IJCRR - 14(1), January, 2022
Pages: 81-86
Date of Publication: 03-Jan-2022
Print Article
Download XML Download PDF
C-reactive Protein and Neutrophil Lymphocyte Ratio Levels as Predictive Biomarkers for Severity of COVID-19 Infection
Author: Prashant Bhardwaj, Manjari Kumari, Esha Singhal
Category: Healthcare
Abstract:Introduction: COVID- 19 is a highly contagious viral disease and spectrum of disease presentation range from asymptomatic to severe disease. Early diagnosis and assessment of disease severity is upmost priority to limit the morbidity, mortality and spread of disease. Aims: The aim of present study was to assess predictive values of C- reactive protein (CRP) levels and neutrophil-to-lymphocyte ratio (NLR), to disease severity to provide reference values for clinical treatment. Methodology: It was a prospective observational study in which 143 COVID-19 positive patients categorized into non-severe and severe groups. Clinical parameters, CRP values and NLR of all patients were recorded and analyzed. The receiver operating characteristic (ROC) curve was applied to determine the thresholds for both biomarkers. Result: Out of 143, total 85(59.44%) were males and 58(40.56%) female in our study. 114(79.72%) patients were non-severe and 29(20.27%) patients in severe group. Mean CRP and NLR of non severe group were 12.24(\?8.5) mg/l, 2.97(\?1.12) and in severe group 58.8(\?43.16) mg/l, 7.85(\?5.51) respectively. ROC curves analysis showed area under the curve (AUC) of 0.884 (95% CI 0.800-0.967; p value< 0.0001) for CRP and 0.817 (95% CI 0.707-0.927; p< 0.0001) for NLR. The cutoff value for CRP and NLR was 24.4mg/L and 3.6 respectively. At cutoff point, sensitivity and specificity for CRP were 82.76% and 88.6% and for NLR 75.86% and 75.44%. Conclusions: CRP and NLR are moderate accuracy diagnostic biomarkers to assess the severity of disease and of them CRP has better overall diagnostic performance than NLR.
Keywords: CRP, NLR, COVID-19 infection, Predictive biomarker
Full Text:
Introduction
The current outbreak of pneumonia which is caused by coronavirus was first reported in Wuhan, China, in December 2019.1-5Later this disease has been officially named as “COVID-19” by World Health Organization (WHO).6,7 Within few months COVID-19 disease spread globally, resulting in a worldwide pandemic.8Corona virus belongs to the family Coronaviridae, subfamily Ortho-coronavirinae and Order Nidovirales. SARS-CoV had also caused the outbreak of severe acute respiratory syndrome in 2003.9 The nature of this disease is rapidly progressive, and severely ill patients can develop acute respiratory distress syndrome, sepsis, and multiple organ dysfunction syndromes in very short period of time.10
To improve the patient outcome early diagnosis, clinicopathological monitoring and appropriate treatment protocol are essential. A Chest CT scan has an important role in assessing the disease.11 Chest CT scan is an expensive diagnostic modality and not readily available all places. So we have to consider some laboratory markers that must be inexpensive, within reach for all people and simultaneously sensitive and specific. Two biomarkers CRP and NLR both are immune-inflammatory parameters in COVID-19 infection and are associated with the progression of the infection.
In 1930s Tillett and Francis discovered CRP as an acute phase reactant. It is synthesized by liver by the action of cytokine interleukin 6 (IL- 6). Not only in bacterial infections even in other pathological processes like injuries, cardiovascular events and other inflammatory states CRP rise to very high levels. The high level of CRP is a biomarker of a pro-inflammatory state and it can be used as a prognostic marker for the underlying disease processes.12 For the early diagnosis of pneumonia C-reactive protein (CRP) levels can play important role and patients suffering with severe pneumonia have high CRP levels.13
Few studies suggest that neutrophil/lymphocyte ratio (NLR) is associated with the progression of the infection,14 or an early warning signal of severe COVID-19 infection.15 It can be considered as an independent biomarker for poor clinical outcomes and mortality in COVID-19 infection.16-20 We assessed the predictive ability of CRP levels, NLR to assess disease severity to provide a reference for clinical treatment.
