IJCRR - 7(11), June, 2015
Pages: 20-25
Print Article
Download XML Download PDF
Antifungal Susceptibility of Candida against Six Antifungal Drugs by Disk Diffusion Method Isolated From Vulvovaginal Candidiasis
Author: Twinkle N. Gandhi, Manish G. Patel, Mannu R. Jain
Category: Healthcare
Abstract:Introduction: Vulvovaginal candidiasis is the second most common cause of vaginitis after bacterial vaginosis and is diagnosed in 40 % women with vaginal discharge. Incidence of fungal infections has increased dramatically over the past few decades. Epidemiologic data from the past decade reveal a paradigm shift in candida species from albicans to non -albicans candida species. The aim of this study was to determine incidence of vulvovaginal candidiasis, identify the species and test their in vitro susceptibility
profile against six antifungal agents \? Fluconazole, Itraconazole, Ketoconazole, Clotrimazole, Amphotericin B and Nystatin. Material and Methods: Two high vaginal swabs were collected from patient with complain of vaginal discharge. One sample is processed for direct microscopic examination and other sample is used for culture on Sabourad's dextrose agar and Blood agar. Isolates identified by battery of test and antifungal susceptibility testing of Candida species done by Disk Diffusion method. Results: A total of 122 isolates of candida species were obtained. Candida albicans was found to be the most frequently isolated species 81(66.39%) of the total isolates, followed by C. glabrata 19 (15.65%),C. tropicalis 12(33.61%), C. parapsilosis 6(4.91%), C. krusei 3(2.4%) and C. gullermondii 1(0.8%).Non-albicans candida constituted 41(33.61%).Antifungal susceptibility pattern showed that candida isolates were more sensitive to Nystatin and Amphotericin \?B, compared to that of Clotrimazole, Fluconazole and Ketoconazole. 78% C. albicans were sensitive to Fluconazole, 5% were dose dependent susceptible, and 17% were resistant. C. krusei had shown 33% resistance against fluconazole and67% resistance against Clotrimazole. Conclusion: The majority of C. albicans isolates were susceptible to Fluconazole. C. krusei had shown maximum resistance among all the isolates. Identification of Candida to species level and their antifungal susceptibility testing should be done to achieve better clinical results.
Keywords: Vulvovaginal candidiasis (VVC), Antifungal susceptibility testing, Disk diffusion method
Full Text:
INTRODUCTION
Vulvovaginal candidiasis (VVC) is a common fungalinfection that affects healthy women of all ages.1 At least75% of women will develop one or more infections once during their lifetime, with 5 to 8% of those individuals developing recurrent infections.1,11Symptoms generally include itching, burning, soreness and abnormal vaginal discharge.4 The principal agent of VVC is Candida albicans, but other species known generally as C. non-albicans (C. glabrata, C. tropicalis, C. krusei, C. parapsilosis, C.gullermondii) are also isolated.19C. glabrata is the second most common yeast, and its treatment is considered a serious clinical challenge. 2 Candida albicans and non-albicans species are closely related but differ from each other with respect to epidemiology, virulence characteristics, and fungal susceptibility, therefore Candida species identification is important for successful management.3 Prolonged therapy and increased use of antifungal for recurrent candidiasis are the most common risk factors for azoles resistance among Candida isolates from vulvovaginitis candidiasis patients.11Azoles have the advantage of being taken orally, which increase their potency. 5 The emergence of drug resistant strains, both chromosomal and plasmid borne, reinforce the need for the study of these pathogens and the surveillance of its susceptibility to drugs commonly used for therapy.2,9Close monitoring of the antimicrobial susceptibility and its resistance mechanism is essential in an environment of rapidly changing resistance patterns.4 Methods for evaluating the susceptibility of yeasts to antifungal agents have been the subject of numerous studies during the last decade.15,16A standard reference procedure has been described by the National Committee for Clinical Laboratory Standards (NCCLS).13 The reference procedure is a macrotube dilution technique which is too cumbersome for use in most clinical laboratories.2,15 A broth microdilution adaptation of that procedure has been found to be acceptable.13,16Although microdilution tests can usually be read after 24 h, 48-h readings are specified for the NCCLS macrotube test.14,15The E test is a proprietary test that has also been found to be capable of giving reliable results that can often be read after 24 h of incubation.2,16 For use in a clinical laboratory, a simplified disk diffusion test has some important advantages.6,8,18For practical reasons, that disk technique should be similar to the disk procedure that is being used to test antibacterial agents.8,12,10 Objectives of this study were to isolate Candida from VVC patients, characterization and in vitro antifungal susceptibility against six antifungal drugs by the Disk Diffusion method.
