IJCRR - 7(7), April, 2015
Pages: 22-27
Print Article
Download XML Download PDF
AUDITORY BRAINSTEM PROCESSING OF COMPLEX SPEECH SOUNDS IN HIGH RISK INFANTS-A PRELIMINARY STUDY
Author: Muhammed Ayas, Hakam Yaseen, Rajashekhar B.
Category: Healthcare
Abstract:Background: The ability of the auditory brainstem process for complex speech sound is precursor for the normal language development. Infants who are born prematurely are at high risk for hearing loss and should be evaluated for their hearing at the earliest to identify the hearing related disorders. Objectives: The aim of the study was to explore how an immature auditory brainstem responds for the complex acoustic stimuli such as speech sounds. Method: Twelve high risk infants were recruited in the prospective, observational study. Speech Evoked Auditory Brainstem Response (SEABR) was recorded in all the high risk infants using stop consonant/da/. Results: The results of the study were promising in such a way that the onset and sustained responses were encoded in the premature auditory brainstem with greater fidelity. Conclusion: The scalp-recorded SEABR offers a unique window in understanding how the human auditory brainstem represents key elements of the speech signal. From the current study, we draw the inference that all the high risk infants should undergo a SEABR recording and such findings would enable the clinicians and researchers to dwell in to the possible onset of APD in these high risk populations.
Keywords: Speech evoked auditory brainstem response (SEABR), High risk infants (HR infants), Frequency following response (FFR), Auditory processing disorders (APD), Language development
Full Text:
INTRODUCTION
The ability of the auditory system to precisely encode the speech sound is vital for the development of speech and language in infants and children. Such an auditory stimulation plays a major role in the maturation of the auditory cortex and makes the auditory areas capable for the intake of acoustic stimulation (Kral et al, 2005, Ponton et al 2002). In newborns and infants, though the central auditory structures are not developed completely yet, continuous acoustic stimulation is important for the synaptic pruning and proliferation, which results in the better development of auditory areas in the brain (Kuhl, 2004, Keuroghlian, and Knudsen ,2007). Due to the widespread application of Universal Newborn hearing screening program (UNHS) program (Shulman et al,2010, Patel and Feldman,2011 , Haves,1999, Wiechbold, Hues and Muller,2006,Gorga et al 2001,Kerchner et al ,2004), today, almost all hospitals are equipped to screen newborns for their hearing. These screening processes have paved a path for the early identification of hearing loss and its intervention (Ching et al 2013,Ptok and Med, 2011, Paludetti et al 2012, Spiyak and Sokol, 2005, Buttros, Vohr et al 2008, Kenna,2004,Kasal et al 2012, Christensen, Thomson, Letson, 2008).However, certain sections of researchers have also pitched their criticism on the growing number of false positive and false negative responses obtained in UNHS (Davis et al 1997,Colgen et al 2012,Nelsen et al 2008, Clemens and Davis 2001). This could be due to the technical failures or procedural flaws during the hearing screening program (Ciorba et al 2007,Gorga et al 2001,Mencher et al 2001). By keeping in mind that the auditory stimulation is vital for the speech and language development in children (Sharp and Hellenbrand, 2008, Tomblin et al 2014, Laws and Hall 2014), it is important to study, how such an auditory processing is categorically processed in these infants. There is a dearth of literature on how speech sounds are processed in the central auditory system of very young infants. Such a research question is imminent as there is a growing population of children who are categorized into poor readers or performers at school level due to apparent breakdown in the central auditory processing of speech signals, though these children has absolutely normal hearing when assessed with pure tones (Ahmmed et al 2014, Allen and Allan, 2014).The possible causes for these APD have paved a ground breaking research in Paediatric audiology, which insists on thorough auditory processing evaluation in children or may be a continuous monitoring at the infant stage itself. The lack of scientific evidence in literature has also been a key reason not to authenticate when and how to proceed for this type of hearing assessment. The basic question arises in the wakening of growing APD is that, can the auditory processing abilities be probed in the infant stage itself, especially in high risk infants. It is clearly understood that the premature babies are at high risk for developing hearing loss or may develop ANSD due to their increased medical complications (Cristobal and Okhalai,2008, Borrdori et al 1997,Marlow and Hunt,2000).These hearing difficulties will have detrimental effect on normal language development. Till date to our