IJCRR - 14(1), January, 2022
Pages: 01-08
Date of Publication: 03-Jan-2022
Print Article
Download XML Download PDF
Homocysteine, Renal Biomarkers, Glucose, Glycated Hemoglobin and Lipid Profile in Diabetic Nephropathy Patients from Gaza Strip
Author: Yassin MM, Alghora SS, Alnajjar MK, Yasin MM
Category: Healthcare
Abstract:Introduction: Homocysteine is recently speculated as an indicator for the development of diabetic nephropathy. Objective: To assess homocysteine in various stages of diabetic nephropathy and its correlation with other biochemical parameters. Methods: A mini questionnaire, and analysis of blood and urine were employed in 120 patients with type 2 diabetes (40 normo-, 40 micro- and 40 macroalbuminuric) and 40 non-diabetic controls. Results: Urinary albumin was significantly elevated in patients with normoalbuminuria (22.3\?5.2 mg/g), microalbuminuria (146.7\?80.7 mg/g) and macroalbuminuria (348.0\?37.6 mg/g) than controls (15.9\?4.8 mg/g), in microalbuminuria and macroalbuminuria versus normoalbuminuria, and in macroalbuminuria versus microalbuminuria. There were also significant increases in serum urea and creatinine in diabetic groups. Serum homocysteine behaves like urinary albumin in being significantly higher in diabetic groups (17.1\?4.8, 20.3\?5.9 and 22.9\?5.5 µmol/l, respectively) than controls (13.4\?3.7 µmol/l), in microalbuminuria and macroalbuminuria versus normoalbuminuria, and in macroalbuminuria versus microalbuminuria. Serum glucose, HbA1c, cholesterol, triglycerides and LDL-C were significantly increased whereas HDL-C was significantly decreased in diabetic groups. There were significant positive correlations of serum homocysteine with urinary albumin (r=0.564, P< 0.001), urea (r=0.654, P< 0.001), creatinine (r=0.561, P< 0.001), glucose (r=0.465, P< 0.001), HbA1c (r=0.517, P< 0.001), and triglycerides (r=0.320, P=0.001) whereas HDL-C showed significant negative correlation (r=-0.517, P< 0.001). Conclusion: Increment of serum homocysteine accompanied with elevation of urinary albumin, urea, creatinine and HbA1c, could innominate homocysteine as a novel biomarker for trans change to diabetic nephropathy.
Keywords: Serum homocysteine, Renal parameters, Glycated hemoglobin, Lipids, Diabetic nephropathy, Gaza Strip
Full Text:
INTRODUCTION
Diabetes mellitus is a metabolic disorder of endocrine origin with persistent increase of blood sugar. Prolonged hyperglycemia leads to serious damage to the heart, blood vessels, eyes, kidneys and nerves. The World Health Organization (WHO) estimated that around 422 million people all over the world are diabetic, and the annual deaths approach 1.6 million as a result of diabetes and its complications.1 These figures are expected to rise steadily over the coming few decades. Two major types of diabetes were identified; type 1 and type 2. Patients with type 2 diabetes have more complications than in type 1 patients, causing higher rate of morbidity and mortality.2 The prevalence of type 2 diabetes mellitus in Gaza Strip is relatively high and estimated at 16.8% in adults aged 42 to 74 years.3
Diabetic nephropathy is the manifestation of diabetic microangiopathy in the kidney, and is characterized by persistent albuminuria and a progressive decline in renal function.4 Appearance of albumin in urine in the range of 30-300 mg/g or microalbuminuria is the first clinical sign of diabetic nephropathy. Increase of urinary albumin excretion to more than 300 mg/24 hours is the case of macroalbuminuria which indicates the disease progression, usually accompanied with other diabetes complications such as retinopathy, cardiovascular disease (CVD) and neuropathy.5 More than 50% of patients with diabetes will progress to diabetic nephropathy in 10 to 20 years, making it one of the main causes of end-stage renal disease.6 Therefore, early detection and proper treatment of diabetic nephropathy are particularly important for patients prognosis.