MATERIAL AND METHODS
This study was conducted during July 2010 to October 2011 in the Department of Microbiology, SMIMER medical college Surat, Gujarat, India.A total number of 410 OPD patients with complains of vaginal discharge attending department of Obstetrics and Gynaecology are included in the study.Two swabs were collected from each patient with the help of sterile cotton swabs. Specimen was collected from the vagina or cervix avoiding the contamination of other organism. Out of two swabs, one was subjected to direct smear examination, and the other was inoculated on Sabouraud’s dextrose agar (SDA) and Blood agar and incubated at 37?C aerobically. The growth of Candidaon SDA was confirmed based on colony morphology and gram stain examination. After growth species of Candida were identified. Species identification- Species identification of Candida isolates was done following standard mycological protocol including germ tube test, fermentation and assimilation of various sugars, chlamydospores production on Corn meal agar and colony color on Hi Chrome Candida agar. Antifungal susceptibility testing- Antifungal susceptibility testing of the isolates was performed by six antifungal drugs disk Fluconazole (25 mcg), Ketoconazole (10 mcg), Itraconazole (10 mcg), Clotrimazole (10 mcg), Nystatin(100 U) and Amphotericin-B (100 U). Antifungal susceptibility testing was performed by disk diffusion method using Mueller-Hinton Agar + 2% Glucose and 0.5 μg/ml Methylene Blue Dye (GMB) Medium as per CLSI guidelines (C.L.S.I. document M44-A2, 2009.). 0.5 McFarland standard was used to standardize the inoculums density. C.albicans ATCC 90028 and C. parapsilosis ATCC 22019 were used as quality control. All the culture media, Antifungal disk, and control strains were obtained from Himedia Laboratories, India. The antifungal susceptibility of the isolates was interpreted as sensitive (S), dose dependent-susceptible (DDS) and resistant (R). The results were interpreted as per the Clinical and Laboratory Standards Institute (CLSI) guidelines.
RESULTS AND ANALYSIS
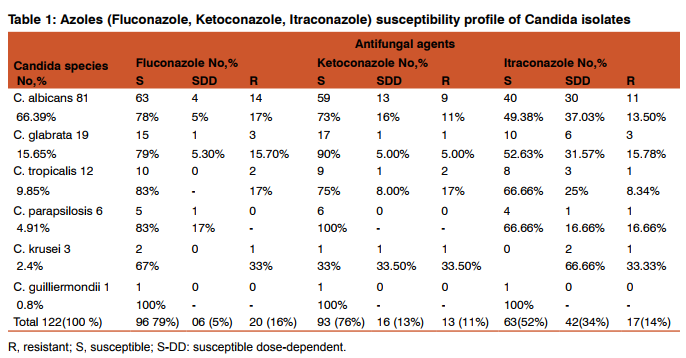
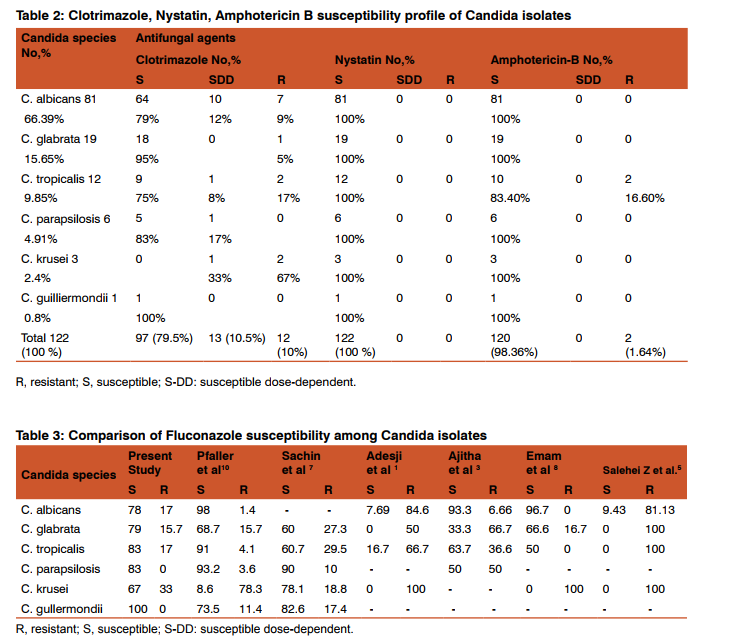
During the study period a total of 122 Candida spp. were isolated from 410 high vaginal swab processed for isolation and identification of Candida. Out of 122 isolates, 81(66.39%) were C. albicans, 19(15.65%) were C. glabrata, 12(9.85%) C. tropicalis, 6(4.91%) C. parapsilosis, 3(2.4%) C. krusei and 1(0.8%) were C. gullermondii. In our study All the Candida spp. had shown 100% sensitivity against Nystatin and almost 100% sensitivity against Amphotericin-B (Except C. tropicalis which showed 83.4% sensitivity to Amphotericin-B). (Table-1, 2) C. albicans had shown maximum sensitivity of 79% and 78% against Clotrimazole