knowledge, there are very few reports on how the speech sounds are processed through auditory brainstem in infants, especially high risk infants. A chunk of studies has been done using non speech sounds like pure tones or tone bursts (Ptok et al 2005, Ahmmed et al 2014).This information is limited to comment on the brainstem processing for speech sounds. Thus it is important to probe into how the brainstem process speech sounds in high risk infants. One of the popular methods to objectively assess the brainstem processing of speech is using speech sounds integrated with ABR (Skoe and Kraus, 2010) Speech evoked Auditory Brainstem Response BR (SEABR) has gained its popularity by using it in various populations such as children, musicians and adults for various aims(Tierney and Kraus,2013,song et al,2011,Anderson et al 2010).SEABR can be defined as the evoked potential recorded in response to a speech sounds from the auditory brainstem in time locked manner.SEBAR has been in the research ground more than a decade by probing the auditory processing abilities in various populations especially using the stimuli stop consonant /da/ (Ruso et al 2004).It is widely believed that such a complex speech sounds can offer an enormous wealth of information on auditory processing in humans (Johnson et al 2005, 2007). Hence, in the current study, the basic aim was to study how the premature auditory brainstem processes the speech sounds and its features such as onset responses, formant transition and frequency following response, which will shed light into the complex auditory processing mechanism in infants.
MATERIALS AND METHODS
a. Participants
The prospective, observational study were carried out in a tertiary care hospital in India, within a specially created sound treated room in the Neonatal intensive care unit (NICU).Twelve high risk infants were included in the study. All the high risk infants were recruited based on the inclusion criteria such as, all the infants should be born before 36 weeks of gestational age during the study period, low birth weight (less than 1500g), hyperbilirubinemia requiring phototherapy. Infants with Congenital malformations and perinatal asphyxia were excluded from the study infants The current study was approved by the institutional research and ethics committee (IREC).Parents were informed about the potential importance and scientific benefit of reporting the data and consensus obtained before proceeding for the study. A duly signed consent form from all the parents was obtained before the SEABR recording.
b. Stimulus and recording parameters
SEABR were elicited by using a stop consonant /da/(Russo et al 2004).The speech stimulus is 40-ms synthesized /da/ which has an initial noise burst and formant transition between the consonant and a steady-state vowel, and was synthesized with a fundamental frequency (F0) that linearly rises from 103 to 125 Hz with voicing beginning at 5 ms and an onset noise burst during the first 10 ms. The first formant (F1) rose from 220 to 720 Hz while the second and third formants (F2 and F3) decreased from 1700 to 1240 Hz and 2580 to 2500 Hz, respectively, over the duration of the stimulus (Russo et al, 2004). The fourth and fifth formants (F4 and F5) were constant at 3600 and 4500 Hz, respectively. The stimuli was presented at repetition rate of 7.1/s. Electrophysiological responses were collected with Ag – Ag Cl electrodes, and recorded from Cz (active)-to right earlobe (reference), with the left earlobe as ground by using Intelligent Hearing System (IHS) version 4.0.3. Electrode impedance were less than 5 kOhms for each electrode and the difference between the electrodes was less than 3KOhms.The stimuli were presented to the right ear through insert earphones (ER-3A) at an intensity of 60 dB SPL while the left ear was unoccluded. 2000 alternating sweeps were collected to extract the final waveform. The analysis time window was kept 60 ms while recording the data.
d. Data Analysis
The brainstem response to a complex speech sound includes transient peaks that reflect the encoding of rapid temporal changes which were compared by means of latencies and amplitudes, as well as sustained elements that comprise the frequency following response (FFR), that encodes the harmonic and periodic sound structure of vowels, The characteristic response to the speech stimulus /da/ includes a positive peak (wave V), likely analogous to the wave V elicited by click stimuli, followed immediately by a negative trough (wave A). Following the onset response, peaks C to F are present in the FFR period and the offset (wave O) indicates the cessation of the stimulus. The latencies of the waveform were identified by two trained audiologists.
e) Statistical Analysis
All the data were entered in a SPSS-version 14 system. The mean and standard deviation of all the 7 peak latencies were obtained.
RESULTS
The result of the study has been categorized into onset and sustained responses (FFR).
