IJCRR - 7(19), October, 2015
Pages: 55-59
Date of Publication: 10-Oct-2015
Print Article
Download XML Download PDF
ANTIOXIDANT ACTIVITY (IN-VITRO) OF CALOTROPIS PROCERA EXTRACT FROM ARID REGIONS OF RAJASTHAN
Author: Sangeeta Loonker, Wasim A. Qadri, Jasmine Singh
Category: Healthcare
Abstract:Aim: To study in-vitro antioxidant activity of Calotropis procera. Methodology: The evaluation of antioxidant activity was done using 1,1-diphenyl-2-picrylhydrazyl (DPPH) free radical scavenging activity via Double Beam UV-Visible Spectrophotometer. Result: The IC50 values in leaves, fruits and flowers of Calotropis procera were found to be 16.08, 16.06 and 10.31\?g/mL respectively. Conclusion: The results explore the strong antioxidant activity of Calotropis procera which can be harvested by medical and pharmaceutical industries for curing several diseases.
Keywords: Calotropis procera, Antioxidant activity, DPPH, Diseases
Full Text:
INTRODUCTION
Calotropis procera is a wild shrub found in tropics of Asia and Africa. It is commonly called as ‘Akra’ in Rajasthan (India) and grows upto a height of 1-3m long with broad10-13cm wide and 17-19 cm long cutaneous leaves.1 It is traditionally used as a medicinal plant in India.2 The latex of Calotropis is used in the treatment of leprosy, eczema, inflammations, malarial and low hectic fevers3 while the leaves, fruits and roots are used in rheumatism, as anti-inflammatory, antimicrobial, antioxidant and hepatoprotective agents. The flowers are found to be effective in asthma, piles and malaria. 4-10 With this view the present research work is concentrated on evaluation of antioxidant activity of Calotropis procera. Antioxidant protects human body against oxidative stress and damage to all types of biomolecules like proteins, lipids and nucleic acid caused by overproduction or inefficient elimination of Reactive Oxygen Species (ROS). 11-12 Scientific evidences reveals that antioxidants play an important role in reducing the risk for chronic diseases including cancer and heart diseases.13
MATERIALS AND METHODS
i. Collection of plant materials and their extraction: The leaves, fruits and flowers of the plant were collected from local areas of Rajasthan. The shade dried materials were then pulverized separately to 40 mesh size, 100g each of which were then extracted in 500mL methanol using a soxhlet extractor. Finally the extracts were filtered and used for antioxidant activity evaluation.
ii. Chemicals: All chemicals were of A.R. Grade and were procured from Ases Chemical Works, Jodhpur Rajasthan
iii. Determination of Antioxidant activity using DPPH via free radical scavenging activity: DPPH free radical scavenging activity was measured according to the procedure described by Blios.14 Methanolic extracts of the samples of different concentrations (100, 200, 300, 400, 500, 600, 700, 800, 900, 1000 ppm) were added separately to each of the 3.5mL, 100µM methanolic DPPH which were then incubated for 30 min. Taking ascorbic acid as the standard, the absorbance of each of the solutions were determined at 517nm using Double Beam UV-Visible spectrophotometer (Rayleigh UV-2601).A blank reading is also noted and finally the DPPH free radical scavenging activity was calculated by the formula:
where are absorbance of blank and sample extract solutions. The IC50 values were calculated from percentage inhibition v/s concentration curves by linear regression analysis.