Materials and Methods
This prospective observational study was started after being reviewed and approved by the institutional ethics committee (ethical committee clearance latter no. RMRI/EC/2021/54 dated on 02/04/21). We enrolled those cases that had clinical manifestations of upper respiratory tract infections like cough, shortness of breath and chest pain, which tested positive for COVID-19 by real-time reverse transcription-polymerase chain reaction (RT-PCR) using nasopharyngeal and/or oropharyngeal swabs and radiological findings of consolidation, ground-glass opacities on high-resolution computed tomography (HRCT).
After considering the inclusion and exclusion criteria we collected data of 143 patients admitted in our hospital from mid of April to May 2021 from medical records of patients. Since this was a time-bound observational study, no formal sample size calculation was done. Patients were clinically categorized into mild, moderate and severe disease according to ICMR guidelines.21 Then we sub-categorized the patients into two groups: non-severe and severe. Patients who had mild and moderate symptoms like fever, upper respiratory tract symptoms without breathlessness, SPO2>90% at room air were grouped into non-severe group and severe group had those patients who had severe symptoms like breathlessness and SPO2 <90% at room air.
The venous blood was collected at the time of admission and along with other investigations, CRP and NLR were also detected. The level of CRP was determined by the immunoturbidometery method using Liquid Microxpress Turbilyte CRP on the Benesphera C61 semiautomatic biochemical analyzer. Complete blood count was determined by five part analyzer Mindray BC-5150. NLR ratio was calculated using the simple formula of- Absolute numbers of neutrophils/ Absolute no of lymphocytes (ANC/ALC).
Statistical Analysis
SPSS (Statistical Package for Social Sciences, version 16.0.1 of IBM, USA) and Excel were adopted for data analysis. All study variables depending on the data type were summarized using appropriate measures of central tendency (mean, median) and dispersion - standard deviation (SD) or interquartile range (IQR). Categorical variables were expressed as frequencies and percentages. Receiver operation curve (ROC) was used, computation of areas under curve (AUCs; with 95% confidence intervals) and cut-off values of CRP and NLR were done. AUC >0.70 were considered to be clinically significant or relevant for good predictive score. For optimal cut-off values of CRP and NLR respective sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) were calculated. p value ≤ 0.05 were considered statistically significant along with the 95% confidence interval for the test statistic computed.
Results
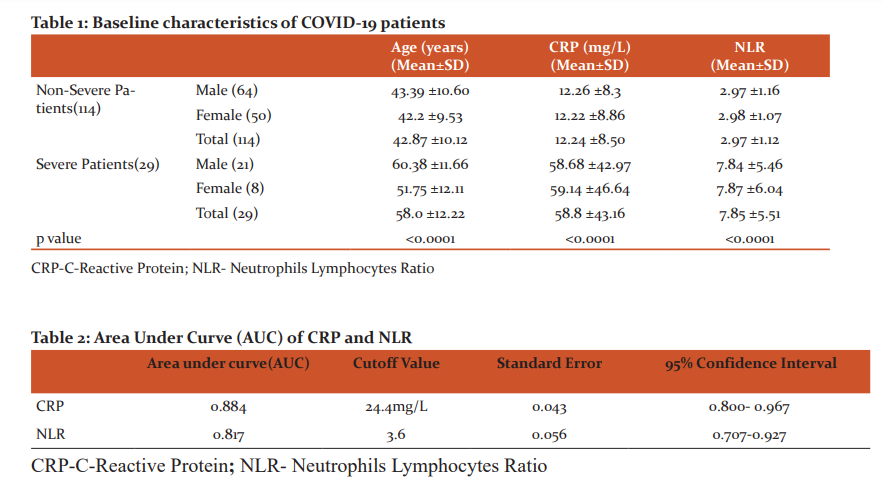
A total of 143 patients with age above 18 years and confirmed diagnosis of COVID-19 were included in during the study period. Patients were divided into two comparison groups according to their clinical profile. Among the enrolled study subjects 114(79.72%) patients were in non-severe group and, 29(20.27%) patients in severe group. There were 85(59.44%) males and 58(40.56%) females in our study. As per laboratory findings mean CRP and NLR of non-severe group was 12.24(±8.5) mg/l, 2.97 (±1.12) and 58.8(±43.16) mg/l, 7.85(±5.51) in severe group respectively. (Table 1)
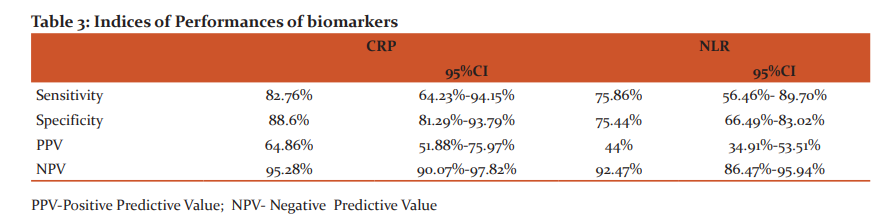
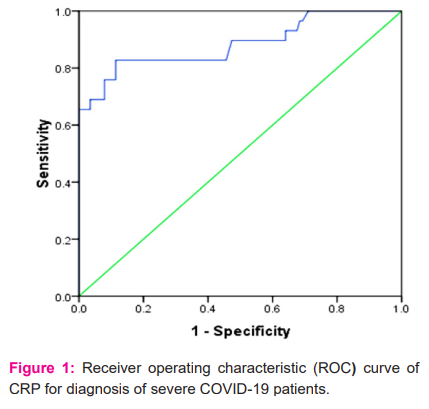
