IJCRR - 14(6), March, 2022
Pages: 71-79
Date of Publication: 15-Mar-2022
Print Article
Download XML Download PDF
Morphological, Phytochemical and Fourier-transform Infrared Spectroscopy (FTIR) Spectroscopic Analysis of Selected Species of Portulacaceae: A Study
Author: Beulah Jerlin S, Santhiya Raichel M
Category: Healthcare
Abstract:Introduction: Morphological characters of plants to assess the differences or similarities in plant taxa and use these characters for plant identification. And also an attempt is made in the present study to analyze the functional group of active compounds present in the leaf and stem extract of selected species of Portulacaceae.
Objective: To study morphological, phytochemical and Fourier-transform infrared spectroscopy (FTIR) spectroscopic analysis of selected species of Portulaceae.
Methodology: Physical examination and measurement of physiognomic features of fresh floral and vegetative sections of the plants were used to conduct a morphological study on the genus Portulaca. Phytochemicals were screened and analyzed with standard methods. 2,2-diphenyl-1-picrylhydrazylquenching assay were selected to investigate antioxidant free radical scavenging activity respectively.
Result: Morphologically they are dissimilar. Leaves are needle in P. grandiflora and obovate in P. umbraticola. Petals are numerous in P.grandiflora and 5 in P. umbraticola. The result of preliminary phytochemical screening indicated that leaf and stem of both species. Both plants were free from steroids. Moreover, quantitative estimation of phytochemicals also exhibited the leaf and stem of both species. Secondary metabolites, which are abundant in plants and have fascinating biological activities, are an important source with a variety of structural arrangements and properties. They have a rich source of protein and have high antioxidant scavenging activity.
Conclusion: Both species of Portulacaceae carry rich protein and have high therapeutic values. Therefore further efficacy and safety studies are encouraged on this potential herb with the hope of replacing some less effective ones in clinical practice especially for antidiabetic and anticancer.
Keywords: Portulaca grandiflora, Portulaca umbraticola, Morphology, FTIR spectroscopy, DPPH, Phytochemicals
Full Text:
INTRODUCTION
Plants are major sources of traditional medicines with a large variety of bioactive constituents, which are effective against different diseases. The dependence of man on plant resources is as old as the various human civilizations. Plant-derived drugs are still a valuable resource in the fight against serious diseases, particularly in developing countries. Since ancient times, higher plants have played a dominant role in the preservation of human health as sources of medicinal compounds.1
Medicinal plants are used by people all over the world to treat a variety of human and animal diseases. These bioactive constituents are responsible for the plants' major biological activities. Medicinal plants are rich sources of antibacterial and antifungal agents and are used as sources of potent and beneficial drugs in many countries.2
Some ornamental plants are grown for medicinal purposes because they contain a variety of bioactive compounds such as phenolic compounds, carotenoids, antioxidants, essential oils, and other secondary metabolites.3 Ornamental plants like Ocimum sp., Nicotiana sp., Ixora, Aloe vera, Agave, etc. Roses, nasturtium, hibiscus, marigold, Calendula, and other ornamental flowers are widely cultivated in homes and have a variety of medicinal uses. Furthermore, plant-based remedies can be much less expensive and have less side effects than pharmaceutical-based medications in terms of protecting against free radicals.
Phytochemicals are compounds found in medicinal plants, leaves, vegetables, and roots that act as a protective mechanism and provide protection against a variety of diseases. Phytochemicals are primary and secondary compounds. Chlorophyll, proteins and common sugars are included in primary constituents and secondary compounds have terpenoid, alkaloids and phenolic compounds.4 According to some generous estimates, almost 80% of the present day medicines are directly or indirectly obtained from plants.5
Any material that delays or inhibits oxidative damage to a target molecule is considered an antioxidant.6 The main characteristic of an antioxidant is its ability to trap free radicals. Antioxidant compounds such as phenolic acids, polyphenols, and flavonoids scavenge free radicals like peroxide, hydroperoxide, and lipid peroxyl, inhibiting the oxidative processes that cause degenerative diseases.7
FTIR used for the identification of functional groups of the compounds present in the sample.8 FTIR enables infrared spectrum measurements to be done quickly and accurately on a wide variety of biological specimens.9
Plant based medicines are safe and effective for the treatment of many ailments. Phytochemical screening is helpful to detect the various important compounds which could be used as the base of modern drugs for curing various diseases. Keeping this in mind the investigator selects the two plants from Portulacaceae the purslane family of flowering plants, in the order Caryophyllales.