RESULTS
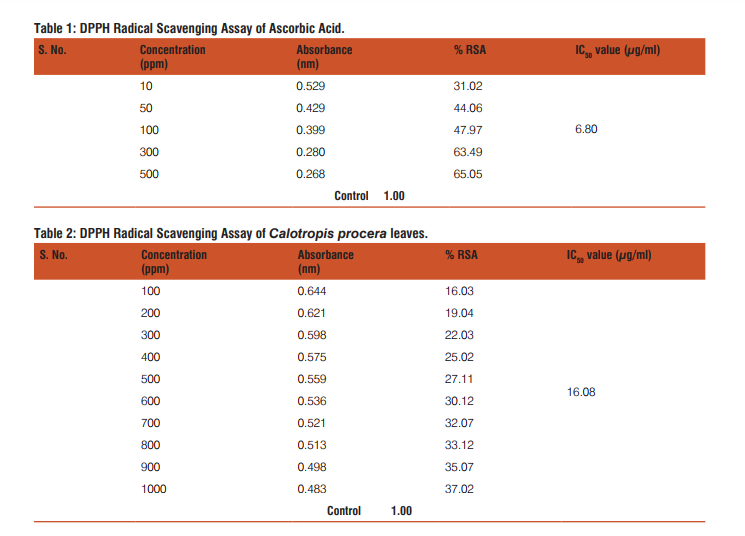
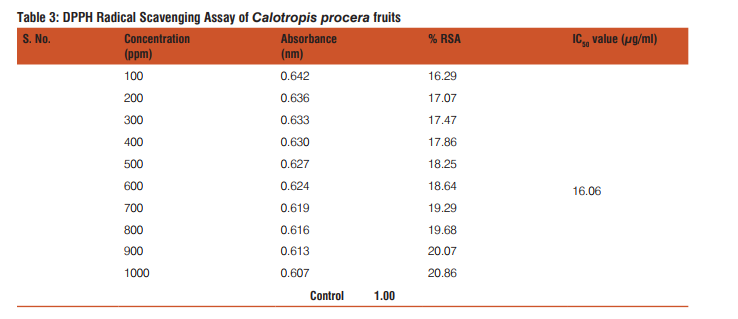
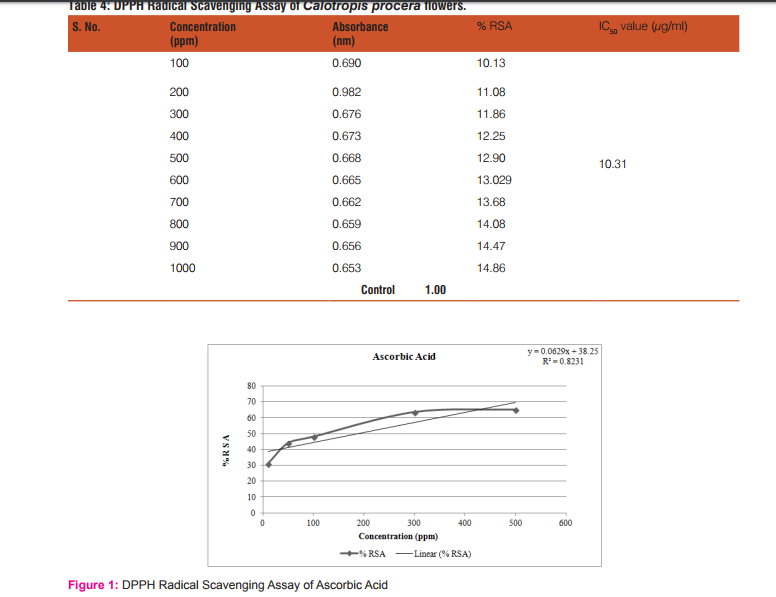
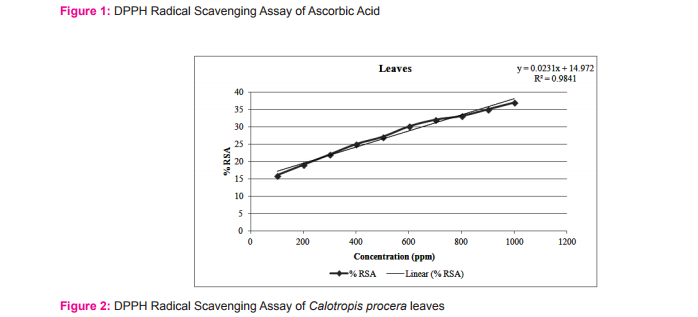
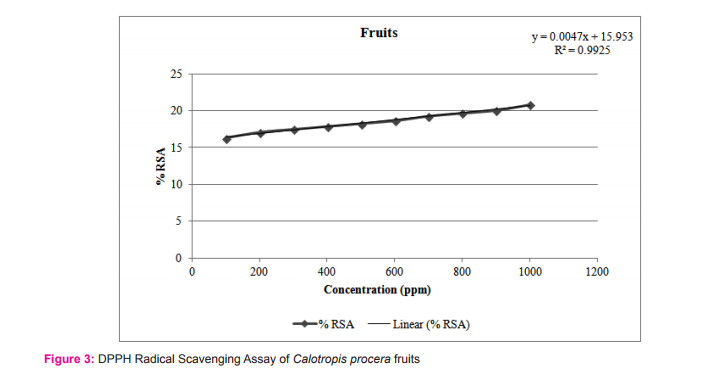
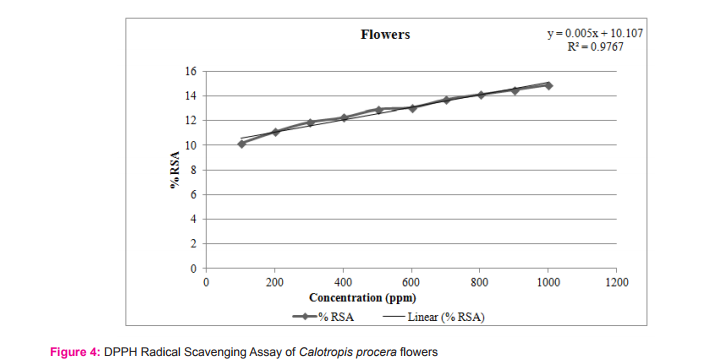
The results of the DPPH free radical scavenging activity of ascorbic acid, leaves, fruits and flowers are tabulated in tables 1-4 respectively. The variation of antioxidant activities in ascorbic acid, leaves, fruits and flowers with the increasing concentration of samples have also been depicted through the curves given in the figures 1-4 respectively.
