IJCRR - 7(4), February, 2015
Pages: 63-67
Print Article
Download XML Download PDF
EFFECT OF TRIAZOPHOS-AN ORGANOPHOSPHATE INSECTICIDE ON MICROBIAL POPULATION IN PADDY SOILS
Author: B. Lakshmi Kalyani, T. Madhuri, V. Indrani, P. Suvarnalatha Devi
Category: Healthcare
Abstract:Triazophos is a Thiophosphoric and is the active ingradient of the insecticide Hostathion. The aim of the study is to determine the effects of triazophos at five different concentrations on soil bacterial and fungal count. The effect of triazophos on bacterial and fungal population by incubation in soil for 7 days was determined. Results revealed that triazophos caused significant reduction in the bacterial and fungal populations. By statistical analysis, the concentrations of the pesticides as well as the dilution of soil sample were analyzed by two way ANOVA. Study reveals that both bacterial and fungal population the concentration of triazophos vs sample dilution has been significantly affected. The change in concentration brought significant reduction in bacterial count. We conclude that triazophos insecticide is using in paddy soils highly toxic pollutant to the bacterial and fungal organisms.
Keywords: Triazophos, Soil, Bacteria, Fungi, Two- way ANOVA
Full Text:
INTRODUCTION
India is primarily an agriculture based country with more than 60-70 per cent of its population dependent on agriculture. It’s fast growing population is projected to cross 1.3 billion by 2020 (Kanekar et al., 2004).Organophosphates are a group of highly toxic pesticides widely used for increasing agricultural productivity in soil. Organophosphates are also reared as non persistent (Racke and Coats, 1988). The reduced persistence of organophosphate insecticides attributed to soil microorganisms has been described by (Chapman and Harris., 1982; Gorder et al., 1982; Sharmila et al., 1989). Triazophos (TAP, O,O-diethyl-O-1-phenyl-1H-1,2,4-triazol-3-ylphosphorothioate) is one of the broad-spectrum, toxic, nonsystemic contact organophosphorus pesticides (OPs). It has been used on various crops, including rice, cotton, okra and maize to contro pest insects, nematodes and acarids acarids some since the late 1970s (Aungpradit et al., 2007; Lin and Yuan., 2005). Pesticides in soil for longer period is undesirable and it leads to the accumulation of the chemicals in soil to highly toxic levels. In addition, these pesticide residues may be assimilated by the plants and get accumulated in edible plant products such as root crops (Babu et al., 2008). Microorganisms are found in large numbers in soil with bacteria and fungi being the most prevalent. It is stated that usually more than 109 microorganisms are present per gram of soil representing 4000 to 7000 different genomes and biomass of 300 to 3000 kg per hectare (Ranjard and Richaume, 2001).Soil bacsteria play a key role in the global recycling of carbon and other essential elements because of their outstanding range of 106 to 109 bacteria per gram of soil and metabolic activities to exploit many sources of energy and carbon (Alexander, 1977). The influence of these pesticides on soil bacteria is dependent on physical, chemical and biochemical conditions, in addition to the nature and concentration of the pesticides (Aurelia, 2009).Fungi form a large part of the total biomass of micro flora in soil. Microbial biomass in soil is considered to be agents of breakdown of organic matter, when organic materials are applied to soil (Jenkenson and Ladd, 1981). They are usually abundant in the upper layers of the soil, where aerobic conditions prevail. However, numerically much less abundant (between 104 and 106 fungal propagules per gram of soil) than bacteria (Alexander, 1977).The present paper deals with impact assessment of commonly used organophosphate insecticide triazophos (40%EC) on soil bacteria and fungal population dynamics.
MATERIALS AND METHODS
Pesticide
Triazophos was selected for the present investigation in view of it’s wide usage in Srikalahasti paddy cultivation for the control of aphids, thrips, midges, beetles, larvaes, cutworms and other soil insects. It is an organophosphorous insecticide as per the international union of pure and applied chemistry (IUPAC),chemical formula of the compound is diethyl o-(1-phenyl-1h-1,2,4-triazol-3-yl) phosphorothioate. It is also known as nicotinamide, and a common insecticide, was obtained from Sudarshan Chemical Industries Ltd., Pune, India.
Collection of Soil
The soil samples were collected from Srikalahasti, Chittoor district a semi-arid region in Andhra Pradesh, India. Samples of about 1 kg were taken from the first 15 cm of depth and then pooled and sieved. Samples were air dried and stored in polyethylene bags at 4 ?C. The texture of the soil was Black loam-sandy clay with a pH of about 7.6, maximum water holding capacity 0.224 ml/g, sand 50%, silt 20%, clay 29%, organic matter 0.9%.
Soil incubation studies
The effect of different concentrations of triazophos on microbial population was determined in soil sample.