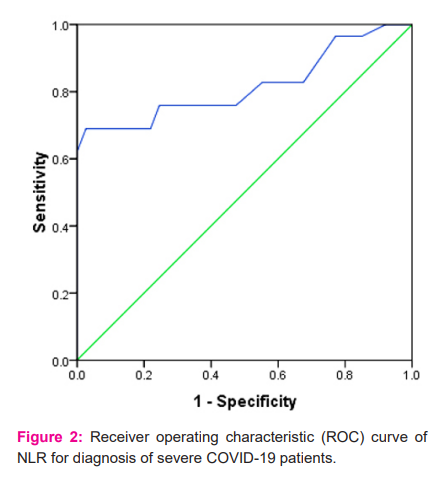
ROC curves analysis revealed moderate accuracy, with an area under the curve (AUC) of 0.884 (95% CI -0.800-0.967; p value<0.0001) for CRP (Figure 1) and 0.817 (95% CI- 0.707-0.927; p<0.0001) for NLR (Figure 2). Defined by the ROC curve, the optimal threshold value was 24.4 mg/L for CRP and 3.6 for NLR (Table 2). At optimal cut-off values, sensitivity and specificity for CRP were 82.76% and 88.6% and for NLR 75.86% and 75.44% respectively (Table 3). Table3 also illustrates Positive predictive value and negative predictive value of respective biomarkers.
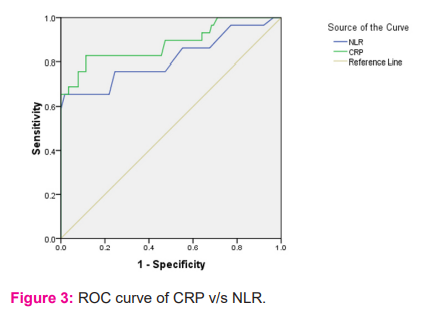
Comparative analysis of AUCs of CRP and NLR shows that CRP is a better diagnostic biomarker for patients with severe COVID-19 disease. (Figure 3)
Discussion
COVID-19 is a pandemic and caused by the SARS-CoV-2 virus infection. The SARS-CoV-2 virus is closely related to the SARS-CoV virus and both virus belong to the β-CoV corona virus family,22 with 79.5% similarity in genetic characteristic.23The spectrum of disease varies from asymptomatic individual to severe disease and death. Clinical observations have found that some patients with mild disease progress to severe disease within a short period of time. In view of this, an opportune assessment of the severity of patient's condition by means of early monitoring of different laboratory parameters is valuable for modifying the treatment.24 The pathological changes of COVID-19 are clearly evident chiefly in respiratory system and immune system damage. 25 Acute inflammatory changes like congestion, edema, exudation and clear membrane formation appear in the lungs.26 Later development of multiple-organ dysfunction occurs which may be related to the "cytokine release storm".27
The concentration of CRP in serum fairly correlates with the severity of inflammation and is unaffected by age, gender, and physical condition.28 Therefore in clinical practice CRP is one of the common inflammatory biomarkers used for identifying infection. CRP released by injured tissues and cells and activates the complement system and facilitates the clearing of microorganisms by enhancing the phagocytic process. CRP has high specificity for identifying bacterial or viral infections and it can be used for early diagnosis of pneumonia.13,29 Patients with severe pneumonia have high CRP levels, and it is used not only for diagnosis, even for the assessment of the severity of pulmonary infection.30 Similar to our study, a study by L. Wang et al. suggests that CRP levels can reflect disease severity and may be a valuable biomarker for monitoring disease.31 Study conducted by G. Wang et al. also suggests that CRP level can be a worthy marker to predict the possibility of aggravation of nonsevere COVID-19 patients.32. Zhou et al. also observed similar findings in their study.33
Neutrophils are major component of the leukocyte population and after invasion of microorganisms in the body, it gets activated and migrates at infection site and play role in host defense and immune regulation.34 Neutrophils release a large amount of reactive oxygen species to kill the virus-infected cells and interact with different cells and produces numerous cytokines and effector molecules. One of them is Vascular endothelial growth factor (VEGF) which stimulates tumor angiogenesis, growth, and metastasis.35 It is observed that the expression of VEGF-A