and Fluconazole respectively. It had shown 17% resistance against Fluconazole. C. glabrata had shown 95% sensitivity against Clotrimazole and 15.78%, 15.70% resistance against Itraconazole and Fluconazole respectively. C. tropicalis had shown maximum 83% sensitivity against Fluconazole. It had shown 17% resistance against Fluconazole, Ketoconazole and Clotrimazole and 16.60% resistance against Amphotericin-B. C. parapsilosis had shown 100% sensitivity against Ketoconazole and Amphotericin-B, followed by 83% sensitivity against Clotrimazole and Fluconazole. It had shown 16.60% resistance against Itraconazole. C.krusei had shown 67% sensitivity against Fluconazole and 67% resistance against Clotrimazole. C. gullermondii had shown 100% sensitivity against all the six antifungal drugs used in the study. In our study overall sensitivity of Candida species against Fluconazole was 79%, Ketoconazole76%, Itraconazole 52%, Clotrimazole 79.5%, Nystatin 100% and Amphotericin-B was 98.36 %.(Table-7)
DISCUSSION
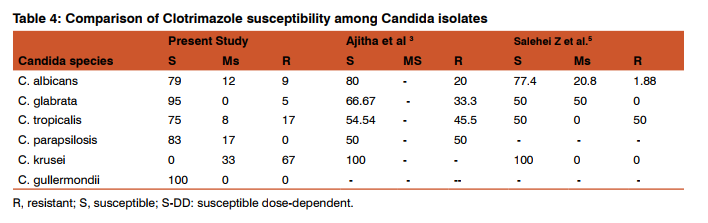
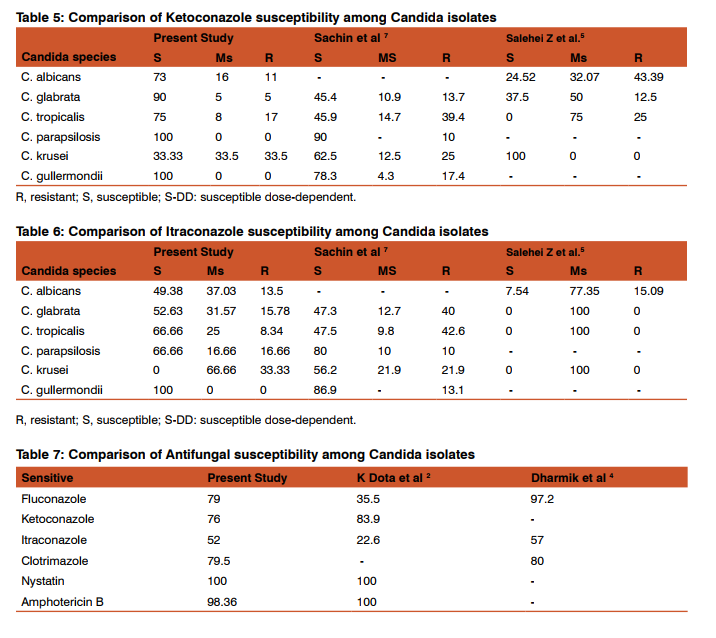
Vulvovaginal candidiasis is one of the most common fungal infections among adult women during their lifetime.3,20Clotrimazole and Fluconazole are the two antifungal drugs that are widely used in the treatment of vulvovaginal candidiasis.5 The main agent of vulvovaginal candidiasis is C. albicans; however, it seems nonalbicans species (C. glabrata and C. tropicalis) of Candida appear to be increasing.5,7 C. glabratais the second commonest agent in vaginal infections in most regions.5 The sensitivity patterns of Candida isolates varies among studies in different countries.2 In our study Fluconazole had shown 78% sensitivity against C. albicans, which is quite comparable with study of Babin et al17(76%),while study done by Ajitha et al3 and Emam et al8 showed higher sensitivity rate of 93.3% and 96.7% respectively. Fluconazole was 33% resistant against C. kruseiin this study while in study conducted by Sachin et al7 it was 18.8% resistance and in study by Adesji et al1 it was 100% resistance. (Table 3) Clotrimazole had highest sensitivity 100 % against C. gullermondii followed by C. glabrata 95%in our study, while 66.67% sensitivity was found in study of Ajitha et al3 against C. glabrata. (Table 4) Ketoconazole had shown maximum resistant 33.5% against C.krusei in our study,while it was 25% in study of Sachin et al7 .(Table 5) Itraconazole was 13.5% resistant among C. albicans in our study which is 13.95% in Babin et al17and15.09% resistance in Salehei Z et al.5 (Table 6) Nystatin was 100% sensitive in all Candida spp. in our study which is quite comparable with Emam et al8 (100%). Amphotericin –B was almost 100% sensitive in all Candida spp., except C.tropicalis83.4% sensitive in our study. Data in Ajitha et al 3 was 81.8% and in Lata et al 9 75.6%. Overall, Fluconazole was 79% sensitive, while in study of Kelen Dota et al2 Fluconazole showed 35.5% and in Dharmik et al4 97.2% sensitivity.