Effect of Triazophos on soil population
Population of bacteria
The effect of different concentrations of triazophos on bacterial population in agricultural soil sample, in triplicate, was determined. Aliquots (0.05 ml) from stock solutions of the pesticides were applied to five-gram portions of soil contained in test tubes (15 X 150mm). The final concentrations (w/w) of each pesticide concentrations 5, 10, 25, 50 and 100 µg/g soil, which are equivalent to 0.5, 1.0, 2.5, 5.0 and 10.0 kg ha-1. The soil samples receiving only distilled water served as controls. According to Shukla and Mishra (1997), Soil samples were then homogenized to distribute the insecticide, and enough distilled water was added to maintain at 60% water holding capacity (WHC) and incubated at room temperature (28 ± 40 C). Seven days after incubation, triplicates of each treatment were withdrawn for estimation of bacterial population. Aliquots were prepared from 10-1 to 10-7 from treated and untreated soil samples by serial dilution plate method on nutrient agar medium and subsequently incubated for 24 hrs in an incubator at 300 C. The experiments were performed in triplicate.
Population of fungi
The effect of different concentrations of triazophos on fungal population(triplicates) in agricultural soil samples was determined, following the same procedure adopted as in the case of bacterial population. Soil plate method was used to asses fungal propagules developing on rose bengal agar medium and subsequently incubated for five days at 25ºC (Shukla and Mishra, 1996).
STATISTICAL ANALYSIS
The data on Triazophos impact on microbial bacterial and fungal populations was interpreted by two way Analysis of variance(ANOVA) and duncan multiple range test (DMR).
RESULTS AND DISCUSSION
Physico-chemical properties of soil
The physico-chemical properties of the experimental soil was shown in (Table 1).
Population of bacteria
The effects of triazophos on the number of bacteria cells per gram of soil was shown in (Table 2) and (Figure1). Results reveals that the reduction in the total bacterial count with increase in the insecticide concentrations when compared with control group. The results of the present study doesn’t agree with many studies used insecticides on soil microbial communities(Stanlake and Clark, 1975; Digrak and Kazanici 2001 ) who notice that the presence of pesticides led to an increase in the total number of soil bacteria, nevertheless, the present study shows that the presence of insecticides led to inhibition in the growth rate of soil bacteria. Bacterial population has been significantly affected by the concentration (F=536.37 ; df 5; p<0.000) of Triazophos and the sample dilution (F=19.700; df 1; p<0.00), the concentration vs sample dilution results (F = 4.979; df 5; p<0.03) as given by ANOVA (Table 3). Treatment of soil with Triazophos brought about a reduction in bacterial population at almost all concentrations (Fig 1). Minimum bacterial count was observed in plate with 100 ppm concentration of triazophos which was significantly very low when compared to control. Short term inhibitory effect on the total bacterial population was observed, which is in accordance with the findings by (Ahmed and Ahmed, 2006; Tawfic et al., 1998 ; Hashem et al., 1999).
Population of fungi
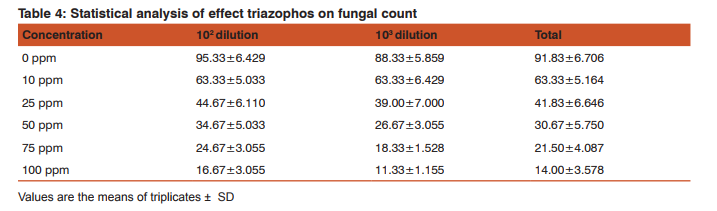
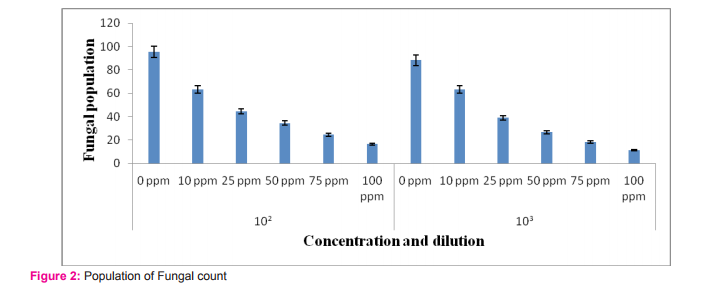
Impact of triazophos on fungi was shown in table 4 and figure 2. The results reveals that decreased in the total fungal count with increasing in the insecticide concentrations compared with control group. Similarly, vapam, captan and nabam at 60 to 400 ppm inhibited fungi in soils (Domsch, 1959;Chandra and Bollen, 1961) demonstrated that soil application of nabam (100 ppm) and mylon (150 ppm) significantly decreased the number of fungal propagules. Fungal population has been significantly affected by the concentration (F=214.25 ; df 5; p<0.000) of Triazophos and the sample dilution (F=10.97; df 1; p<0.003), the concentration vs sample dilution results (F = 0.497; df 5; p<0.775) as given by ANOVA (Table 5).