Homocysteine is a sulphur-containing intermediary amino acid which is derived by the demethylation of the essential amino acid methionine.7 The main metabolic organs of homocysteine are liver and kidney. The normal range of serum homocysteine level is between 4.4 and 10.8 µmol/l. A condition of increasing levels of serum homocysteine beyond 15µmol/L is named as hyperhomocysteinemia.8 Elevation of serum homocysteine may be due to genetic insufficiencies of the enzymes needed for its metabolism, to nutritional deficits in vitamins B12, B6 and folic acid, or to medical conditions such as CVD.9,10 Several studies have also been indicated that homocysteine is closely related to the development of diabetic nephropathy.11,12 Hyperhomocysteinemia is reported to be a risk factor for diabetic nephropathy; which can directly produce cytotoxicity, lead to oxidative stress and synergistic glycation end products, and thereafter damage vascular endothelium and induce microvascular injury.13
Although type 2 diabetes and its complications account for approximately 5.7% of all Palestine mortalities3, there is a lack of diagnosis and/or under-reporting of the disease and its progression towards the end-stage renal disease. This necessitates searching for other diagnostic markers for diabetic nephropathy, besides the traditional ones. The published articles on diabetic nephropathy among Palestinians are few and recent.5 There is no previous study investigates homocysteine status among diabetic nephropathy patients in Gaza Strip. To our best knowledge, this is the leading investigation of homocysteine level and its correlations with other biochemical parameters in diabetic nephropathy among type 2 diabetic patients in Gaza Strip. Understanding of homocysteine status and its variation, as well as its relation during trans changes to diabetic nephropathy, could offer diagnostic and management values on the progression of this life-threatening disease.
MATERIALS AND METHODS
Type of study design and target population
This is a case-control investigation. A total of 120 patients with type 2 diabetes were chosen from the main and representative Diabetic Care Unit in Gaza Strip (Al Rimal Medical Center); previously diagnosed according to the World Health Organization diagnostic criteria for diabetes.14 The diabetic patients were three groups: Group I comprised 40 patients (20 males and 20 females) with normoalbuminuria (Urinary albumin<30 mg/g), Group II included 40 patients (20 males and 20 females) with microalbuminuria (Urinary albumin=30-300 mg/g) and Group III comprised 40 patients (20 males and 20 females) with macroalbuminuria (Urinary albumin>300 mg/g). The exclusion criteria were urinary tract infection, hypertensive patients (Blood pressure ≥140/90 mmHg), pregnant women, and women under hormonal therapy. Forty non-diabetic healthy individuals (20 males and 20 females) were chosen randomly from general population in Gaza Strip, and constituted the control group. Patients and controls were age matched (40 to 65 years old).
Ethical consideration
Helsinki committee provided us with written approval to perform this study under the ethical number PHRC/HC/28/13 in Gaza Strip, and an informed consent form was signed by all patients and controls prior to commence the study.
Questionnaire interview and patients’ records
Patients and controls filled in a mini questionnaire designed to suit the purpose of the study. The questions were simple, clear, direct, and depend on diabetes clinic questions adopted by the Palestinian Ministry of Health with some modifications.15 The majority of the questions were of the yes/no category, which provide a dichotomous choice.16 The questionnaire was piloted with 12 individuals (3 from each study group) not involved in the population sample. The questions were related to age, education, employment, family history of diabetes and diet. Clinical data including diagnosed diabetic complications and duration of diabetes were obtained from the patients’ records.
Urine and blood analysis
Blood and random urine samples were obtained from all participants. A fraction of blood was placed into EDTA vacutainer tube to determine HbA1c. The remainder quantity of blood was placed into a plastic tube, and left for a short period without anticoagulant to allow blood clotting. Blood samples in plastic tubes and urine samples were centrifuged at 4000 rpm/10 min using a Rotina 46 Hettich Centrifuge, Japan. Then, the obtained serum and urine samples were analyzed. Albumin concentration in urine was measured by Immunoturbidimetry-Latex assay using BioSystems kit, Spain.17 Serum homocysteine was quantitatively analyzed by enzymatic colorimetric method, using Globe diagnostics kit, Italy.18 The urease glutamate dehydrogenase/UV and the alkaline picrate procedures were employed for serum urea and creatinine measurement, using the BioSystems kit, Spain.19,20 The method employed for determination of serum glucose was glucose oxidase/glucose peroxidase (POD), using Labkit Kits, Spain.21 HbA1C was estimated through determination of glycated hemoglobin in blood as a whole with the use of Stanbio Kit, Texas-USA.22 The cholesterol oxidase/POD and the glycerol phosphate oxidase/POD assays were followed in the determination of serum cholesterol and triglycerides, using the BioSystems kit, Spain.23,24 The precipitating procedure was applied for measurement of high-density lipoprotein cholesterol, using Labkit kit, Spain25, and finally, low-density lipoprotein cholesterol was calculated using the empirical relationship of Friedewald.26
Statistical analysis
The obtained data were computer analyzed using IBM SPSS statistics version 22. A simple frequency of the study variables was presented. Chi-square (c2) was applied to show the difference between variables. Yates's continuity correction test, c2(corrected), was employed when not more than 20% of the cells had an expected frequency of < 5. The continuous variables were expressed as mean ± standard deviation and compared using the independent one-way analysis of variance (ANOVA) to test the relationship between various diabetic groups and controls. Bonferroni test served to test the difference within different studied groups. To examine the correlations between homocysteine and other studied parameters, the test of Pearson's correlation was used. The level of significance was considered when the probability values (P)<0.05.
