IJCRR - 6(14), July, 2014
Pages: 01-06
Date of Publication: 22-Jul-2014
Print Article
Download XML Download PDF
EFFECT OF SLEEP DEPRIVATION ON MEMORY IN RATS
Author: Damel Lakshmi, Kayalvizhi, Chandrasekhar, Priyadarshini
Category: Healthcare
Abstract:Objective: To study the effect of sleep deprivation on memory in rats. Methods: The experiment was done at Department of Physiology, Meenakshi Medical College and Research Institute, Enathur, Kanchipuram. Ethical clearance was obtained from CPCSEA. 12 male Wistar albino rats were used in the study, control group (6) and study group (6). Sleep deprivation was performed by using single platform method (Flower pot method). Study group was exposed to 5 hours of sleep deprivation for 5 days. Behavior, learning and memory was assessed using T maze and radial arm maze. Results: Rats subjected to sleep deprivation showed alteration in behavior and memory impairment. In T maze, there was significant increase in time spent in left arm, number of fecal bolus count with significant decrease in number of rearing, grooming and number of urination were decreased when compared with control group. In Radial arm maze impairment in memory was observed when compared with the control group. Conclusion: Rats showed memory impairment after subjected to sleep deprivation, which was assessed by standardized T maze and Radial arm maze.
Keywords: Single platform method, T Maze, Radial arm Maze.
Full Text:
INTRODUCTION
Sleep is a naturally or artificially induced state of sensory and motor activity1 . Sleep occupies approximately one-third of a person’s life19 . Normal adult have 8 – 9 hours of sleep in a day. Sleep has two phases; Non-rapid eye movement sleep (NREM) and rapid eye movement sleep (REM). Sleep includes 5-6 cycles / day. Each cycle has NREM sleep for 70 minutes and REM sleep for 20 minutes12. NREM sleep is generated by pre optic region of hypothalamus and its adjacent basal forebrain. REM sleep is generated by pons and adjacent portions of midbrain10. Suprachiasmatic nucleus (SCN) maintains the circadian cycle in the mammalian body. One of the most prominent circadian rhythm is the cycle of sleep and wakefulness. All physiologic and behavioral functions are generated by a circadian pacemaker of SCN located in the anterior hypothalamus11. Sleep plays a major role in wound healing, basal metabolic rate, energy conservation, development of brain, learning and memory processing, cognitive functions, dreaming22 . Sleep deprivation is the condition of not having enough sleep. It is considered as an extreme case of sleep reduction, in that the organism is simply awake for a prolonged period of time2 . Deprived sleep may occur due to various reasons like physiological stress, a poor sleep environment and an inconsistent sleep schedule. Common physiological effects of deprived sleep are headache, malaise, increased blood pressure, increased stress hormone levels, irritability, confusion, memory lapses, and depression21 . Recalling of learned events at conscious or sub conscious level is called memory, which includes non-declarative (procedural) memory and declarative memory constituting short term (working) memory and long term memory9 . Memory consolidation is a slow process which converts learned events into a more permanent or enhanced form by uniting the medial temporal lobes and neocortical areas7 . Paradoxical sleep (PS) has a major role in the development of nervous system and neural plasticity; it is also assumed that immediate or short term memory storage takes place during PS4 . Brain-derived neurotrophic factor (BDNF) is a member of the neurotrophin family, which plays important roles in the survival, maintenance, and growth of neurons. It supports the growth of many neurons, complexity of dentrites in the cerebral cortex. It also enhances synaptic transmission and long term potentiation which is considered to be a potent cellular mechanism in learning and memory. The purpose of this experiment is to study the effect of paradoxical sleep deprivation on memory in rats.
MATERIALS AND METHOD
The experiment was carried out in the Department of Physiology, Meenakshi Medical College hospital and Research Institute, Enathur, Kanchipuram. Proper ethical clearance was obtained from Institutional Animal Ethical Committee. 12 male Wistar albino rats weighing 800 to 900g were used for the study. The animals were grouped into 2 groups with 6 animals in each; Control group (6) and study group (6). The animals were adapted to the laboratory environment 7 days prior to the initiation of the experiment and 12 hours dark/light cycle was maintained (4). Study group rats were subjected to sleep deprivation by single platform method. Control group rats were left free in home cage with free access to food and water in the same environment were sleep deprivation is performed during the period of experiment. Single platform method: This apparatus consists of a small platform (6.5 cm in diameter and 10 cm high) inside the chamber (22 x 22 x 35 cm) (Fig:1). The platform was submerged in water until 1-2 cm below its surface (Jouvet et. al. 1964)16 . Rats were placed over the small platform for 5 hours for 5 days between 9.00 am to 2.00 pm for sleep deprivation. After 5 days of sleep deprivation the rats were allowed to explore in T maze and radial arm maze. T maze: It is used to assess rodent behavior with reference and working memory. A wooden T maze apparatus was made which has a stem (50x16 cm), right arm and left arm (50x10 cm). The side walls are raised to 30 cm height4 (Fig: 2). Rats were exposed to T maze for 10 minutes then observed for side preference.