(Table 7) In this study the overall sensitivity of Clotrimazole and Itraconazole were 79.5% and 52% respectively, which were comparable to study of Dharmik et al4 80% and 57% respectively. (Table 7)
CONCLUSION
This study provides information on species pattern and antifungal susceptibility of Candida species isolated from VVC cases attending Obstetrics and Gynecology OPD of SMIMER Hospital, Gujarat, India. In the present study, though C. albicans was the commonest spp. isolated, there was a slight increase in the prevalence of non-albicans Candida spp. Among the non-albicans Candida, C. glabrata was the commonest species. Majority of C. albicans isolates were susceptible to Fluconazole. Antifungal susceptibility pattern showed that Candida isolates were more sensitive to Amphotericin –B and Nystatin, compared to that of Clotrimazole and Azoles. C. krusei had shown maximum resistance among all the Candida species. In vitro susceptibility testing of the yeast to antifungal agents will play a vital role in appropriate selection of antifungal agents for the treatment of fungal infections.
ABBREVIATIONS
VVC-Vulvovaginal Candidiasis
SDA-Sabourad’s Dextrose agar
NCCLS-National Committee for Clinical Laboratory Standards
ACKNOWLEDGEMENT
Authors would like to thank Dean and Medical Superintendent, SMIMER Medical College and hospital for allowing us to carry out this study and for providing the facilities and help. They are also thankful to the Head of Department Obstetrics and Gynecology, SMIMER Medical College and hospital for allowing us to collect the specimens of their patients. Authors would also like to extend their gratitude to authors whose articles are cited and included in the references of the present study. They are also grateful to authors/editors/publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed
References:
1. Adesiji Y.O., N. Ndukwe, B. M. Okanlawon. C Isolation and Antifungal Sensitivity to Candida Isolates in Young Females. Central Europen Journal of Medicine. Accepted 21 September 2010.
2. Kelen F. D. Dota, Alessandra R. Freitas, Marcia E. L. Consolaro, Terezinha I. E. Svidzinski. A Challenge for Clinical Laboratories: Detection of Antifungal Resistance in Candida Species Causing Vulvovaginal Candidiasis. Journal of Labmedicine February 2011 Volume 42 Number 2
3. Dr. Ajitha Reddy, Dr. Maimoona Mustafa. Phenotypic identification of candida species and their susceptibility profile in patients with genitourinary candidiasis International Journal of Advanced Research (2014), Volume 2, Issue 12, 76-84.
