IJCRR - 6(24), December, 2014
Pages: 43-47
Date of Publication: 20-Dec-2014
Print Article
Download XML Download PDF
BIOCHEMICAL CHANGES HUMAN IMMUNODEFICIENCY VIRUS INFECTED PATIENTS BEFORE AND AFTER ANTIRETROVIRAL THERAPY
Author: Usha kiran P., Komala P.
Category: Healthcare
Abstract:Objectives: The aim of present study was to evaluate the levels of liver enzymes and lipid abnormalities in patients receiving antiretroviral therapy compared to controls. Lactic acidosis was one of the known complications of ART which is the major cause of discontinuation of treatment. Methods: Siemens Dimension Expand Plus fully automated random access analyzer was used for analyzing different parameters. Results: Mean plasma levels of ALT and AST were more in HIV infectedpatients than control group. These parameters were further increased in HIV infected patients after ART. Mean plasma levels of cholesterol and HDL are lower than control group in HIV infected patients before treatment. Cholesterol levels increased after HAART whereas HDL level decreased. Mean plasma levels of triglycerides, LDL and lactic acid are high in HIV infected patients than control and they are further increased after ART. Conclusion: Serum liver enzymes are significantly increased after ART and may be used as prognostic marker of lactic acidosis. This study helps in diagnosing lactic acidosis which can be treated successfully so as to render patients to adhere to treatment to decrease mortality.
Keywords: HIV, ART, Lactic acid, Liver enzymes, Lipid profile
Full Text:
INTRODUCTION
Most antiretroviral agents have been associated with hepatic toxicity. NRTIs can cause hepatic steatosis generally after more than 6 months of therapy1 , probably via mitochondrial toxicity. NRTIs can cause hepatitis in first 2-3 months of therapy, sometimes as part of a hypersensitivity reaction. Protease inhibitors can also cause hepatitis2 by an unknown mechanism, particularly in patients co-infected with hepatitis B or hepatitis C and with raised hepatic aminotransferases concentration and less commonly have been associated with variceal bleeding in patients with cirrhosis3 . Some cases of hepatitis with antiviral seem to represent a side effect of an improved immune response, where immune restoration leads to recognition of hepatitis B4 or C in chronic carriers and results in a clinical episode of hepatitis with sero conversion. What proportion of hepatic reaction to any retroviral is caused by such mechanism is not known. Unconjugated hyperbilirubinemia can occur with indinavir (about 7%) but does not represent liver toxicity5 . Pre – HAART dyslipidemia was caused by an initial decrease in serum levels of total cholesterol, HDL and LDL followed by increase in triacylglycerol during advanced stages of HIV infection.6, 7 The severity of observed dyslipidemia was related inversely to decreased CD4 lymphocyte count particularly when CD4 counts were below 200.8 In contrast, correlation between CD4 lymphocyte count / viral load levels and hyperlipidemia in patients taking PI containing HAART regimen have not been found.9, 10 Several cross-sectional studies have established a relationship between PI use and hypercholesterolemia and hypertriglyceridemia.11,12 Studies which followed patients before and after PI exposure noted higher cholesterol and triglyceride levels after PI initiation.13,14 The average increase of total cholesterol and triglyceride levels was 28% and 29% respectively from baseline pretreatment levels.15 Following a sample of 221 HIV- infected patients for 5 years, Tsiodras and colleagues 2000 found that PI therapy was associated with a 2.8 fold increased rate of hypercholesterolemia and a 6 fold increased rate of hypertriglyceridemia. Lactic acidosis Lactic acidosis results from an increase in blood lactate levels when lactate production exceeds consumption and body buffer systems become overburdened. Lactic acidosis is classified by Cohen and Woods into 2 broad categories Type A lactic acidosis is due to decreased tissue perfusion may be due to over production or underutilization. Type B2 lactic acidosis is due to drugs without evidence of poor tissue perfusion or oxygenation or with occult tissue hypo perfusion. NRTIs have been associated with functional and structural mitochondrial abnormalities leading to lactic acidosis.16 Mild to moderate asymptomatic hyperlactatemia has been frequently reported in patients treated with an estimated prevalence between 15% and 35%. On the contrary, symptomatic, severe hyperlactatemia and lactic acidosis are less common, with an incidence ranging from 1.7 to 25.2 cases per 1000 person- years of antiretroviral treatment, and are associated with a remarkable mortality rate, which varies from 30% to 60% in different studies.17
MATERIALS AND METHODS
The present study was conducted in department of Biochemistry, Rangaraya Medical College, Kakinada, Andhra Pradesh, India. The present study was undertaken to determine biochemical changes in HIV infected patients before and 2 months after ART in venous blood consisting of 100 patients. These values are compared with 50 normal healthy persons. All of these subjects were taken from ART centre, Government General Hospital, Kakinada. IRB/ Ethics Committee decided approval was not required for this study. Lactic acid is estimated by Marbach and Weil method. Principle: L-Lactate + NAD LDH Pyruvate + NADH + H+ Liver enzymes are estimated by Reitmane Frankel method. Cholesterol is estimated by CHOD-PAP method.