DISCUSSIONS
The study shows that %RSA increases gradually with increase with concentration of samples. The concentration at which the %RSA value i.e. the inhibition value reaches 50% is called the IC50 value. The lower IC50 value indicated high antioxidant value in analyte.15 The IC50 values in leaves, fruits and flowers of Calotropis procera were found to be 16.08, 16.06 and 10.31µg/mL respectively.
CONCLUSION
The results showed a good antioxidant activity in the plant which can be efficiently used in the pharmaceutical purposes in arid regions of Rajasthan and world over.
ACKNOWLEDGEMENT
The authors are thankful to the Department of Chemistry, J.N.V. University, Jodhpur for providing all the laboratory facilities. The authors greatly acknowledge the indispensible help received from the scholars whose articles have been cited in this manuscript. The authors are also thankful to the authors, editors and publishers of all those articles and journals from where the literature of this manuscript is received and discussed.
DECLARATION Of CONFLICT OF INTEREST
The manuscript has no conflict of interest.
References:
1. Mainasara MM, Aliero BL, Aliero AA, Yakubu M. Phytochemical and antibacterial properties of root and leaf extracts of Calotropis procera. Nigerian J of Basic and Applied Science 2012; 20(1): 1-6.
2. Ahmed MKK, Rana AC, Dixit VK. Calotropis species (Ascelpediaceae) a comprehensive review. Pharmacog Mag 2005;1 (2):48-52.
3. Yesmin MN, Uddin SN, Mubassara S, Muhammad AA. Antioxidant and antibacterial activities of Calotropis procera Linn. American-Eurasian J Agric and Environ Sci 2008 4 (5): 550-553
4. Chandrawat P, Sharma RA. GC-MS analysis of fruits of Calotropis procera: A medicinal shrub. Res J Recent Sci 2015; 4: 11-14.
5. Mukherjee B, Bose S, Dutta SK. Phytochemical and pharmacological investigation of fresh flower extract of Calotropis procera Linn. Int J of Pharmaceutical Sciences and Research 2010; 1(2):182-187.
6. Meena AK, Yadav A, Rao MM. Ayurvedic uses and pharmacological activities of Calotropis procera Linn. Asian J of Traditional Medicines 2011; 6(2): 45-53.
7. Ansari SH, Ali M. Norditerpenic ester and pentacyclic triterpenoids from root bark of Calotropis procera (Ait) R.Br. Pharmazie 2001; 56(2):175-177.
8. Begum N, Sharma B, Pandey RS. Evaluation of insecticidal efficacy of Calotropis procera and Annona squamosa ethanol extracts against Musca domestica. J Biofertil Biopestici 2010; 1(1): 1-6.
9. Jain SC, Sharma R, Jain R, Sharma RA. Antimicrobial activity of Calotropis procera.Fitoterapia1996; 67(3): 275-277.
10. Yazna srividya B, Ravishankar K, Priya Bhandhavi P. Evaluation of in vitro antioxidant activity of Calotropis procera fruit extract, Inter J Res in Pharm and Chem 2013, 3,2231-2781
11. Dorge W. Free radicals in the physiological control of cell function. Physiol Rev 2002; 82: 47-95.
12. Thambiraj J, Paulsamy S, R. Sevukaperumal. Evaluation of in vitro antioxidant activity in the traditional medicinal shrub of western districts of Tamilnadu, India, Acalypha fruticosa Forssk. (Euphorbiaceae).Asian Pacific J of Tropical Biomedicine 2012: S127-S130.
13. Himaja M, Anand R, Ramana MV, Anand M, Karigar A. Phytochemical screening and antioxidant activity of rhizome part of Curcuma zedoaria. IJRAP 2010;1(2):414-417.
14. Blios MS. Antioxidant determinations by the use of a stable free radical. Nature 1958; 26:1199-1200.
15. Murali A, Ashok P, Madhaban V. In vitro antioxidant activity and HPTLC studies on the roots and rhizomes of Smilax zeylanica L. (Smilacaceae). Int J Pharm Sci. 2011; 3(1):192–195.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License