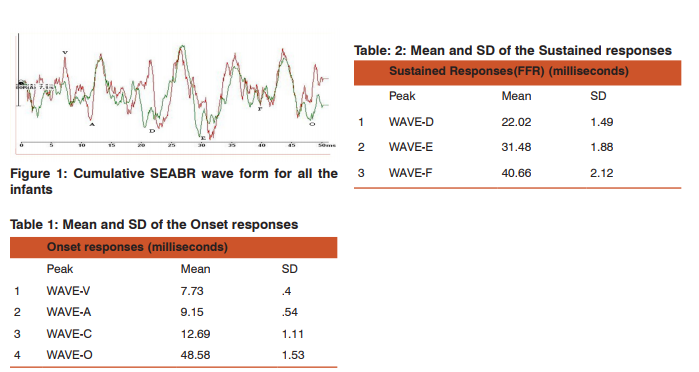
A) Onset response: The onset responses were highly replicable both within and across subjects (figure 1). Peaks V, A, and C were detectable in all the infants. However,3 infants, peak c were not been recorded. The onset response waves V and A were largest in magnitude and followed by peak C. The mean and standard deviation obtained for wave V is 7.73ms (SD-.40) and for wave A is 9.15ms (SD-.54). For Wave C the mean values were 12.69ms (SD-1.11). There was a slight variation seen in the wave C across the infants. The reason for this will be discussed later. In the offset measures, Wave O were obtained with a mean value of 48.58ms (SD-1.51). General variance is noted in offset responses as well. (table-1)
B) Sustained Response (FFR): The FFR was evident in all infants (figure-1). The FFR is the phase locked Reponses obtained between in 14ms to 40ms. The wave D-E-F were prominently recorded with a mean value of 22ms-31ms-40ms respectively with a standard deviation of 1.49-1.89- 2.12.The slight variation in the peak responses could be due to the infant related factors during the recording time.(table-2).
DISCUSSION
The auditory brainstems ability to respond for complex speech sound is precursor for the normal language development. The SEABR faithfully reflects many acoustic properties of the speech signal. These responses provides a mechanism for understanding the neural bases of normal auditory brainstem function, by providing a quantifiable tool to asses an infant’s attention-independent neural encoding of speech sounds. The aim of the current study was to explore the auditory brainstem encoding of speech sounds in premature infants. The results of the current study are promising in such a way that the complex speech sounds are able to record in such versatile population. The onset and sustained responses-FFR were recorded with good precision with good amplitude and it was replicated in all infants with good fidelity. The current study will explore on how the onset and sustained responses were encoded in High Risk (HR) infants. The onset response (transient) measures included latency and amplitudes of peaks V, A, C, and O, slope, area and amplitude of the VA complex as a unit. The onset responses of the stop consonant /da/ were recorded in all HR infants. The wave V to A slope were seen as similar that of the responses recorded in children in other studies (Anderson et al 2010, Johnson at all 2007 Russo et al 2004).However in few recordings, wave V-A were prolonged, which could be attributed to the neural conduction delay. In contrast, evidence from the literature states that due to the maturational process, the latency of wave v can be negligibly prolonged in infants (Thaivan et al 2007, Ponton, Moore and Eggermont 1996). It is important to note that these responses could well give an important information on how well a developmental auditory system respond to a burst portion of a complex speech sounds such as stop consonants. It is to be noted that in 3 infants the peak C was not prominent compare to others. Peak C depicts the formant transition feature of the complex stimuli /da/. The acoustic features of this information would give an insight into how the complex signals are routed through the auditory brainstem to the auditory cortex. Studies have reported that the disruption in formant transition of the signals is evidently seen in children who are diagnosed with APD (Moore 2011, Allen and b Allen 2014, Banai and Kraus, 2006). The offset responses peak O is also an important component of SEABR as it marks the cessation of voicing of the stimulus during the recording. The FFR is synchronous to the sound, with each cycle faithfully representing the temporal structure of the sound. Thus the FFR reflects the neural phase-locking response with an upper limit of about 1000 Hz (Russo et al 2004) In the current study, FFR were recorded in all the infants. The periodicities of the stimulus were faithfully encoded in the immature brainstem revealing a possible inherent mechanism in infants to extract the speech sounds especially fundamental frequency. The fidelity of these responses opens up a debate on an old school of thought on how an infant identifies mother’s voice from that of others. The fundamental frequency, which is defined as the lowest lowest frequency in the signal is important for the speaker identification and differentiation of high and low pitch sounds. Results from the current study strengthen the notion that the