and VEGF-C are significantly higher in COVID-19 patients as compared to normal individuals.36 Severe reduction of neutrophils count is associated with compromised immunity and increased risk of infection.37 Considering the other side of coin, human immune response to viral infections mainly taken care by lymphocytes.38 The systemic inflammation causes depression of cellular immunity resulting into a significant decrease in CD4+ T lymphocytes and increase CD8+ suppressor T lymphocyte.39 NLR is increased in viral infections and associated with worsening of clinical profile in COVID-19 patients. Ai-Ping Yang et al found in their study that NLR may be related to the severity of infection and may also stipulate the outcome.40 Study by M. Imran et al. also suggest that NLR can be used as an early alarming signal for clinician to assess the progression of severity in COVID-19 infection.41, 42 Findings of the study,41 are very much in alignment with our study.
Our study is not free from limitations. First, stronger evidences can be reported by conducting multicentric studies with larger sample size. Secondly, we classified the patients into two groups on the basis of clinical findings. It could have been better if we should have included some other radiological parameter like CT score. Thirdly, patients with mild and moderate symptoms may deteriorate to severe clinical condition at any point of time where investigating other biochemical parameters may be necessitated. Exacerbation of nonsevere disease to severe requires meticulous follow-up of patients which was not included in our study design. Lastly, severe disease can also be associated with some co-morbid conditions which might require multivariate analysis for confirmation.
Conclusion
To summarize with, we observed that both laboratory parameters CRP and NLR are moderate accuracy diagnostic biomarkers to assess the severity of disease and of them CRP has better overall diagnostic performance than NLR. Elevated CRP and NLR can be used early warning indicators of deterioration of non-severe to severe COVID-19 disease irrespective radiological findings and can help clinicians to indentify severe COVID-19 cases promptly and intervene accordingly. However, furthermore, studies are needed to strengthen these facts.
Acknowledgements
We would like to thank nursing staffs of COVID ward for their support in data collection from patient’s bed head tickets. We would convey our sincere thanks to technical staffs of our central laboratory for their excellent record keeping of reports of all investigations.
Conflict of Interest- Nil
Funding -Nil
Authors contributions
Dr Prashant Bhardwaj- Conceptualization, Methodology, Validation, Formal analysis, Investigation, Resources, Writing – Original Draft, Writing – Review & Editing, Visualization, Project administration
Dr Manjari Kumari- Methodology, Formal analysis, Investigation, Writing– Original Draft, Writing – Review & Editing, Visualization, Project administration
Dr Esha Singhal- Conceptualization, Methodology, Investigation, Writing – Review & Editing
References:
1. Lu H, Stratton CW, Tang YW. The outbreak of pneumonia of unknown etiology in Wuhan, China: the mystery and the miracle. J Med Virol 2020; 92:401–2.
2. Hui DS, Azhar EI, Madani TA,. The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health - the latest 2019 novel coronavirus outbreak in Wuhan, China. Int J Infect Dis 2020; 91:264–6.