Radial arm maze: It is used to evaluate working memory. It consists of eight equally spaced arms from octagonal central platform. Each arm measures 50x12 cm extends from octagonal shaped central hub of 30 cm diameter. The platform is elevated 40 cm above the floor. Small food wells (3cm in diameter) were placed at the end of each arm (Fig:3). Rats were exposed for 10 min to observe how rats were using the spatial cues and working memory for choice of arm. Rearing, grooming, urination and fecal bolus count were also observed to assess the level of stress due to sleep deprivation method.
RESULTS
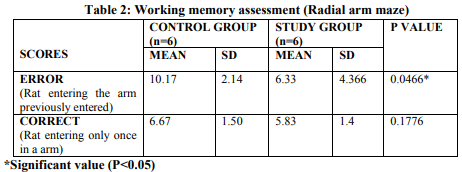
The scores were tabulated, mean and standard deviation was calculated using student t-test (Table 1.1) Significant increase (P< 0.05) in the time spent in left arm of T maze was observed in sleep deprived rats when compared with control group rats. In T maze rearing, grooming and fecal bolus count were also increased and urination was decreased in sleep deprived rats than control group (Table 1.2). In Radial arm maze, impairment in the memory level was observed by significant increase (P< 0.05) in the error scores of sleep deprived rats than control rats (Table 2).
DISCUSSION
In our study, we have found that 5 hours of sleep deprivation for 5 days produces various behavioral alterations linked with memory impairments. Sleep is a common feature of many sleep disorders in humans. For this reason effect of behavioral and memory performances were seen in animal models of interest2 . Sleep deprivation can have negative consequences on both the body and the mind interfering with physical energy and cognition. Memory loss is often associated with old age, but even young people may experience the cognitive impairment if they have sleep reduction. This study also correlates well with the report given by Health News Team (2011) which shows that adenosine reduction after sleep deprivation that could be due to the increased levels of nucleosides which might lead to decrease in memory loss and attention deficits. In our study, we have identified that sleep deprivation is a potent stressor inducing behavioral changes which is also similar to the findings of Shyamala et. al., (2012) reported that REM sleep deprivation enhanced the plasma corticosterone level. Increased level of lipid peroxidation was also reported with free radical induced neuronal damage, in thalamus and hypothalamus. The sleep deprivation performed by using flower pot method showed a significant increase in error scores of radial arm maze when compared with control group rats which indicates impairment in memory level which is also similar to the findings Shenghui Li, 2009, who reported that 48 hours of REM sleep deprived rats showed impairment in retention of acquired spatial reference memory in Morris water maze than control group. The impairment continuously existed even after 24 and 48 hours of release from sleep deprivation could be due to decrease in the levels of acetylcholine and synaptic connections in hippocampus, which mainly impairs spatial reference memory. This shows REM sleep is mainly required for memory consolidation (Chumin Zhu et. al., 2008). The increase in error scores in Radial arm maze also correlates well with the findings of Saha et. al.,2010, who explained that the alteration of memory and behavior could be due to decrease in level of melatonin which leads to increase in free radicals, a probable effect of sleep deprivation. Simultaneously, there was an increase in the time spent in left arm of T maze of sleep deprived rats showing inability to recognize the spatial cues which might be due to inactivation of Zif268 gene during sleep deprivation which inhibits consolidation of memory. (Carmel et. al., 2003). He also reported that sleep deprivation impaired performance on hippocampus – dependent spatial learning task, reduced neuronal excitability in CA1 pyramidal neurons. He also explains that consolidation of memory was reduced due to inhibition of CA1 pyramidal neurons and dendate gyrus granule cell because of long term potentiation of synaptic strength inhibition.
CONCLUSION
Thus rats on exposed to sleep deprivation showed impairment in memory. Failure to acquire adequate sleep produces various changes in the development of memory due to several cellular and molecular level alterations inhibiting hippocampal functioning. But still the exact mechanism causing the alterations in memory remains unclear. Hence further studies involving molecular parameters might help in understanding the mechanism of memory impairment following sleep deprivation.
ACKNOWLEDGEMENT
We are very much thankful to all the authors whom we have referred for this study.
