IJCRR - 6(22), November, 2014
Pages: 47-49
Date of Publication: 21-Nov-2014
Print Article
Download XML Download PDF
ANAGEN EFFLUVIUM - A REVIEW
Author: Shashikant, Nagaraj Mallashetty
Category: Healthcare
Abstract:Anagen effluvium is a type of hair loss that follows the administration of chemotherapeutic drugs, radiation treatment and various chemical agents. It is characterized by hair breakage rather than hair loss. Hair shafts break at about the same time when the thin portion reaches the scalp surface. The hair loss in anagen effluvium is quite disturbing to patients and their family members. Dermatologists have little experience with anagen effluvium.
Keywords: Anagen effluvium, Diffuse hair loss
Full Text:
INTRODUCTION
Anagen effluvium (AE) is a type of diffuse hair loss that follows the administration of cytotoxic drugs, radiation treatment or various chemical agents and is characterized by hair breakage rather than hair loss.1 Anagen effluvium has an estimated incidence of 65% and is considered to be one of the most traumatic aspects of chemotherapy in female patients.2a Hair shafts break at about the same time when the thin portion reaches the scalp surface. Anagen effluvium can be managed with various drugs, even if anagen effluvium cannot be prevented.1
ETIOPATHOGENESIS
Anagen effluvium occurs during the anagen phase of growth because hair bulb cell divide rapidly and are sensitive to cytotoxic agents. Cytotoxic drugs impair the mitotic and metabolic processes in actively growing hair follicles, leading to thinning of the shaft, which becomes fragile and susceptible to fracture with minimal trauma. The molecular mechanism of AE has been associated with premature apoptosis-driven hair follicle regression, and p53, Fas and c-kit are the involved factors.1These agents can impair or totally disrupt the anagen cycle and cause varying degree of hair follicle dystrophy. The net result is either anagen hair that break off within the hair follicle or at the level of scalp (secondary to a weak point in the structurally inferior hair shaft) and are then shed without roots, or dystrophic anagen hair that are easily dislodged from the usual follicular moorings.3 Various causes of anagen effluvium has been presented in table 1.
CLINICAL FEATURES
Anagen effluvium is typically reversible. Severity of hair loss depends on the route of administration as well as the dose and frequency of administration. Hair shedding usually begins at 1-3 weeks after initiation of chemotherapy and becomes most clinically apparent in 1-2 months.
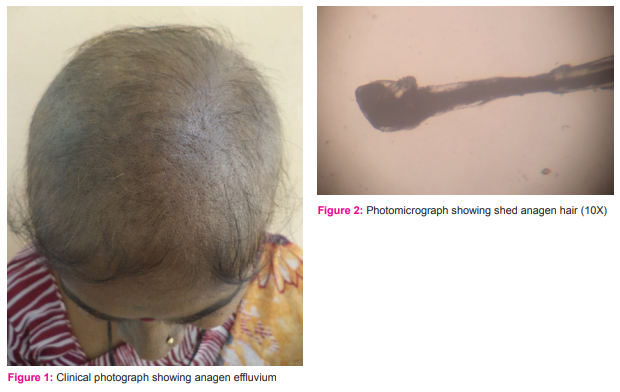
It most commonly affects scalp hair due to long anagen phase and to a variable degree terminal hair at other sites such as eyebrows, eyelashes, axillary and pubic hair.7 The World Health Organization criteria for alopecia is grade 0 = no loss, grade 1 = mild hair loss, grade 2 = moderate hair loss grade 3= reversible complete hair loss and grade 4 = irreversible complete hair loss.8 A careful history is an important key to identify triggers in any patient with diffuse hair loss.9 Anagen effluvium is a reversible condition, and hair regrowth begins several weeks after the cessation of chemotherapy. Hair loss is known to start from the area of mechanical friction such as crown and side of the head above the ears because these areas come in contact with bed linens, pillow and head covering. Nearly 85% of the total number of anagen hair are shed after chemotherapy and scalp hair those are in the telogen phase are not affected (figure 1).1 When hair regrows, approximately 65% of the patients experience a change from their previous hair. Some patients experience alteration in the colour, texture or type of hair.1 Regrowth of hair after radiation therapy depends upon type, depth, and dose-fractionation but it commonly leads to permanent follicular destruction, most likely as a result of irreversible hair follicle stem cell damage leading to scarring alopecia. In fact, this scarring alopecia may progress long after radiation therapy has been discontinued; possibly due to persistent radiation-induced inflammatory changes that progressively damage hair