3. Huang C, Wang Y, Li X,. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020; 395:497–506.
4. Guan WJ, Ni ZY, Hu Y,. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020; 382:1708–20.
5. Wang D, Hu B, Hu C,. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA 2020; 323:1061-1069.
6. Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu R, Niu P, Zhan F, Ma X, Wang D, Xu W, Wu G, Gao GF, Tan W; China Novel Coronavirus Investigating and Research Team. A novel Coronavirus from patients with pneumonia in China, 2019. N Engl J Med 2020; 382: 727-733.
7. WHO. WHO Director-General’s remarks at the media briefing on 2019-nCoV on 11 February 2020.
8. Munster VJ, Koopmans M, van Doremalen N, van Riel D, de Wit E. A novel Coronavirus emerging in China - key questions for impact assessment. N Engl J Med 2020; 382: 692-694.
9. Zhang W, Du RH, Li B, Zheng XS, Yang XL, Hu B, et al. Molecular and serological investigation of 2019-nCoV infected patients: implication of multiple shedding routes. Emerg Microbes Infect. 2020; 9:386–9.
10. Han, Q., Lin, Q., Jin, S. & You, L. Coronavirus 2019-nCoV: a brief perspective from the front line. J. Infect. 80; 373–377.
11.Lin C, Ding Y, Xie B, Sun Z, Li X, Chen Z, et al. Asymptomatic novel coronavirus pneumonia patient outside Wuhan: the value ofCT images in the course of the disease. Clin Imaging 2020; 63:7–9.
12. Sproston NR, Ashworth JJ. Role of C-reactive protein at sites of inflammation and infection. Front Immunol. 2018; 13:754.
13. Warusevitane A, Karunatilake D, Sim J, Smith C, Roffe C. Early diagnosis of pneumonia in severe stroke: clinical features and the diagnostic role of C-reactive protein. PloS one 2016; 11:e0150269.
14. Feng X, Li S, Sun Q,. Immune-inflammatory parameters in COVID-19 cases: a systematic review and meta-analysis. Front Med (Lausanne). 2020; 7:301.
15. Xia X, Wen M, Zhan S, He J, Chen W. An increased neutrophil/ lymphocyte ratio is an early warning signal of severe COVID-19. Nan Fang Yi Ke Da Xue Xue Bao [Journal of Southern Medical University]. 2020;40:333-336.
16. Yang A, Liu J, Tao W, Li H. The diagnostic and predictive role of NLR, d-NLR and PLR in COVID-19 patients. Int Immunopharmacol. 2020; 84:106504.
17. Liu Y, Du X, Chen J,. Neutrophil-to-lymphocyte ratio as an independent risk factor for mortality in hospitalized patients with COVID-19. J Infect. 2020; 81:e6-e12.
18. Ye W, Chen G, Li X,. Dynamic changes of D-dimer and neutrophil-lymphocyte count ratio as prognostic biomarkers in COVID-19. Respir Res. 2020;21:169.
19. Ciccullo A, Borghetti A, Zileri Dal Verme L,. Neutrophil-to lymphocyte ratio and clinical outcome in COVID-19: a report from the Italian front line. Int J Antimicrob Agents. 2020; 56:106017.
20. Li H, Zhao M, Xu Y. Biochemical analysis between common type and critical type of COVID-19 and clinical value of neutrophil/lymphocyte ratio. Zhejiang da xue xue bao. Yi Xue ban [Journal of Zhejiang University. Medical Sciences ]. 2020; 40:965-971.
21. ICMR New Delhi. Indian council of medical research, New Delhi[Internet]. Gov.in. http://www.icmr.gov.in/
22. Meo SA, Alhowikan AM, Al-Khlaiwi T,. Novel coronavirus 2019-nCoV: prevalence, biological and clinical characteristics comparison with SARS-CoV and MERS-CoV. Eur Rev Med Pharmacol Sci. 2020; 24:2012-2019.
23. Ren L-L, Wang Y-M, Wu Z-Q,. Identification of a novel coronavirus causing severe pneumonia in human: a descriptive study. Chin Med J (Engl). 2020; 133:1015-1024.
24. Li G, De Clercq E. Therapeutic options for the 2019 novel coronavirus (2019-nCoV). Nat Rev Drug Discov 2020; 19:149–50.