STATISTICAL METHODS
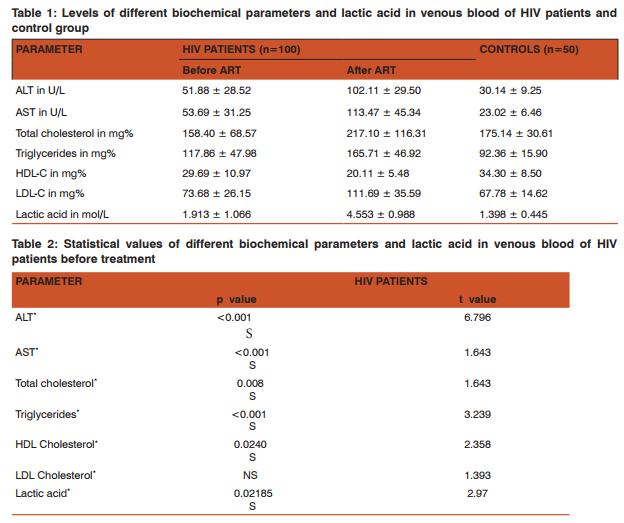
The statistical software used was graphpadquickcalcs. Student t test was used. RESULTS Mean plasma levels of ALT and AST were more (51.88 ± 28.52), (53.69 ± 31.25) than control group (30.14 ± 9.25), (23.02 ± 6.46). These parameters were further increased in HIV infected patients after ART (102.11 ± 29.50), (113.47 ± 45.34). Mean plasma levels of cholesterol were lower (158.40 ± 68.57), than the control group (175.14 ± 30.61) in patients before treatment and it is more than the control group after ART (217.10 ± 116.31). Mean plasma levels of triglycerides were more (117.86 ± 47.98) than the control group (92.36 ± 15.90) before ART and it is further increased after ART (165.71 ± 46.92). Contrarily the mean levels of HDL cholesterol were high in control group (34.30 ± 8.50) than in HIV infected patients (29.69 ± 10.97) and it is further decreased in HIV infected patients (20.11 ± 5.48) after ART. Mean levels of LDL cholesterol were high in HIV infected patients (73.68 ± 26.15) than the control (67.78 ± 14.62) and it is further increased in HIV infected patients after ART (111.69 ± 35.59). Mean levels of lactic acid were high in HIV infected patients (1.913 ± 1.066) than the control (1.398 ± 0.445) and it is further increased in HIV infected patients after ART (4.553 ± 0.987).
DISCUSSION
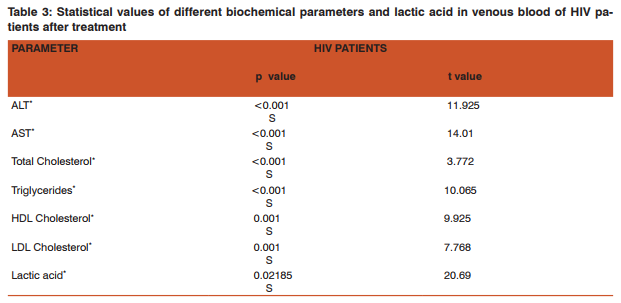
Highly Active Antiretroviral therapy (HAART) has led to decreased morbidity and mortality from HIV infection due to immune reconstitution and viral suppression and increased recognition of both acute and long term toxicities of ART. Hepatitis, lipid abnormalities and lactic acidosis are the common complications seen with HAART. In the present study serum ALT and AST values were significantly elevated in HIV infected patients when compared with controls (p< 0.001). Serum total cholesterol levels, tri-glyceride levels and LDL levels were also significantly elevated when compared with controls (p< 0.001). These results suggest that elevated serum liver enzymes and lipid levels are complications of ART. Similar observations are also made by M.Nunez, R.Lana, J.L.Mendoza, L.Marttin- Carbonero and V. Soriano. 18 The raised serum ALT and AST enzymes are markers of liver injury due to antiretroviral therapy. The total cholesterol and triglycerides which are increased are markers of cardiac risk for the patients receiving antiretroviral therapy in future on long term treatment. The increased cholesterol and triglyceride levels in turn lead to excess formation of pyruvate. This leads to excess production of lactate leading to lactic acidosis. Lactic acid levels are increased significantly (p<0.001) in both groups of patients than control groups and it is further increased in both groups after ART. These results strongly suggest that lactic acidosis is due to ART. Similar observations were also made by Lima A, Van Bommel J, Jansen TC, Ince C, Bakker J.19 Lipid levels have been reported to rise as early as 1 week and stabilize within 3-4 months after initiation of PI therapy.20 Studies by Nerurkas PV, Pearsa l, Frank JE, Yanagihara R, have shown that NRTI and PI are capable of producing lactic acidosis.21 NRTI interfere with life cycle of retrovirus at the point at which RNA is translated into DNA that can be incorporated into genetic blue print of cell. PIs work later in the life cycle of virus preventing release of virions from the cellular machinery that has been subverted into a virus factory. The d4 T might however be responsible for the apparent acceleration