ability of the auditory brainstem to identify the speech sounds could have been inherently developed in infants in early days of life. This offers a path breaking evidence for a further investigation on how the FFR responses changes during the developmental stages in life. Evidence from the literatures suggests that during the developmental course the auditory system undergoes vigorous synaptic pruning and myelination (Moore and Linthicum 2001, Moreno et al 2009). The FFR finding from the study points out atypical mechanism that process the complex speech sounds with great fidelity. However it is premature to comment such a mechanism functions in the immature auditory system. Also the scalp-recorded FFR probably reflect multiple sources in the immature brainstem (LL, CN, IC) (Chandrashekaran et al 2012). SEABR being a non invasive method to examine the subcortical encoding of speech features helps one to probe more closely the representation of speech features. The findings of the study also suggest that the close proximity of FFR responses in infants indicate that the Fo and harmonic structure of vowels are well developed and encoded with precision. There are reports stating that disruption in FFR could be seen in children with APD ( Banai et al 2007, Russo et al 2009,Hornicket et al 2012). By taking all these evidence into account, from the current study we can state that the infants who are at risk for hearing loss showed a satisfactory evidence of brainstem processing for complex acoustic stimuli such as speech stimuli. However, such inference cannot be drawn from the non speech stimulus recording using clicks or tone bursts. This underlies the importance of using speech stimuli for electrophysiological research and a paradigm shift from non speech stimuli to speech stimuli research in young children coming years.
CONCLUSION
The scalp-recorded SEABR offers a unique window in understanding how the human brainstem represents key elements of the speech signal. From the current study we draw the inference that all the high risk infants should undergo a SEABR recording in order to rule out APD in the early stage itself. Such recordings would enable the clinicians and researchers to dwell in to the possible onset of APD in these populations. Though the findings from the current study cannot be generalized in to the whole population owing to the limited data, by widening the potential importance of the study with more data these findings can be replicated in to clinical settings for hearing evaluation in infants and children.
Acknowledgements
Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed. The authors also would like to thank all the babies and parents for taking part in the study.
References:
1. Ahmmed, AU., Ahmmed, AA., Bath, JR., Ferguson, MA., Plack, CJ.and Moore, DR.(2014). Assessment of children with suspected auditory processing disorder: a factor analysis study. Ear Hear, 35(3), 295-305.
2. Allen, P and Allan, C. (2014). Auditory processing disorders: relationship to cognitive processes and underlying auditory neural integrity. Int J Pediatr Otorhinolaryngol, 78(2), 198- 208.
3. Anderson S, Skoe E, Chandrasekaran B, Zecker S, Kraus N. (2010) Brainstem Correlates of Speech-in-Noise Perception in Children. Hearing Research. 270:151-157
. 4. Banai K, Kraus N. (2006) Neurobiology of (central) auditory processing disorder and language-based learning disability. In: Handbook of Central Auditory Processing Disorder: Volume I: Auditory Neuroscience and Diagnosis,Chermak GD, Musiek FE. (eds) San Diego: Plural Publishing Inc. 89- 116.
5. Banai K, Abrams D, Kraus N. (2007) Speech evoked brainstem responses and sensory-based accounts of learning disability. International Journal of Audiology 46: 524-532.
6. Buttross, SL., Gearhart, JG. and Peck, JE. (1995). Early identification and management of hearing impairment Am Fam Physician, 51(6), 1437-46, 1451-2.
7. Borradori, C., Fawer, CL., Buclin, T. and Calame, A. (1997). Risk factors of sensorineural hearing loss in preterm infants. Biol Neonate, 71(1), 1-10.
8. Chandrasekaran B, Kraus N. (2012). Biological factors contributing to reading ability: subcortical auditory function. In: Developmental Dyslexia: Early Precursors, Neurobehavioral Markers and Biological Substrates. April A. Benasich and R. Holly Fitch (eds.) 83-98.
9. Cristobal, R. and Oghalai, JS. (2008). Hearing loss in children with very low birth weight: current review of epidemiology and pathophysiology. Arch Dis Child Fetal Neonatal Ed, 93(6), F462-8.