References:
1. Essentials of Medical Physiology, Anil Baran Singha Mahapathra, Third Edition, Higher Neural Functions, Sleep Page no 469, ISBN 81- 86793-60-0
2. Sergio Tufik, Monica L. Andersen, Lia R.A. Bittencourt and Marco T. De Mello.Paradoxical Sleep Deprivation: neurochemical, hormonal and behavioral alterations. Evidence from 30 years of research. Anais da Academia Brasileira de Ciências (2009) 81(3): 521-538
3. Fundamentals of Medical Physiology, Lingala Prakasam Reddy, third edition, Sleep, page 129,130; ISBN: 81-8191-016-8
4. Jerome M. Siegel, The Neurobiology of Sleep, Semin Neurol 2009; 29;277-296
5. Kathryn J. Reid, Phyllis C. Zee, Circadian Rhythm Disorders, Semin Neurol 2009; 29;393-405
6. Sleep Wikipedia, http://en.wikipedia.org/wiki/Sleep
7. Sleep deprivation Wikipedia, http://en.wikipedia.org/wiki/Sleep_deprivation
8. Deborah Suchecki, Leticia L, Lobo, DeBora C . Hipolide And Sergio Tufik, Increased ACTH and corticosterone secretion induced by different methods of paradoxical sleep deprivation. J. Sleep Res. (1998)
7, 276–281 Accepted in revised form 25 June 1998; received 26 November 1997
9. Chumin Zhu, Progressive paradoxical sleep deprivation impairs partial memory following learning tasks in rats
10. Robert M J Deacon and J Nicholas P Rawlins Tmaze alternation in the rodent, 27 June 2006; doi:10.1038/nprot.2006.2
11. Researchers Discover How Lack of Sleep May Cause Memory Loss By Health News Team• May 18th, 2011 • Category: Memory Problems, True Health News
12. Cordova CA, Said BO, McCarley RW, Sleep deprivation in rats produces attentional impairments on a 5-choice serial reaction time task. SLEEP 2006; 29(1): 69-76.
13. D.C. Mathangi, R. Shyamala and A.S. Subhashini, Effect of REM sleep deprivation on the antioxidant status in the brain of Wistar rats. Annals of Neurosciences, Volume 19, Number 4 October 2012
14. Stress, http://www.medicinenet.com/script/main/art.as p?articlekey=488
15. Text book of medical physiology, Indu khurana, 2006, learning and memory, ISBN: 978-81-8147-850-4
16. Makoto Mizuno,Kiyofumi Yamada,Ana Olariu,Hiroyuki Nawa, and Toshitaka Nabeshima, Involvement of Brain-Derived Neurotrophic Factor in Spatial Memory Formation and Maintenance in a Radial Arm Maze Test in Rats, The journal of Neuroscience, September 15, 2000, 20(18):7116–7121
17. Shenghui Li, Ying Tian, Yu Ding, Xinming Jin, Chonghuai Yan, Xiaoming Shen, The effects of rapid eye movement sleep deprivation and recovery on spatial reference memory of young rats, Learning and behavior, August 2009, Volume 37, Issue 3, pp 246-253.
18. Habibur Rahman, P. Muralidharan, D. Sivaraman, Dipankar Saha. Continuous sleep deprivation for 5 days produces loss of memory in mice and may be a cause of Alzheimer’s disease Scholars Research Library. Annals of Biological Research, 2010, 1 (4) : 185-193.
19. Bruno Bozon, Sabrina Davis, and Serge Laroche, A Requirement for the Immediate Early Report Gene zif268 in Reconsolidation of Recognition Memory after Retrieval. Neuron, Vol. 40, 695–701, November 13, 2003.
20. Maquet,P.,. The role of sleep in learning and memory. Science, 2001, 294, 1048-1051.
21. Reimund, E., The free radical flux theory of sleep. Medical Hypothesis, 1994, 43, 231-233.
22. Smith TD, Adams MM, Gallagher M, et. al. Circuit- Specific alteration in hippocampal synaptophysin immune reactivity predict spatial learning impairment in aged rats. J.Neuroscience; 20(17): 6587-93.
23. Smith. CT, Nixon MR, Nadar RS. Post training increases in REM Sleep intensity implicate REM sleep in memory processing and provide a biological marker of learning potential. Learn Mem 2004; 11 (6): 714-9.
24. Youngblood BD, Zhou J. Smagin GN, et. al. Sleep deprivation by the “flowerpot” technique and spatial reference memory. Physiol Behavior 1997; 61 (2): 249-56.
25. T.W. Boonstra, J.F.Stins, A. Daffertshofer, P.J.Beek, Effects of aleep deprivation on neural functioning: an integrative review. Cell.Mol.Lofe Sci. 64 (2007) 934-946.
26. Geraldine Rauchs, Beatrice Desgranges, Jean Foret, Francis Eustache, The relationships between memory systems and sleep stages. J.Sleep Res (2005) 14, 123-140

|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License