RESULTS
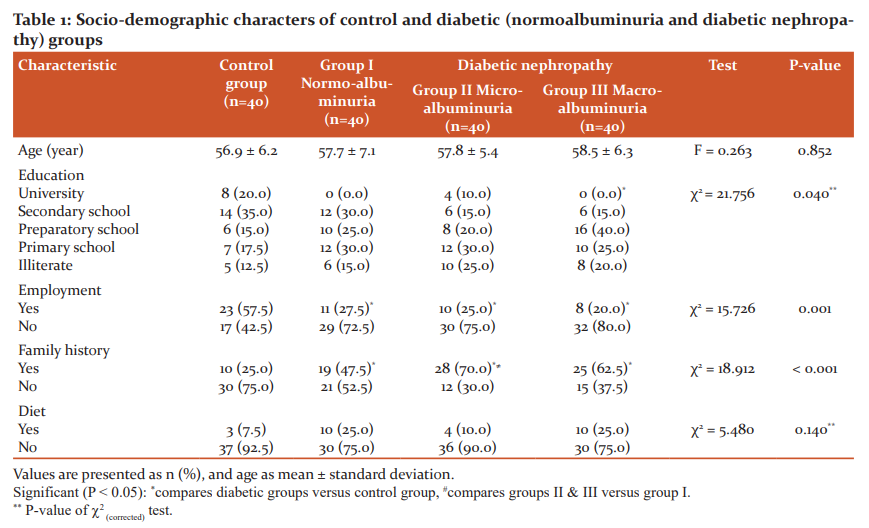
Socio-demographic characteristics of the study population
There were no significant differences between diabetic groups and control with respect to age and diet (Table 1). On the other hand, education, employment and family history of diabetes revealed significant differences among the groups (c2=21.756, P=0.040; c2=15.726, P=0.001 and c2=18.912, P<0.001, respectively). Within groups, the educational level was significantly lower in group III versus control group (P<0.05). Employment was significantly lower whereas family history of diabetes was significantly more frequent among diabetic groups when compared to control group. Also, family history was significantly higher in group II than group I.
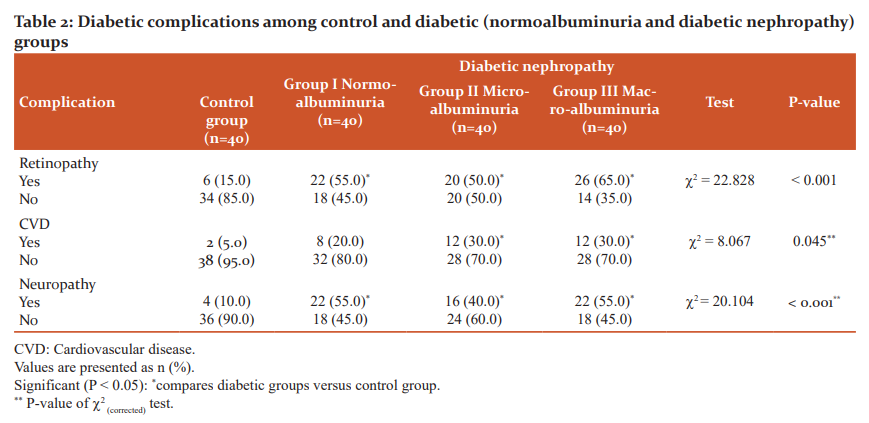
Diabetic complications and duration of diabetes
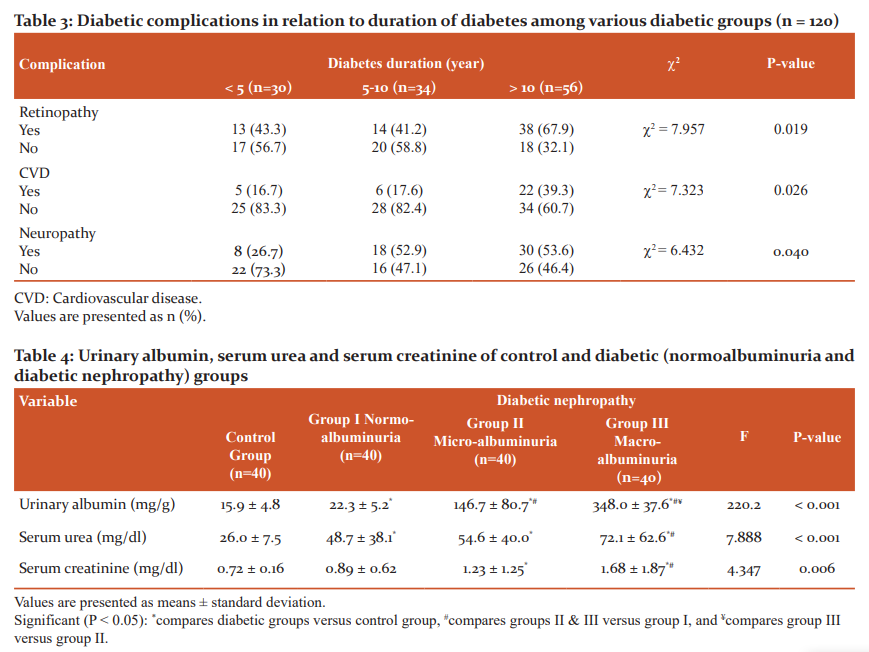
As illustrated in Table 2, diabetic complications namely retinopathy, CVD and neuropathy displayed significant differences among the different groups (c2=22.828, P<0.001; c2=8.067, P=0.045 and c2=20.104, P<0.001, respectively). Within groups, in general, retinopathy, CVD and neuropathy were significantly more frequent in diabetic groups compared to control one (P<0.05). However, no significant differences were found among the diabetic groups (P>0.05). When related to duration of the disease (Table 3), diabetic complications showed a significantly increasing trend with increasing duration of diabetes (c2=7.957, P=0.019 for retinopathy; c2=7.323, P=0.026 for CVD and c2=6.432, P=0.040 for neuropathy).
Urinary albumin, serum urea and creatinine