of the increase of total cholesterol and triglyceride probably related to the lipodystrophy reported in patients on d4 T treatment and the possible association of HIV-1 infection with decreased triglyceride clearance following food intake.22 Concentration of triglyceride had an effect in the calculated values of LDL and VLDL; however, the smaller increase in levels of LDL-C and triglyceride in NVP regimen than for the EFV might suggest that LDL-C and triglyceride results are valid. Furthermore increased cholesterol has been observed with the use of EFV and NRTIs such as d4 T. A great improvement of atherogenecity indices (total cholesterol/ HDL-C and LDL-C/ HDL-C) on NVP compared to EFV was observed. CONCLUSION By this study, we came to the conclusion that serum lactic acid concentrations are significantly increased in HIV infected patients after ART. Serum lactate levels may be used as prognostic marker in HIV infected patients on ART. Major lipid abnormalities showed that in HIV infected patients, triglycerides, total cholesterol, LDL were increased significantly but HDL was decreased. Also there is significant increase in serum liver enzymes. It is suggested that lipid abnormalities which are major risk factors for heart disease should be monitored regularly by assessment of lipid profile. Lactic acidosis which can be treated successfully should be identified at the earliest so that the patients may not discontinue ART which is useful for them to lead a better life. All HIV infected patients who are starting HAART should have a baseline fasting lipid profile checked, a repeat test in 3-6 months and then a test annually.23Counseling and healthy diet for heart and exercises are also recommended. Use HAART regimen which is not associated with significant increased cholesterol levels.
ACKNOWLEDGEMENTS
Authors acknowledge Dr S Sanjeevi Rao, Professor and Head of department of Biochemistry, Rangaraya Medical College, Kakinada for his technical support. Authors also acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed. Instruments for analysis were provided by Rangaraya Medical College. There are no conflicts of interest.
Abbreviations: HIV – Human Immunodeficiency
Virus ART – Anti Retroviral Therapy
ALT – Alanine Transaminase
AST – Aspartate Transaminase
HDL – High Density Lipoprotein
LDL – Low Density Lipoprotein
VLDL – Very Low Density Lipoprotein
NRTI – Nucleoside Reverse Transcriptase Inhibitors
PI – Protease Inhibitors
NVP – Nevirapine
EFV – Efaverinz
HAART – Highly Active Anti Retroviral
Therapy LDH – Lactate dehydrogenase
References:
1. M. Nunez and V. Soriano, “Hepatotoxicity of antiretrovirals: incidence, mechanisms and Management,” Drug Safety, vol.28, no. 1, pp. 53–66, 2005.
2. Lactic Acidosis and Hepatic Steatosis Associated with Use of Stavudine: Report of Four Cases Kirk D. Miller, MD; Miriam Cameron, MD; Lauren V. Wood et al; Ann Intern Med. 2000;133(3):192-196.
3. M. S. Sulkowski, D. L. Thomas, S. H. Mehta, R. E. Chaisson,and R. D. Moore, “Hepatotoxicity associated with nevirapine or efavirenz-containing antiretroviral therapy: role of hepatitis C and B infections,” Hepatology, vol. 35, no. 1, pp. 182–189,2002.
4. A. R. Lai, K. T. Tashima, and L. E. Taylor, “Antiretroviral medication considerations for individuals coinfected with HIV and hepatitis C virus,” AIDS Patient Care and STDs, vol.20, no. 10, pp. 678–692, 2006.
5. M. Bonacini, “Liver injury during highly active antiretroviral therapy: the effect of hepatitis C coinfection,” Clinical Infectious Diseases, vol. 38, supplement 2, pp. S104–S108, 2004.