25. General office of the national health commission of China. Covid-19 diagnostic and therapeutic regimen (trial 7th, edition). J Cardiopulm Rehabil Prev2020;39(02):103–7.
26. Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med 2020; 8:420–2.
27. Channappanavar R, Perlman S. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin Immunopathol 2017; 39:529-539.
28 Bilgir O, Bilgir F, Calan M. Comparison of pre-and post-levothyroxine high-sensitivity C-reactive protein and fetuin-A levels in subclinical hypothyroidism Clinics 2015; 70:97–101.
29. Knudsen TB, Larsen K, Kristiansen TB, Møller HJ, Tvede M, Eugen-Olsen J, Kronborg G. Diagnostic value of soluble CD163 serum levels in patients suspected of meningitis: comparison with CRP and procalcitonin. Scand J Infect Dis 2007; 39: 542-553.
30. Sharanya K, Lakshmi K, Vinod K, Chitralekha S. Association of CRP Levels as an Inflammatory Marker in Prognosis of COVID-19 Cases. Int J Cur Res Rev. 2nd Wave of COVID-19: Role of Social Awareness, Health and Technology Sector, June, 2021, 98-100.
31. L. Wang, C-reactive protein levels in the early stage of COVID-19, Med Mal Infect 2020; 50: 332-334.
32. Wang G, Wu C, Zhang Q, Wu F, Yu B, Lv J, Li Y, Li T, Zhang S, Wu C, Wu G, Zhong Y, C-Reactive Protein Level May Predict the Risk of COVID-19 Aggravation, Open Forum Infectious Diseases. 2020; 7: ofaa153.
33. Y.-Z. Zhou, X.-B. Teng, M.-F. Han, J.-F. Shi, C.-X. Li, X.-H. Zhang, D.-Y. Hou, L.-L. Yang The value of PCT, IL-6, and CRP in the early diagnosis and evaluation of COVID-19 Eur Rev Med Pharmacol Sci 2021; 25: 1097-1100
34. Mercier J, Voutsadakis I. The platelets-neutrophils to lymphocytes ratio: a new prognostic marker in metastatic colorectal cancer. J Gastrointest Oncol. 2018; 9: 478-486.
35. Hanrahan V, Currie MJ, Gunningham SP, Morrin HR, Scott PAE, Robinson BA, Fox SB, The angiogenic switch for vascular endothelial growth factor (VEGF)-A, VEGF-B, VEGF-C, and VEGF-D in the adenoma-carcinoma sequence during colorectal cancer progression, J. Pathol. 2003; 200: 183–194.
36. Kim S, Lee S, Trang KTT, Kim SH, Kim IH, Lee SO, Kim DG, Kim S, Parthenolide exerts inhibitory effects on angiogenesis through the downregulation of VEGF/VEGFRs in colorectal cancer, Int. J. Mol. Med. 2014; 33: 1261–1267.
37. Yeo A, Henningham A, Fantino E,. Increased susceptibility of airway epithelial cells from ataxia-telangiectasia to S. pneumonia infection due to oxidative damage and impaired innate immunity. Sci Rep. 2019; 9:1-10.
38. Rabinowich H, Cohen R, Bruderman I, Steiner Z, Klajman A, Functional analysis of mononuclear cells infiltrating into tumors: lysis of autologous human tumor cells by cultured infiltrating lymphocytes, Cancer Res. 1987; 47: 173–177.
39. Menges T, Engel J, Welters I, Wagner RM, Little S, Ruwoldt R, Wollbrueck M, Hempelmann G, Changes in blood lymphocyte populations after multiple trauma: association with posttraumatic complications, Crit. Care Med. 1999; 27: 733–740.
40. Yang AP, Liu JP, Tao WQ, Li HM. The diagnostic and predictive role of NLR, d-NLR and PLR in COVID-19 patients. Int Immunopharmacol. 2020; 84:106504.
41. Imran MM, Ahmad U, Usman U, Ali M, Shaukat A, Gul N. Neutrophil/lymphocyte ratio-A marker of COVID-19 pneumonia severity. Int J Clin Pract. 2021; 75:e13698.
42. Sahu P, Rai A, Sahu S, Raut K. Haematological findings and pattern analysis in patients with COVID- 19 infection. Int J Cur Res Rev 2021;13(6):36-41
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License