Portulaca grandiflora, Hook.is a common garden plant in Japan. The entire plant is depurative. Hepatitis, cirrhosis of the liver with ascites, swelling, and pain in the pharynx are all treated with it. Externally, the fresh juice of the leaves and stems is used to treat snake and insect bites, burns, scalds, and eczema.
Portulaca umbraticola, also known as the wing pod purslane, is a perennial succulent in the genus of flowering plant Portulaca. While it is easy to confuse this species with P. oleracea, the foliage of this species is much smaller and wider than that of other species. Roots are edible. In reality, it contains more nutrients than spinach. It has no bitter taste and can be consumed raw or fried. It's best for soups and stews because of its mucilaginous consistency. Seeds can be eaten raw or ground and used to make bread. Hence, the present work is aimed at with the following objectives
-
To find out the Morphological characters to eliminate possible confusion to identify them.
-
To assess the status of phytochemical properties of leaf and stem of Portulaca grandiflora and Portulaca umbraticola.
-
To analyze the phytochemicals quantitatively.
-
To identify the functional group of phytochemicals by using FTIR spectroscopy.
-
To evaluate the antioxidant activity of leaf and stem samples of Portulaca grandiflora and Portulaca umbraticola by using DPPH free radical assay.
MATERIALS AND METHODS
Collection and processing of the plant materials
Portulaca grandiflora and Portulaca umbraticola were collected from the home garden of Thoothukudi. The Leaf and stem of Portulaca grandiflora and Portulaca umbraticola were cut into pieces, sun dried to reduce the moisture level. The plant material was pulverised after drying to obtain a coarser powder material, which was then deposited in an airtight plastic container. About 10 g of each fine powdered sample was weighed and separately soaked in 100 ml of Methanol and distilled water. These were permitted to sit at room temperature for 24 hours. All the extracts were filtered through Whatman No. 41 filter paper and the filtrate was used for further analysis.
Preliminary Phytochemical Screening (Qualitative analysis):
The qualitative phytochemical test for tannins, saponins, carbohydrate, protein and phenol were carried out on the concentrated extracts using the standard procedures.10 flavonoids and quinones by Kokate.11 Coumarins, terpenoids and steroids are by Yadev.12 Alkaloids, carbohydrate, glycosides, protein and phenol were carried out on the concentrated extracts using the standard procedures to identify the constituents in leaf and stem of Portulaca grandiflora and Portulaca umbraticola.
PHYTOCHEMICAL QUANTITATIVE ESTIMATION
Total Soluble Protein.13
100 mg of sample was homogenized in 10 ml of distilled water and filtered through a muslin cloth and centrifuged at 3000 rpm for 10 minutes. To the supernatant 10% trichloro acetic acid (TCA) was added in 1:1 ratio and left in an ice bath for 30 minutes to precipitate protein. The supernatant was then discarded after centrifugation at 3000 rpm for 5 minutes. In 0.1N sodium hydroxide, the precipitate was dissolved and diluted to a known amount. 5 mL alkaline copper reagent was applied to 0.5 mL protein extract. After thorough mixing 0.5 ml of folin ciocalteu reagent was added and allowed to stand for 30 minutes; the blue colour appeared and absorbance was measured at 650 nm using UV visible spectrophotometer (Model No: UV 2371). The analysis was performed in triplicates and the amount of protein was calculated and expressed as mg/g DW.
Estimation of Carbohydrate
The total carbohydrate content was determined.14 An aliquot of 1ml sample (1mg/ml) was mixed with 5ml of 5% phenols and conc. Sulfuric acid. Tubes were vortexed and incubated for 15 minutes at a boiling water bath. After cooling to room temperature, the absorbance was measured against blank at 490 nm using a UV-VIS spectrophotometer.