6. Stein JH.Dyslipidemia in the era of HIV protease inhibitors. Prog Cardiovasc Dis. 2003 Jan-Feb; 45(4):293-304.
7. Penzak SR, Chuck SK, Stajich GV. Safety and efficacy of HMG CoA Reductase inhibitors for treatment of hyperlipidemia in patients with HIV infection. Pharmacotherapy, 2000; 1066-1071
8. Constans J, Pellegrin JL, Peuchant Eet al (1993) High plasma lipoprotein (a) in HIV –positive patients. Lancet 341; 1099-1100
9. Tsiodras S, Mantzoros C, Hammer S, Samore M. Effects of protease inhibitors on hyperglycemia, hyperlipidemia and lipodystrophy. Arch Intern Med.200; 160:2050-2056
10. Galli, M., Cozzi-Lepri, A., Ridolfo, A. L., Gervasoni, C., Ravasio,L., Corsico, L., et al. (2002). Incidence of adipose tissue alterations in first-line antiretroviral therapy: The LipoICoNaStudy.Archives of Internal Medicine162(22), 2621-2628.
11. Koppel K. Bratt G. Eriksson M. Sandstrom E. Serum lipid levels associated with increased risk for cardiovascular disease is associated with highly active antiretroviral therapy (HAART) in HIV-1 infection.Int J STD AIDS. 2000;11:451– 455. [PubMed]
12. Behrens G, Dejam A, Schmidt H,et al: Impaired glucose tolerance, beta cell function and lipid metabolism in HIV patients under treatment with protease inhibitors. AIDS 1999; 13: F63- F70
13. Sergerer, S and Bogner, JR and Walli, R and Loch, O and Goebel, FD, “Hyperlipidaemia under treatment with protease inhibitors”, Infection, vol. 27, 1999, p.77-81
14. Periard, D and Telenti, A and Sudre, P, “Atherogenic dyslipidemia in HIV-infected individuals treated with protease inhibitors. The Swiss HIV Cohort Study”, Circulation, vol. 100, 1999, p.700-705
15. Friis-Møller N1 , Weber R, Reiss P, et al.Cardiovascular disease risk factors in HIV patients--association with antiretroviral therapy. AIDS. 2003 May 23;17(8):1179-
16. Joly V, Flandre P, Meiffredy V et al. Assessment of lipodystrophy in patients previously exposed to AZT, ddI or ddC, but naive for d4T and protease inhibitors (PI), and randomized between d4T/3TC/ indinavir and AZT/3TC/ indinavir (NOVAVIR Trial). VIII Conference on Retroviruses and Opportunistic Infections. Chicago, IL, February, 2001 [Abstract 539].
17. Brinkman K, Hofstede H.J, Veerkamp M.J, Kolke H.J, Williams J.L and Wesserling P. (1998). Fatal lactic acidosis following HAART containing stavudine (d4T), lamivudine (3TC) and saquinavir. In program and Abstracts of the Twelfth World AIDS Conference, Geneve, 1998.Abstract 60998
18. M. Núñez, R. Lana, J. L. Mendoza, L. Martín-Carbonero, and V. Soriano, “Risk factors for severe hepatic injury after introduction of highly active antiretroviral therapy,” Journal of Acquired Immune Deficiency Syndromes, vol. 27, no. 5, pp. 426–431, 2001.
19. Jansen TC, van Bommel J, Bakker J. Blood lactate monitoring in critically ill patients: a systematic health technology assessment. Crit Care Med. 2009;3(10):2827–2839.
20. Dubé MP, Stein JH, Aberg JA, et al. Guidelines for the Evaluation and Management of Dyslipidemia in Human Immunodeficiency Virus (HIV)–Infected Adults Receiving Antiretroviral Therapy: Recommendations of the HIV Medicine Association of the Infectious Disease Society of America and the Adult AIDS Clinical Trials Group.* Clin Infect Dis. 2003;37:613–627.
21. Nerurkar PV, Pearson L, Frank JE, Yanagihara R, Nerurkar VR. Highly active antiretroviral therapy (HAART)-associated lactic acidosis: in vitro effects of combination of nucleoside analogues and protease inhibitors on mitochondrial function and lactic acid production. Cell Mol Biol. 2003; 49:1205–1211.
22. Miro O, Gomez M, Pedrol E, Cardellach F, Nunes V, Casademont J. Respiratory chain dysfunction associated with multiple mitochondrial deletions in antiretroviral therapyrelated lipodystrophy AIDS:18 August 2000 - Volume 14 - Issue 12 - pp 1855-1857 Research Letters.
23. Shikuma CM, Shiramiju B. Mitochondrial toxicity associated with nucleoside reverse transcriptase inhibitor therapy. Current Inf Dis Reports 2001; 3:553-560.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License