As depicted from Table 4, urinary albumin, serum urea and serum creatinine exhibited significant differences among various groups (F=220.2, P<0.001; F=7.888, P<0.001 and F=4.347, P=0.006, respectively). Within groups and in general, Bonferroni test revealed significant increases in these biomarkers of groups I, II and III compared to control group (P<0.05). Significant increase in urinary albumin was also registered in groups II and III against group I, and in group III against group II. Additionally, serum urea and creatinine exhibited significant increments in group III with respect to group I.
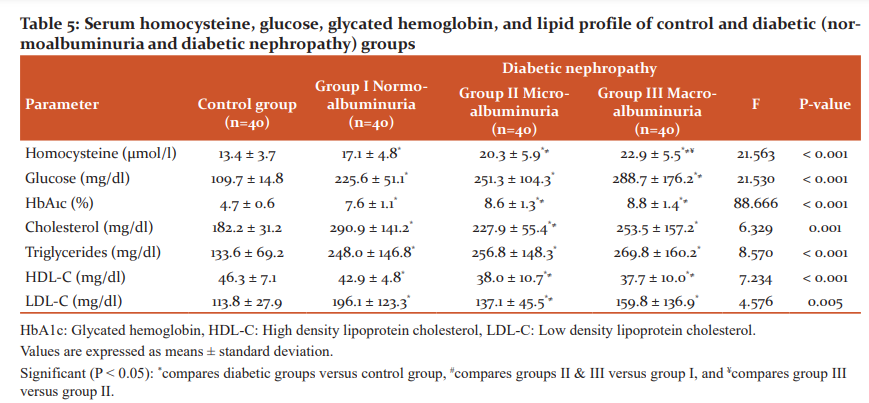
Serum homocysteine, glucose, HbA1c, and lipid profile
Serum homocysteine level shows an increasing trend upon disease progression, with significant differences among various groups (F=21.563, P<0.001), as indicated in Table 5. Like urinary albumin, homocysteine was significantly elevated in diabetic groups than control group, and in groups II and III versus group I (P<0.05). Also, homocysteine level was significantly higher in group III than group II. Significant differences among the groups was recorded for glucose and HbA1c (F=21.530, P<0.001 and F=88.666, P<0.001, respectively), with progressive significant increments in diabetic groups than control one, in group III than group I. Also, HbA1c was significantly higher in group II than group I. Lipid profile in the serum represented by cholesterol, triglycerides, HDL-C and LDL-C showed significant differences among the studied groups (F=6.329, P=0.001; F=8.570, P<0.001; F=7.234, P<0.001 and F=4.576, P=0.005, respectively). Cholesterol, triglycerides and LDL-C were significantly increased in diabetic groups in comparison to control one (P<0.05). In contrast, the level of HDL-C was significantly lower in diabetic groups than control, and in groups II and III versus group I (P<0.05).