Total flavonoid.15
Procedure
100 mg of sample was homogenized with 10 ml of distilled water and filtered through muslin cloth. 0.5 ml of extract was added with 2.5 ml distilled water and mixed. After 6 minutes, 0.15 ml NaNO was added and again after 6 minutes, 0.3 ml of 10% AlCl3 was added. After 5 minutes 1 ml of 1M NaNH and 0.5% ml of water were added. The absorbance against a blank was measured at 510 nm after the solution had been thoroughly mixed. Quercetin was used as standard and the results were expressed as mg quercetin equivalents (QE)/g dry weight
Vitamin-C (Ascorbic acid)16
100 mg of each sample was homogenized with 10 ml of 5% trichloro acetic acid (TCA). The homogenate was centrifuged at 3000 rpm. To 2 ml of protein free supernatant, 1 drop of indophenol reagent and 0.5 ml of DT reagent were added and incubated at 10°C for 1 hour. Then cooled in an ice bath and 2.5 ml of 85% sulphuric acid was added. After intermittent shaking for 30 minutes (until red colour appeared), 30 absorbance was measured at 540 nm. L-ascorbic acid was used as standard and the results were expressed as mg/g DW.
Vitamin E (Tocopherol).17
The sample (2.5 g) was homogenized in 50 ml of 0.1 N sulphuric acids and allowed to stand overnight. The content in the flask was shaken vigorously and filtered through Whatman No.1 filter paper. Aliquots of the filtrate were used for estimation. Into stoppered centrifuge tubes, 3 ml of extract and 3 ml of water were pipetted out separately. To both the tubes, 3 ml of ethanol and 3 ml of xylene were added, mixed well and centrifuged. The Xylene (2.0 ml) layer was transferred into another stoppered tube. To each tube, 2.0 ml of dipyridyl reagent was added and mixed well. The mixture (3 ml) was pipetted out into a cuvette and the extinction was read at 460 nm. Ferric chloride solution (0.66 ml) was added to all the tubes and mixed well. After 15 minutes, the red colour formed and read at 520 nm.
ANTIOXIDANT ACTIVITY
Crude sample extracts were prepared by pouring 100ml of distilled water in a conical flask containing 10g of each sample separately in the ratio of 10:1 (V/W). After 24 hours, the mixture was filtered through whatman no:1 filter paper and the filtrate was evaporated to dryness. Crude (aqueous) extracts of all samples (1mg/ml) were used for the determination of free radical scavenging activity.
Free radical scavenging assay.18
Free radical scavenging assay was measured by 2-2 Diphenyl, 1-picryl hydrazine (DPPH) method proposed with slight modifications. 1ml of aliquot of test sample was added to 3ml of 0.004% DPPH solution prepared in methanol. The absorbance was measured at 517 nm after the mixture was vortexed for 1 minute and held at room temperature for 30 minutes in darkness. Allow absorbance of the reaction mixture indicated a high free radical scavenging activity. Ascorbic acid was used as standard.
DPPH scavenging activity (%)
A control –A test / A control * 100 Where, A control is the absorbance of the DPPH solution without test solution. A test is the absorbance of DPPH with the test solution. Aqueous extract was used as blank.
FT-IR Spectroscopy: Using a mortar and pestle, a small amount of plant specimen powder was combined with KBr salt and compressed into a thin pellet. Infra-red spectra were recorded as KBr pellets on a Thermo Scientific NicotiS5ID1 transmission, between 4000-400 cm-1.