CONCLUSION
Our research showed that natural growth of beneficial soil bacterial and fungal species is affected by Triazophos when applied in agricultural farm. The obtained results indicated that triazophos used had adverse effect on bacterial and fungal population. We conclude that triazophos is highly toxic to the soil microbes.
ACKNOWLEDGEMENT
Authors acknowledge the immense help received from the scholars whose articles are cited and included in reference of this manuscript. The authors are also grateful to authors/ editors/ publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed. Authors are highly grateful to the Department of Applied Microbiology, DST-FIST, CURIE, Sri Padmavathi Mahila Visavidyalayam, Tirupati, Andhra Pradesh, India for providing Research facilities.


References:
1. Ahmed S and Ahmed M S. Effect of Insecticides on the total number of soil bacteria under laboratory 2006.
2. Alexander M. Introduction to Soil Microbiology. John Wiley and Sons, Inc., New York.and field conditions. Pakistan Entamologist 1977; 28(2):63-67.
3. Anderson J F. Energy content of spider eggs . Oecologia 1978; 37 :41-57.
4. Aungpradit T, Sutthivaiyakit P, Martens D, Sutthivaiyakit S and Kettrup A A F. Photocatalytic degradation of triazophos in aqueous titanium dioxide suspension: identification of intermediates and degradation pathways. Journal of Hazardous Materials 2007;146(1):204–13.
5. Babu J N, Bhalla V, Kumar M, Mahajan R K and Puri R K. A chloride selective sensor based on a calyx arene possessing a urea moiety. Tetrahedron Letters 0040- 4039. 2008 ;49: (17) 2772-2775.
6. Chandra P and Bollen W B. Effect of nabam and mylon on nitrification, soil respiration and microbial numbers in four Oregon soils. Soil Science 1961; 92:387.
7. Chapman R A and Harris C R. Persistence of isofenphos and isazophos in a mineral and an organic soil. Journal of Environmental Science and Health section B 1982;17: 355-361.
8. Digrak M and Kazanici F. Effect of some organophosphorus insecticides on soil microorganisms. Turkey Journal of Biology 2001; 25: 51-58.
9. Domsch K H. The effects of soil fungicides. III. Quantitative changes in soil microflora. Z.Planzendau Pflanzenschutz 1959; 66:17.
10. Gorder G W, Dahm P A and Tollefson J J. Carbofuran persistence in cornfield soils. Journal of Economic Entomology 1982; 75: 637-642.
11. Hashem F, Hafes H and El-Mohandes M A O. Isolation and identification of pyrethroid insecticides degrading bacteria from soil. Ann. Agric. Sci. Cairo., 1999; 44(1): 123-137.
12. Jenkinson D S and Ladd J N. Microbial biomass in soil: Measurement and turnover. In: Paul EA, Ladd JN (eds) Soil Biochemistry, vol 5. Marcel Dekker, New York, Basel 1981 ;415-471.
13. Kanekar P P, Bhadbhade B, Deshpande N M and Sarnaik S S. X Biodegradation of organophosphorus pesticides, IN : Proceedings of Indian National Science Academy 2004;70:57- 70.
14. Lin K D, Yuan D X. Degradation kinetics and products of triazophos intertidal sediment. Jounal of Environmental Sciences 2005;17(6):933–6.
15. Racke K D and Coats J R. Comparative degradation of organophosphorus insecticides in soil : Specificity of enhanced microbial degradation. Journal of Agriculture and Food Chemistry 1988; 36: 193- 199.
16. Ranjard L and Richaume A S. Quantitative and qualitative microscale distribution of bacteria in soil. Research in Microbiology 2001;152: 707-716.
17. Sharmila M, Ramanand K and Sethunathan N. Effect of yeast extract on the degradation of organophosphorus insecticides by soil enrichment and bacterial cultures. Canadian Journal of Microbiology 1989; 35: 1105-1110.
18. Shetty P K and Magu S P. Effect of metalaxyl on soil microbial population. Journal of Tropical Agriculture 2000 ; 38: 63-65.
19. Shukla P K and Mishra R R. Response of microbial population and enzyme activities to fungicides in potato field soil. Proceedings of Indian National Science Academy section B 1996; 62 (5): 435-438.
20. Stanlake G J and Clark J B. Effects of a commercial malathion preparation on Selected Soil Bacteria. Applied Microbiology 1975; 30(2) :335-336.
21. Tawfic M A, Ismail S M M and Mabrouk S S. Residues of some chlorinated hydrocarbon pesticides in rainwater, soil and ground water and their influence on some soil microorganisms. Environmental International 1998 ; 24(5-6): 665-670.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License