Correlations of serum homocysteine with studied parameters
Table 6 summarized correlations between serum homocysteine and the other studied parameters. Significant positive correlations of serum homocysteine with urinary albumin (r=0.564, P<0.001), urea (r=0.654, P<0.001), creatinine (r=0.561, P<0.001), glucose (r=0.465, P<0.001), HbA1c (r=0.517, P<0.001), and triglycerides (r=0.320, P=0.001) were obtained upon applying Pearson correlation test. Conversely, HDL-C showed significant negative correlation with homocysteine (r=-0.517, P<0.001).
DISCUSSION
Diabetic nephropathy is a complicated life-threatening disease that usually develops to end-stage renal disease followed by renal failure. However, these trans changes are somewhat interdigitated and their onsets are difficult to be determined. Therefore, the proper management of the gap between the initial stage of diabetic nephropathy and renal failure is expected to improve and prolong the patient's life. This necessitates searching for other biomarkers besides the traditional ones to strengthen the diagnostic process of diabetic nephropathy. Serum homocysteine was recently innominate to be an indicator for transition to diabetic nephropathy, without testing its relation to diabetic parameters.13 Consequently, the present study assessed serum homocysteine level and examined its relation to metabolic profile in various stages of diabetic nephropathy among type 2 diabetic patients in Gaza Strip. Evaluation of homocysteine level with diabetic parameters and their correlations in diabetic nephropathy could offer a potential diagnostic tool for the progression of the disease for the seek of its management and protection.
In general, the educational level and employment were significantly lower, whereas family history of diabetes was significantly more frequent among diabetic groups in comparison to controls. This implies that low educational level, unemployment and family history are all risk factors of type 2 diabetes and its complication. The higher educated diabetic patients had good management of the disease.27 Unemployment raises poverty which consequently participates largely in raising type 2 diabetes incidence and inequality of care in spite of global health coverage.28 In addition, many authors indicated that family history is a risk factor of diabetes, particularly in type 2.29,30 The frequent diabetic complications recorded included retinopathy, CVD and neuropathy, were more prevalent in diabetic patients in comparison to controls. Such complications showed a significantly increasing trend with increasing duration of diabetes, implying that patients with long periods of diabetes are more likely to develop nephropathy. This is will be accelerated in uncontrolled disease. Similar results were previously reported.5
It is not surprising that urinary albumin level was markedly and obviously raised in diabetic groups than controls. Even within diabetic groups this traditional biomarker is still increasing with the progression of the disease, reaching its maximal level in the macroalbuminuric group. Progressive elevation of urinary albumin is largely attributed to frequent disturbance of renal filtration capacity31 and implies that employment of urinary albumin concentration as a marker for different stages of diabetic nephropathy in this study is convenient and satisfactory. Clinical screening and diagnosis of diabetic nephropathy is chiefly based on the presence of albuminuria.32 The significant increase in serum urea and creatinine in patients with diabetes than controls, and in macroalbuminurics than normoalbuminurics do confirm the occurrence of renal impairment towards the progression of diabetic nephropathy. Higher serum urea and creatinine levels were found in patients with diabetic nephropathy than in patients without.33
Serum homocysteine behaves like urinary albumin in being progressively elevated in diabetic patients, reaching its maximal level in macroalbuminuria stage of diabetic nephropathy. In this context, the significant positive correlation of serum homocysteine with urinary albumin found in this study do support this view. Hyperhomocysteinemia was recorded in diabetic nephropathy or even in hemodialysis patients, and was proposed to be associated with renal endothelial and mesangial cells dysfunction in glomerular capillaries, chronic inflammation and oxidative stress.34,35 These findings strongly nominate homocysteine to be an outstanding marker for transition into diabetic nephropathy stage, and may be valuable in diagnostic and even in therapeutic strategies. Just recently, one study supports such conclusion.13 Consequently and from clinical point of view, serum homocysteine level must be kept under control in patients with diabetic nephropathy, and this can be achieved by administration of vitamins B12, B6 and folic acid supplements i.e. medications can delay the progress of diabetic nephropathy. However, such clinical trials are not in the range of this investigation and needs further assessment.