RESULT
The morphological characters of the studied taxa are presented in (Table1). Both taxa were ornamental perennial herbs. In P.grandiflora stem are whitish herbaceous, prostate, cylindrical thick and succulent with scarcious or hairy stipular appendage. Leaves are needle like, simple alternate one leaf per node along the stem, small group at the end of the branched. The flowers are solitary, axillary, bisexual, small groups at the end of branches, but flowers only flourish one after another and come in various colours. Flowers bracteates, hermaphrodite, actinomorphic, showy, complete and hypogynous. The lifespan of flowers is one day they are open during day time. Subconnate at the base, sepals 2, green in colour persistent, united at the base. Petals many, obovate with entire margin. Stamens 8-9 in one whorl, adnate to petals inserted on calyx, filaments basally connate, about 2.5-6 mm long, anthers 2-4 locular, dorsifixed. Semi inferior, unilocular ovary with 4-many ovules on free central placentation, type 5 fid, 8-13 mm long, stigmas 5 linear, 2.5-3 mm long. (Table 2)
QUALITATIVE PHYTOCHEMICAL SCREENING
The preliminary phytochemicals screening of leaf and stem of selected plants such as Portulaca grandiflora and Portulaca umbraticola were presented in Table 3. Leaf samples of P.grandifloracontain Flavonoids, Alkaloids, Glycosides, Terpenoides, Betacyanin and Quinones were present in aqueous solution. Flavonoids, Alkaloids, Glycosides carbohydrates Terpenoides, Quinones and Anthraquinone were present in Methanol extract. Leaf extract of P. umbraticola containing Alkaloids, Glycosides, Protein, Terpenoids, and Betacyanins were present in aqueous solution. Alkaloids, Glycosides, Coumarins and Anthraquinone were present in methanol extract. Stem samples of P.grandiflora contain Alkaloids, Protein and Betacyanin were present in aqueous solution. Flavonoids, Alkaloids, Carbohydrates, and Anthraquinone were present in methanol extract. Stem of P. umbraticola possible presence of Saponins, Flavonoids, Alkaloids, Carbohydrates, Betacyanin and Anthraquinone in the methanol extracts. Alkaloids and Anthraquinone were present in aqueous extracts. The result of preliminary phytochemical screening indicated that tannins, saponin, phenol and steroids are totally absent in both the species.
QUANTITATIVE ANALYSIS
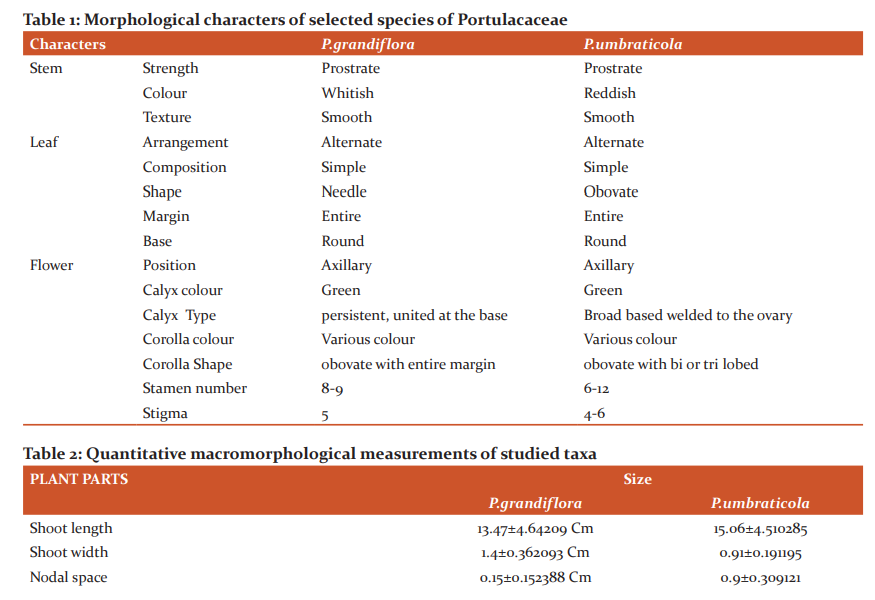