4. Dharmik Preeti G., Gomashe A. V., Upadhyay V. G. Susceptibility Pattern of Various Azoles Against Candida Species Causing Vulvovaginal Candidiasis. The Journal of Obstetrics and Gynecology of India (March–April 2013) 63(2):135–137
5. Zahra Salehei , Zahra Seifi, Ali Zarei , Mahmoudabadi A .Sensitivity of Vaginal Isolates of Candida to Eight Antifungal Drugs Isolated From Ahvaz, Iran. Jundishapur J Microbiology. 2012;5(4):574-577.
6. Arul Sheeba Malar S., Viswanathan T., Malarvizhi A., Lavanya V, Moorthy K. Isolation, characterisation and antifungal susceptibility Pattern of Candida albicans and non albicans Candida from Integrated counseling and testing centre (ICTC) patients African Journal of Microbiology Research Vol. 6(31), pp. 6039-6048, 16 August, 2012.
7. Sachin C Deorukhkar, Santosh Saini. Vulvovaginal Candidiasis due to non albicans Candida: its species distribution and antifungal susceptibility profile Int. J. Curr. Microbiol App. Sci (2013) 2(12): x-xx .
8. Sherin M Emam, Abeer A Abo Elazm1 and Ahmed Walid A. Exoenzymes Production and Antifungal Susceptibility of Candida Species Isolated from Pregnant Women with Vulvovaginitis. Journal of American Science 2012;8(12).
9. Lata Patel, Jayashri Pethani, Palak Bhatia et al . Prevalence of Candida infection and its Antifungal susceptibility pattern in tertiary care hospital, Ahmedabad. National journal of Medical researchVol.2 Issue4 Oct-Dec 2012.
10. M. A. Pfaller, D. J. Diekema, 1D. L. Gibbs,V. A. Newell et al Results from the ARTEMIS DISK Global Antifungal Surveillance Study, 1997 to 2007: a 10.5-Year Analysis of Susceptibilities of Candida Species to Fluconazole and Voriconazole as Determined by CLSI Standardized Disk Diffusion journal of clinical microbiology, Apr. 2010, p. 1366–1377.
11. Chander J. Candidiasis. In: A textbook of Medical Mycology, 3rd ed. Mehta Publishers, New Delhi, 2009; 266-90.
12. Fran Fisher, Norma cook, Fundamentals of diagnostic mycology, Philadelphia. W.B saunders company 1998;197-222.
13. National Committee for clinical Laboratory standards. Metod for antifungal disk diffusion susceptibility for yeasts. Approved guidelines.2004.wayne.
14. Vandenbossche, I., Vaneechoutte, M., Vandevenne, M., Baere, T. D., and Verschraegen G. 2002. Susceptibility testing of fluconazole by the NCCLS broth macrodilution method, E- test, and disk diffusion for application in the routine laboratory. J ClinMicrobiol.40 (3): 918-921.
15. Arthur L. Barry and Steven D. Brown. Fluconazole Disk Diffusion Procedure for Determining Susceptibility of Candida Species journal of clinical microbiology, Sept. 1996, p. 2154–2157
16. Debora Moreira, Marcos Ereno Auler, Luciana da Silva Ruiz et al. Species Distribution and Antifungal Susceptibility Of Yeasts Isolated From Vaginal Mucosa. Rev Patol Trop Vol. 43 (1): 48-56. Jan.-Mar. 2014.
17. Deepa Babin, Subbannayya Kotigadde, P. Sunil Rao and T.V Rao. Clinico-mycological profile of vaginal candidiasis in a tertiary care hospital in Kerala. International Journal of Research in Biological Sciences 2013; 3(1): 55-59.
18. Keyvan Pakshir, Leila Bahaedinie, Zahra Rezaei et al. In vitro activity of six antifungal drugs against clinically important dermatophytes. Jundishapur Journal of Microbiology (2009); 2(4): 158-163.
19. E. E. Akortha, V. O. Nwaugo and N. O. Chikwe. Antifungal resistance among Candida species from patients with genitourinary tract infection isolated in Benin City, Edo state, Nigeria African Journal of Microbiology Research Vol. 3(11) pp. 694-699, November, 2009.
20. Parisa Badiee , Abdolvahab Alborzi , Mohammad Ali Davarpanahb et al. Distributions and Antifungal Susceptibility of Candida Species from Mucosal Sites in HIV Positive Patients Archives of Iranian Medicine, Volume 13, Number 4, July 2010.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License