follicle stem cells. Low dose cytotoxic agents more often cause only telogen effluvium, because they induce premature catagen. High dose busulfan which is used in the preparatory treatment for bone marrow transplantation may lead to permanent alopecia due to irreversible damage to hair follicle stem cells.3 In a study by Korean author Jung Yun S, 20 among the 38 female patients of anagen effluvium had patterned hair loss. They did not notice any significant difference in the pattern of hair loss depending upon age, associated symptoms and chemotherapeutic agent groups.1 Hair loss resembling androgenetic alopecia and changes in the structure and colour have been reported with tamoxifen therapy.10
INVESTIGATIONS
Clinically diffuse hair loss can be diagnosed by hair pull test.11 The hair pull test is positive in anagen effluvium. Light microscopic examination shows dystrophic anagen hair (figure 2) with tapered ends and thinning or constriction of the hair shafts called Pohl-Pinkus constriction.9, 10
TREATMENT
If the insult ceases, growth of hair restarts within weeks.9 Various measures have been tried in order to prevent hair loss. Topical minoxidil has been found to decrease the duration of hair loss caused by chemotherapy. Minoxidil is not effective in preventing initial hair loss due to chemotherapeutic agents. It should not be used in patients undergoing chemotherapy for hematological malignancies with a curative intent.7 Scalp cooling has been reported as an effective method of preventing chemotherapy-induced alopecia.4 It involves cooling of the scalp with cold air or liquid. It produces vasoconstriction of the scalp vessels leading to reduced blood flow to the follicles during chemotherapy thus minimizing concentration of the antineoplastic agent in plasma.7 However it may not be effective when multiple drug regimes or very high doses of individual drugs are used. There are no specific guidelines on optimal method, temperature, and duration of scalp cooling at present.7
CONCLUSION
Anagen effluvium is one of the causes of diffuse hair loss. Severity of the hair loss depends on the route as well as dose and frequency of administration of chemotherapeutic drugs which is typically reversible.
ABBREVIATION
AE – Anagen effluvium
ACKNOWLEDGEMENT
We acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. We are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
References:
1. Yun SJ, Kim SJ. Hair loss pattern due to chemotherapyinduced anagen effluvium: a cross-sectional observation. Dermatology 2007;215:36-40.
2. Trüeb R. Chemotherapy-induced Hair Loss. Skin Therapy Lett 2010;15:5-7.
3. Paus R, Olsen EA, Messenger AG. Hair growth disorders. In: Wolff K, Goldsmith LA, Katz SI, Gilchrest BA, editors. Fitzpatrick’s dermatology in general medicine, 7thedn. NewYork: McGraw Hill; 2008.p.- 753-778.
4. Chadha V, Shenoi SD. Hair loss in cancer chemotherapeutic patients. Indian J Dermatol Venereol Leprol 2003;69:131- 132.
5. Habif TP. Hair diseases. In: Habif TP, editor. Clinical dermatology: A color guide to diagnosis and therapy, 3rd edn. St. Louis: Mosby; 1996.p.- 739-747.
6. Wadhwa SL, Khopkar U, Nischal KC. Hair and scalp disorders. In: Valia RG, Valia AR, editors. IADVL Text book of dermatology, 3rd edn. Mumbai: Bhalani Publishing House; 2010.p.- 864-948.
7. Atanaskova Mesinkovska N, Bergfeld WF. Hair: what is new in diagnosis and management? Female pattern hair loss update: diagnosis and treatment. Dermatol Clin 2013;31:119- 127.
8. Protière C et al. Efficacy and tolerance of a scalp-cooling system for prevention of hair loss and the experience of breast cancer patients treated by adjuvant chemotherapy. Support Care Cancer. 2002;10:529-37.
9. Harrison S, Bergfeld W. Diffuse hair loss: its triggers and management. Cleve Clin J Med 2009;76:361-367.
10. Lindner J et al. Hair shaft abnormalities after chemotherapy and tamoxifen therapy in patients with breast cancer evaluated by optical coherence tomography. Br J Dermatol 2012;167:1272-1278.
11. Rustom A, Pasricha JS. Causes of diffuse alopecia in women. Indian J Dermatol Venereol Leprol 1994;60:266-271.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License