10. Christensen, M., Thomson, V. and Letson, GW. (2008). Evaluating the reach of universal new-born hearing screening in Colorado.Health Technol Assess, Am J Prev Med, 35(6), 594-7.
11. Colgan , S., Gold, L., Wirth, K., Ching, T., Poulakis, Z., Rickards, F.and Wake, M.(2012). The cost-effectiveness of universal newborn screening for bilateral permanent congenital hearing impairment: systematic review. Acad Pediatr, 12(3), 171-80.
12. Clemens, CJ. and Davis, SA. (2001). Minimizing false-positives in universal newborn hearing screening: a simple solution. Pediatrics.;107(3):E29.
13. Ching, T.Y.C., Day, J., Seeto, M., Dillon, H., Marnane, V., and Street, L.(2013). Predicting 3-year outcomes of earlyidentified children with hearing impairment. B-ENT.Suppl 21: 99–106.
14. Ciorba, A., Hatzopoulos, S., Camurri, L., Negossi, L., Rossi, M., Cosso, D., Petruccelli, J., and Martini, A.(2007). Neonatal newborn hearing screening: four years’ experience at Ferrara University Hospital (CHEAP Project): Part 1. Acta Otorhinolaryngol Ital, 27(1), 10–16.
15. Davis, A., Bamford, J., Wilson, I., Ramkalawan, T., Forshaw, M. and Wright, S. (1997). A critical review of the role of neonatal hearing screening in the detection of congenital hearing impairment. Health Technol Assess.1(10)1-176.
16. Gorga, MP., Preissler, K., Simmons, J., Walker, L.and Hoover, B. (2001). Some issues relevant to establishing a universal newborn hearing screening program. J Am Acad Audiol, 12(2), 101-12.
17. Johnson KL, Nicol T, Kraus N. (2005) The brainstem response to speech: a biological marker. Ear and Hearing 26(5): 424-433.
18. Johnson K, Nicol T, Zecker S, Kraus N. (2007) Auditory brainstem correlates of perceptual timing deficits. Journal of Cognitive Neuroscience 19: 376-385.
19. Hayes,D. (1999).State programs for universal new born hearing screening. Pediatric Clinic North America, 46(1), 89-94.
20. Hornickel J, Kraus N. (2012) Breaking news: cABR can predict auditory-based communication skills. Hearing Journal. 65(9): 28-30.
21. Kenna, MA. (2004). Medical management of childhood hearing loss. Pediatric Annals. 2004;33(12):822-32.
22. Kasai, N., Fukushima, K., Omori, K., Sugaya, A.and Ojima, T.(2012). Effects of early identification and intervention on language development in Japanese children with prelingual severe to profound hearing impairment. Ann Otol Rhinol Laryngol Suppl, 202, 16-20.
23. Kerschner, JE., Meurer, JR., Conway, AE., Fleischfresser, S., Cowell, MH., Seeliger, E.and George, V.(2004). Voluntary progress towards universal newborn hearing screening. International Journal of Pediatric Otorhinolaryngology.;68(2):165-74.
24. Kral, A., Tillein, J., Heid, S., Hartmann, R.and Klinke, R. (2005). Postnatal Cortical development in Congenital Auditory Deprivation. Cerebral cortex, 15 (5), 552-565.
25. Kuhl, P. K. (2004). Early language acquisition: Cracking the speech code. Nature Reviews Neuroscience, 5, 831–843.
26. Keuroghlian, A. S., and Knudsen, E. I. (2007). Adaptive auditory plasticity in developing and adult animals. Progress in Neurobiology, 82,109–121.
27. Laws, G. and Hall, A. (2014). Early hearing loss and language abilities in children with Down syndrome. Int J Lang Commun Disord, 49(3), 333-42.
28. Marlow, E., Hunt, L., and Marlow, N. (2000). Sensorineural hearing loss and prematurity. Arch Dis Child Fetal Neonatal Ed, 82(2), F141–F144.
29. Mencher, GT., Davis, AC., DeVoe, SJ., Beresford, D.and Bamford, JM.(2001). Universal neonatal hearing screening: past, present, and future. Am J Audiol.;10(1):3-12.
30. Moore JK, Linthicum FH Jr. 2007,. The human auditory system: a timeline of development. International Journal of Audiology 46 460–478.
