and diabetic complications( Perspectives in Diabetes). Diabetes, 1993;42:801-813.
9. Aiello LP, Northrup JM, Keyt BA, Takagi H, Iwamoto MA. Hypoxic regulation of vascular endothelial growth factor in retinal cells. Arch Ophthalmol, 1995;113:1538-1544.
10. Lu M, Kuroki M et al. Advanced glycation end products increase retinal vascular endothelial growth factor expression. J Clin Invest,1998;101:1219-1224.
11. Armstrong D, Ueda T et al. Lipid hydroperoxide stimulates retinal neovascularization in rabbit retina through expression of tumor necrosis factor-alpha, vascular endothelial growth factor and platelet derived growth factor. Angiogenesis,1998;2:93-104.
12. Yagi K. Lipid peroxides and human diseases. Chem Phys Lipids,1978;45:337- 351.
13. Sinnhuber RO, Yu TC. Characterization of red pigment formed in thiobarbituric acid determination of oxidative rancidity. Food Res,1958;23:626-630.
14. Beutler E, Duron O, Kelly BM. Improved method for determination of blood glutathione. J Lab Clin Med, 1963;61:882- 888.
15. Baker and Frank. Determination of serum tocopherol by colorimetric method. In Varley’s practical clinical biochemistry, Heinmann professional publishing 1998;6th edition.902.
16. Tietz NW(Ed). Textbook of Clinical Chemistry. W.B.Saunders Company, Philadelphia, London, Toronto.2004;pp.960- 962.
17. Kempen JH, O’ colmain BJ et al. Eye Diseases Prevalence Research Group. The prevalence of diabetic retinopathy among adults in the United States. Arch Ophthalmol, 2004;122:552-563.
18. International Diabetes Federation. The Diabetes Atlas, 2009; 4th edition. Brussels. 19. The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long term complications in insulin dependent diabetes mellitus. New Engl J Med,1993;329:977-986.
20. Rema M, Prem kumar S et al. Prevalence of diabetic retinopathy in urban India: the Chennai Urban Rural Epidemiology Study(CURES) eye study. Invest Ophthalmol Vis Sci,2005;46:2328-33.
21. Rema M, Ponnaiya M, Mohan M. Prevalence of retinopathy in non insulin dependent diabetes mellitus at a diabetes center in southern India. Diab Res Clin Pract.1996;34:29-36.
22. Nirmalan PK, Katz J, Robin AL et al. Prevalence of vitreoretinal disorders in a rural population of Southern India: the Aravind Comprehensive Eye Study. Arch Ophthalmol,2004;122:581-586.
23. Kim TW.Pathogenesis of diabetic retinopathy. J Korean Diabetes Assoc,1997;21:115-21.
24. Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature,2001;414:813-20.
25. Sundaram RK, Bhaskar A, Vijayalingam S et al. Antioxidant status and lipid peroxidation in type 2 diabetes mellitus with and without complications. Clin Sci,1996;90:255-60.
26. Jain SK, Mc Vie R, Jaramillo JI et al. The effect of modest vitamin E supplementation or lipid peroxidation products and other cardiovascular risk factors in diabetic patients. Lipids,1996;31:87-90.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License