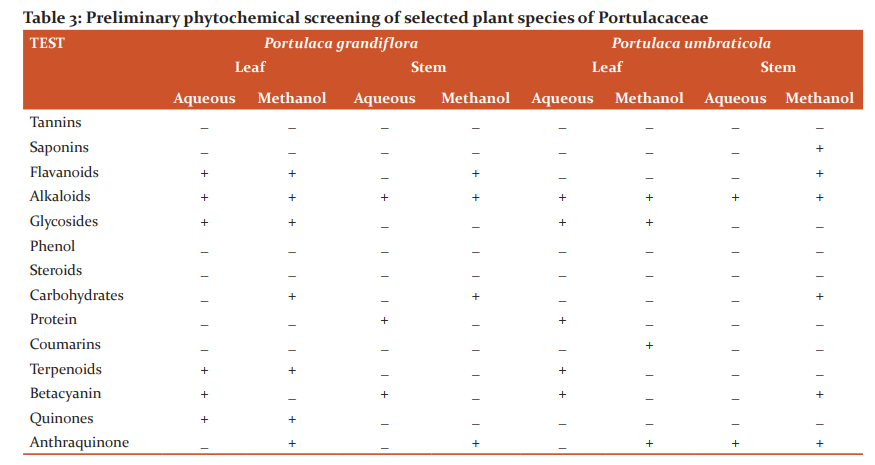
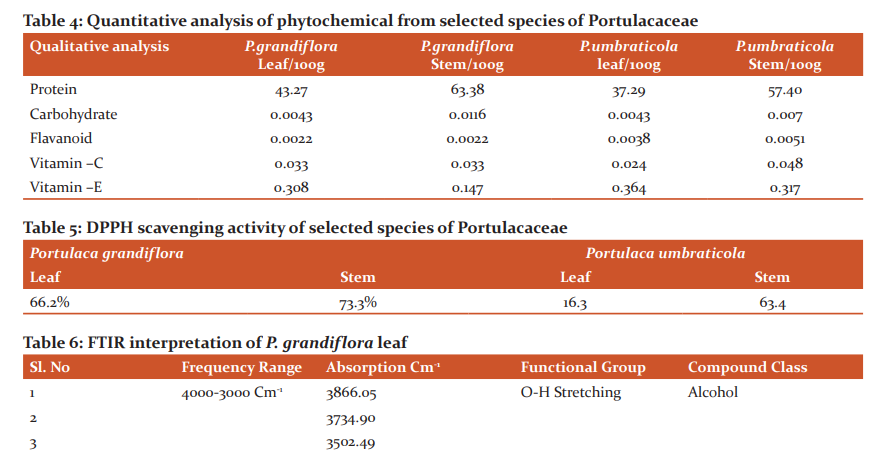
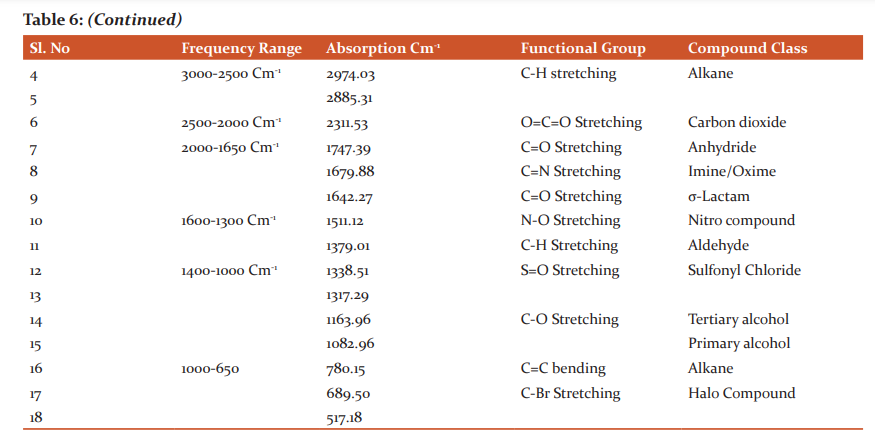
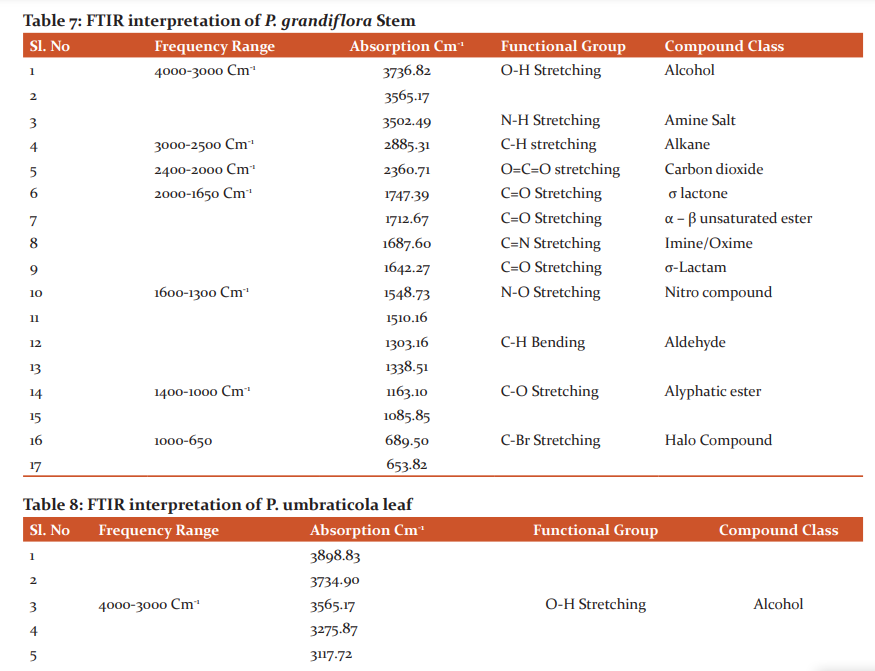
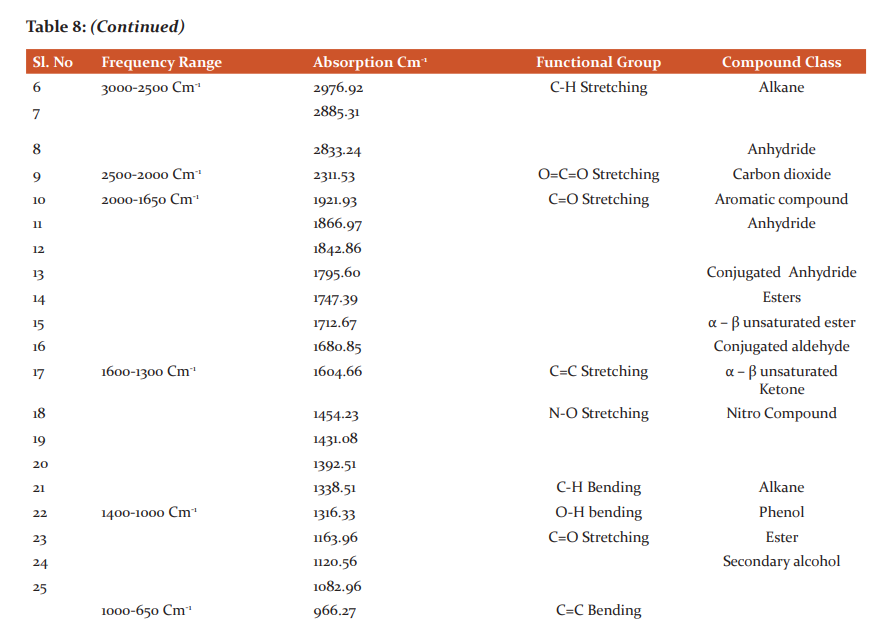
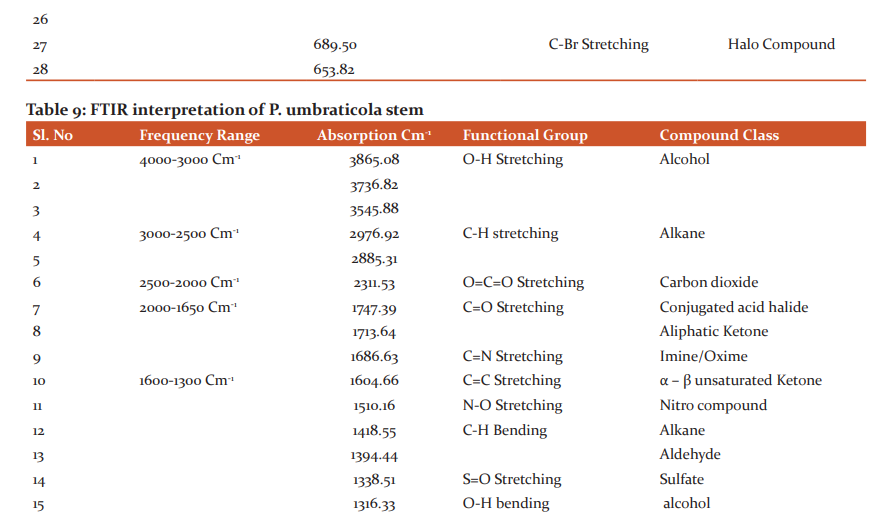
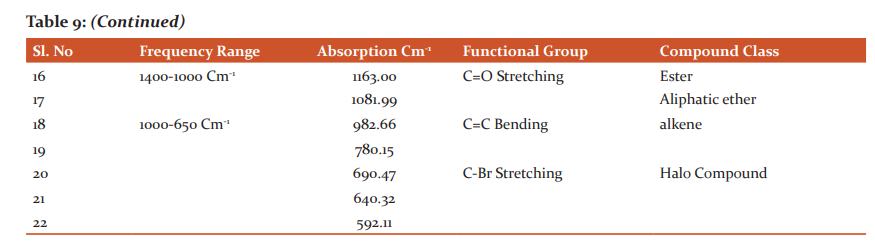
More amount of Protein was observed in the stem than the leaf. Carbohydrates and Flavonoids were poor in all the samples. Vitamin E is higher than Vitamin C. The level is maximum in all the samples. Carbohydrates are very less. The results were presented in Table 4. The results of anti–oxidant DPPH scavenging activity shows more percentage in stem than leaf of P.umbraticola. In Portulaca pilosa leaf shows more than stem. The result is present in Table 5. Based on the peak value in the infrared radiation region, the FTIR spectrum was used to classify the functional group of the active components. The results of FTIR peak values and functional groups were represented in Table 6-9. Fig.1-4. The FTIR spectrum confirmed the presence of alcohols, phenols, alkanes, alkynes, alkyl halides, aldehydes, carboxylic acids, aromatics, nitro compounds and amines in different samples.