It is expected and of logic that hyperglycemia is overt in all diabetic groups, and even reached its maximal value in macroalbuminuria. Hyperglycemia was reported to be a modifiable risk factor that promotes the development of diabetic kidney disease in type 2 diabetes.36 In parallel, blood HbA1c was significantly higher in diabetic groups than controls as well as in micro- and macroalbuminuric than normoalbuminuric diabetic patients. Therefore, intensive glycemic control can reduce the risk of the onset of diabetic nephropathy and slows its progression when it has occurred. Serum cholesterol, triglycerides and LDL-C were significantly increased whereas HDL-C was significantly decreased in the three diabetic groups with respect to control group. In addition, HDL-C showed significant lower levels in microalbuminuria and macroalbuminuria than normoalbuminuria. Such findings are in concord with other studies.37,38 It is recently accepted that hyperglycemia is, in part, involved in the determination of the initial step in renal injury, whereas high triglycerides and low HDL-C levels, might be the actual triggers of the onset and progression of diabetic kidney disease.39
Serum homocysteine showed significant positive correlations with urinary albumin, serum urea, creatinine, glucose, HbA1c, and triglycerides whereas a significant negative correlation was found with HDL-C. Such findings are in line with that obtained by other authors.35,40However, few studies have investigated these associations. Alterations of serum homocysteine level throughout transitional changes to diabetic nephropathy and its strong correlations with various metabolic parameters reflect its involvement in the pathophysiology of the disease. The increment of serum homocysteine accompanied with increments of urinary albumin, urea, creatinine and HbA1c, could make homocysteine a novel indicator for transition to diabetic nephropathy. To complete the scenario, further research is recommended to assess serum homocysteine status and its relations to other biochemical parameters in end-stage renal disease or even in hemodialysis patients. In combination with other biomarkers, homocysteine will no doubt open new avenues in diagnosis, management and therapeutic strategies of diabetic kidney diseases.
CONCLUSION
Serum homocystein and urinary albumin have similar trend in being significantly higher in diabetic patients than controls, in micro- and macroalbuminuria than normoalbuminuria, and in macroalbuminuria than microalbuminuria. In general, serum urea, createnine, glucose, HbA1c, cholesterol, triglycerides, LDL-C, were significantly increased in diabetic groups compared to non-diabetics, and reaching their maximum change in macroalbuminuria, whereas HDL-C showed significant decrease and reached its maximum decrease in macroalbuminuria. Serum homocystein showed significant positive correlations with urinary albumin, urea, creatinine, glucose, HbA1c, and triglycerides, whereas with HDL-C it showed a significant negative correlation.
ACKNOWLEDGEMENT
The authors acknowledge the immense help received from the scholars whose articles are cited and included in the references of this manuscript. The authors are also grateful to authors/editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
Conflicts of interest:
There is no conflict of interest
Source of Funding:
No funding has been received for this study.
Authors’ contribution
Prof. Dr. Maged Yassin designed the study, wrote the protocol, helped in the statistical analysis and wrote the first draft of the manuscript. Dr. Said Alghora performed data analysis and revised the final draft of the manuscript. Dr. Mahmoud Alnajjar performed the experimental work. Dr. Mohammed Yasin managed the literature searches.
 ???????
???????
References:
-
World Health Organization. Health topics, Diabetes, Overview; 2020. https://www.who.int/health-topics/diabetes#tab=tab_1. Accessed on 5 April, 2021.
-
Yassin MM, Masoud AD, Yasin MM. Serum vitamin D status in type 2 diabetic patients from Gaza Strip. Diabetes Metab Syndr 2019;13(3):1865-70.