31. Moreno S, Marques C,Santos A,Santos M,Castro SL,Besson M (2009). Musical training influences linguistic abilities in 8-year-old children: more evidence for brain plasticity. Cerebral Cortex 19: 712–723.
32. Nelson, HD., Bougatsos, C. and Nygren, P. (2008). Universal newborn hearing screening: systematic review to update the 2001 US Preventive Services Task Force Recommendation. Pediatrics,122(1),e266-76.
33. Ponton, C., Eggermont, J.J., Khosla,D., Kwong, B.and Don, M.(2002). Maturation of human central auditory system activity: separating auditory evoked potentials by dipole source modelling. Clinical Neurophysiology.;113(3):407-20.
34. Ponton CW, Moore JK, Eggermont JJ (1996). ABR generation by parallel pathways: Differential maturation of axonal conduction time and synaptic transmission. Ear Hear. 17: 402–410.
35. Patel, H., Feldman, M. (2011). Universal new-born hearing screening. Pediatric Child Health.; 16(5): 301–305.
36. Ptok M, Med.(2011) Early Detection of Hearing Impairment in Newborns and Infants. Dtsch Arztebl Int.; 108(25): 426–431.
37. Ptok, M., Buller, N., Kuske, S. and Hecker, H. (2005).Subjective assessment of auditory processing deficits and perceptual performance in children. HNO, 53(6),568-72.
38. Paludetti, G., Conti, G., Di Nardo, W., De Corso, E., Rolesi, R., Picciotti, P.M. and Fetoni A.R.(2012). Infant hearing loss: from diagnosis to therapy Official Report of XXI Conference of Italian Society of Pediatric Otorhinolaryngology. Acta Otorhinolaryngol Ital. 32(6): 347–370.
39. Russo N, Nicol T, Musacchia G, Kraus, N. (2004) Brainstem responses to speech syllables. Clinical Neurophysiology 115: 2021-2030.
40. Russo NM, Zecker S, Trommer B, Chen J, Kraus N. (2009) Effects of background noise on cortical encoding of speech in autism spectrum disorders. Journal of Autism and Developmental Disorders 39: 1185-1196.
41. Shulman, S., Besculides, M., Saltzman, A., Ireys, H., White, KR., Forsman, I.(2010). Evaluation of the universal newborn hearing screening and intervention program. Paediatrics;126(1)19-27.
42. Spivak, L. and Sokol, H. (2005). Beyond newborn screening: early diagnosis and management of hearing loss in infants. Adv Neonatal Care, 5(2),104-12.
43. Sharp, HM. and Hillenbrand,K.(2008). Speech and language development and disorders in children. Pediatr Clin North Am,55(5),1159-73.
44. Skoe E, Kraus N. (2010) Auditory brainstem response to complex sounds: a tutorial. Ear and Hearing 31(3): 302- 324.
45. Song J, Nicol T, Kraus N. (2011) Test-Retest Reliability of the Speech-Evoked Auditory Brainstem Response. Clinical Neurophysiology. 122, 346-355.
46. Thai-Van H, Cozma S, Boutitie F, Disant F, Truy E, Collet L. (2007). The pattern of auditory brainstem response wave V maturation in cochlear-implanted children. Clinical Neurophysiology.118(3):676-89.
47. Tomblin, JB., Oleson, JJ., Ambrose ,SE., Walker, E ., and Moeller, MP.. (2014). The Influence of Hearing Aids on the Speech and Language Development of Children with Hearing Loss. JAMA Otolaryngol Head Neck Surg.;140(5):403- 9
48. Tierney A, Kraus N. (2013) The ability to move to a beat is linked to the consistency of neural responses to sound. Journal of Neuroscience. 33(38):14981–14988.
49. Vohr, B., Jodoin-Krauzyk, J., Tucker, R., Johnson, MJ., Topol, D.and Ahlgren, M.(2008). Early language outcomes of early identified infants with permanent hearing loss at 12 to 16 months of age. Pediatrics, 122(3), 535-44.
50. Weichbold, V., Heis, D. and Mueller, K. (2006). Universal newborn hearing screening and postnatal hearing loss,117(4),631-636.
51. Wesson, B., Vohr, BR., Sininger, YS., Widen, JE., Folsom, RC., Gorga, MP. and Norton ,SJ. (2000). Identification of neonatal hearing impairment: infants with hearing loss. Ear hear, 21(5), 488-507.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License