DISCUSSION
Morphological characters of selected species of Portulacaceae were dissimilar. Leaves are needles in P.grandiflora Obovate in P. umbraticola. This was proven by earlier literature of Shide.19,20 Phytochemicals are substances that carry multiple biological properties. Preliminary phytochemical screening of both samples shows the possible presence of Alkaloids, Glycosides, Protein, Terpenoids, Coumarins, Carbohydrates, Anthraquinone and Betacyanins. The important thing is that Flavonoids were present in all samples. Flavonoids are an economically valuable main class of secondary metabolites and are good for human health and also help the plants to protect against adverse environmental conditions.21,22 By using FTIR spectrum, we can confirm the functional group. This result coincides with previous observations by various biologists.23 DPPH scavenging activity of P.grandiflora Obovate is more than P. umbraticola. 24
Conclusion
Morphologically they are dissimilar. Leaves are needle in P.grandiflora and obovate in P.umbraticola. Petals are numerous in P.grandiflora petals are 5 in P. umbraticola. The result of preliminary phytochemical screening indicated that leaf and stem of both species. Both plants were free from steroids. Moreover, quantitative estimation of phytochemicals also exhibited the leaf and stem of both species. The plant's rich source of secondary metabolites with interesting biological activities in general these secondary metabolites are an important source with a variety of structural arrangements and properties. They have a rich source of protein and have high antioxidant scavenging activity. Therapeutic value is also rich. Therefore further efficacy and safety studies are encouraged on this potential herb with the hope of replacing some less effective ones in clinical practice especially for antidiabetics and anticancer.
ACKNOWLEDGEMENT
This research is a part of the M.Sc project of second author. Authors gratefully acknowledge St. Mary’s College (Autonomous) Thoothukudi. For providing an enormous resource and peaceful environment for the successful project completion.
Conflicts of Interest: The author reports no conflict of Interest.
Source of Funding: Nil

References:
-
Farombi. E.O. African indigenous plants with chemotherapeutic potentials and biotechnological approach to the production of bioactive prophylactic agents. Afr. J. Biotechnol. 2003. Vol 2:Issue 12 pp 662-671.
-
Mahesh B, Satish S. Antimicrobial activity of some important medicinal plant against plant and human pathogens.WJAS., 2008 Vol 4:Issue (S) PP 839-843.
-
Kaushik P, Dhiman A K. Medicinal Plants and Raw Drugs of India. 2000. ISBN-13 978-8121101745.
4. Krishnaiah D, Sarbatly R, Bono A. Phytochemical antioxidants for health and medicine – A move towards nature. Biotechnology and Molecular Biology Review 2007. Vol. 1 Issue (4), pp. 097-104.
-
Vidhya, R, Umavandhana, R. Phytochemical screening and GC-MS analysis of methanolic leaf extract of Cleome viscosa L.IAJMR (IAJMR) 2016.Vol 2 Issue (6):863-868. ISSN-2454-1370.
-
Yamagishi S, Matsui T. Nitric oxide, a Janus-faced therapeutic target for diabetic microangiopathy-Friend or foe? Pharmacol. Res. Commun 2011. Vol 64 Issue (3) 187–194.
-
Wu YY, Li W, Xu Y, Jin EH, Tu YY. Evaluation of the antioxidant effects of four main aflavin derivatives through chemiluminescence and DNA damage analyses. J Zhejiang Univ Sci B. 2011.Vol 12 Issue(9):744–7518.
-
Mittal1 P, Goswami1 M, Airi M. Phytochemical, FTIR and NMR Analysis of Crude Extract of Duranta plumieri leaves. JPSR. 2020. Vol. 12 Issue (1), 182-185.
9. Griffiths P R, de Haseth J A: Fourier Transform Infrared Spectroscopy, Chemical Analysis- A Series of Monographs of Analytical Chemistry and Its Applications, 1986. John Wiley + Sons, Chichester, New York, Brisbane, Toronto, Singapore 1986. Vol. 83 656 Seiten
10. Harbone J.B. Phytochemical Method,1998 Elsevier Science 3:11Chapman and Hall, London, 60-66.
11. Kokate, C.K. Practical Pharmacognosy,2008 4th Ed. New Delhi: Vallabh Prakashan, 107-111.
12. Yadav, M., Chatterji, S., Gupta, S.K., Watal, G. Preliminary phytochemical screening of six medicinal plants used in traditional medicine. Int J Pharm Pharm Sci . 2014. Vol 6 Issue (5):539-542.
13. Lowry, O. H., Rosebrough, N. J., Farr, A. L., Randall, R. J. "Protein measurement with the Folin phenol reagent". J Biol. Chem,1951. Vol 193 Issue (1): Pp 265–75.
14. Dubois M, Gilles K.A, Hamilton J.K, Rebers, P. A. J, Calorimetric Dubois method for Determination os sugar and related substance, Analytical Chemistry. 1956. Vol 28 Issue (3) PP 350-356.
15. Zhishen J. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chemistry,1999. Vol .64, PP.555-559, Available from: Accessed: Mai. 2013. doi: 10.1016/S0308-8146(98)00102-2. ZUR, I.A. et al. Cytological analysis of infection proc.
16. Baker H, Frank O Tocopherol. Methods and Interpretation, New York Interscience Publisher, John Wiley and Sons, Inc, 1968 Clinical Vitaminology.PP 172-3.
17. Rosenberg, H. R., Chemistry and Physiology of the Vitamins.1992. Inter Science Publisher Inc.
18. Hatano T, Kagawa H, Yasuhara T, Okuda T. Two new flavonoids and other constituents in Licorice root: their relative astringency and radical scavenging effects Chemical and Pharmacetical Bulletin., (1988), Vol 36 Issue (6) pp. 2090-2097
19. Shinde P. R, Wagh K. R, Patil P. S, Bairagi V. A. Pharmacognostic standardization and antibacterial potential of aerial herbs of Portulaca grandiflora Hooker (Portulaceae). WJPS 2014. ISSN (Print): 2321-3310; ISSN (Online): 2321-3086.
20. Netala S, Priya AM, Pravallika R, Tejasri NS, Sumaiya Shabreen Md, Nandini Kumari S. Comparative Pharmacognostic Studies on Three Species of Portulaca. IJPPR. 2014. Vol 6 Issue (4), PP 704-714.
21. Kumar V, Suman U, Rubal, Kumar S, Yadev, Flavonoid Secondary Metabolite: Biosynthesis and role in growth and development in plants. Recent trends and techniques in plant metabolic Engineering 2018. pp 19-45
22. Lakshmi T, Geetha R.V, Anitha Roy Acacia Catechu Willd: IJCRR 2012 Vol 03 Issue 05, PP 101-111
23. Sahayaraja APJ,Gowri a* V, Dharmalingama R, ShobanaaA. Angelin Premab Phytochemical screening by FTIR spectroscopic analysis of leaf and stem Extracts of wedelia biflora Int. j. nano corros. sci. eng. (2015) Vol 2 Issue (5) PP 322-334.
24. Chithra, Dharanyshri , Das A, Bindhu Structural and Antimicrobial Analysis of Methanolic Extract of Punica granatum PEEL A. IJCRR 2021 Vol 13 Issue (3) PP 175-184.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License