-
Jebril M, Liu X, Shi Z, Mazidi M, Altaher A, Wang Y. Prevalence of type 2 diabetes and its association with added sugar intake in citizens and refugees aged 40 or older in the Gaza Strip, Palestine. Int J Environ Res Public Health 2020;17(22):8594.
-
Selby NM, Taal MW. An updated overview of diabetic nephropathy: Diagnosis, prognosis, treatment goals and latest guidelines. Diabetes Obes Metab 2020;22(Suppl 1):3-15.
-
Yassin MM, AbuMustafa AM, Yassin MM. Serum leptin in diabetic nephropathy male patients from Gaza Strip. Diabetes Metab Syndr 2019;13:1245-50.
-
Nagib AM, Matter YE, Gheith OA, Refaie AF, Othman NF, Al-Otaibi T. Diabetic nephropathy following posttransplant diabetes mellitus. Exp Clin Transplant 2019;17(2):138-46.
-
Martella BM, Veiga GRL, Alves BCA, Azzalis LA, Junqueira VBC, Gehrkei FS, et al. The importance of homocysteine levels in the prognosis of patients with chronic renal disease and in hemodialysis patients. J Bras Patol Med Lab 2018;54(3):170-6.
-
Rujaswini T, Praveen D, Chowdary PR, Aanandhi MV, Shanmugasundaram P. A review on association of serum homocysteine in diabetic neuropathy. Drug Invent Today 2018;10(2):154-6.
-
Chrysant SG, Chrysant GS. The current status of homocysteine as a risk factor for cardiovascular disease: a mini-review. Expert Rev Cardiovasc Ther 2018;16(8):559-65.
-
Haloul M, Vinjamuri SJ, Naquiallah D, Mirza MI, Qureshi M, Hassan C, et al. Hyperhomocysteinemia and low folate and vitamin B12 are associated with vascular dysfunction and impaired nitric oxide sensitivity in morbidly obese patients. Nutrients 2020;12(7):2014.
-
Wang T, Wang Q, Wang Z, Xiao Z, Liu L. Diagnostic value of the combined measurement of serum hcy, serum cys C, and urinary microalbumin in type 2 diabetes mellitus with early complicating diabetic nephropathy. ISRN Endocrinol 2013;2013:407452.
-
Guo J, Ren W, Li X, Xi G, Liu J. Correlation between hyperhomocysteine and serum cystatin C in diabetic nephropathy. Biomed Res 2017;28(11):5153-57.
-
Xu W, Tang S, Xiang M, Peng J. Serum homocysteine, cystatin C as biomarkers for progression of diabetic nephropathy. Pteridines 2019;30:183-8.
-
World Health Organization. Definition and diagnosis of diabetes mellitus and intermediate hyperglycemia Report of a WHO/IDF Consultation. Geneva: WHO; 2006.
-
Palestinian Ministry of Health. Diabetic questionnaire, Diabetic clinic records. Palestine: Gaza Strip; 2006.
-
Backestrom C, Hursh-Cesar G. Survey research. Pennsylvania, United States: Literary Licensing, LLC; 2012.
-
Harmoinen A, Ala-Houhala I, Vuorinen P. Rapid and sensitive immunoassay for albumin determination in urine. Clin Chem Acta 1985;149(2e3):269e74.
-
Refsum H. Clinical Laboratory News May 2002; pp 2-14.
-
Bergmeyer HU. Methods of enzymatic analysis. second ed. New York: Academic Press; 1974. Weinheim: Verlag Chemie.
-
Fabiny DL, Ertingshausen G. Automated reaction-rate method for determination of serum creatinine with the CentrifiChem. Clin Chem 1971;17:696e700.
-
Trinder P. Determination of glucose in blood using glucose oxidase. Ann Clin Biochem 1969;6:24e33.
-
Trivelli LA, Ranney HM, Lai HT. Hemoglobin components in patients with diabetes mellitus. N Engl J Med 1971;284(7):353e7.
-
Meiatlini F, Prencipe L, Bardelli F, Giannini G, Tarli P. The 4-hydroxybenzoate/ 4aminophenazone chromogenic system used in the enzymic determination of serum cholesterol. Clin Chem 1978;24(12):2161e5.
-
Bucolo G, David H. Quantitative determination of serum triglycerides by use of enzymes. Clin Chem 1973;19:476e82.
-
Grove TH. Effect of reagent pH on determination of HDL cholesterol by precipitation with sodium phosphotungstate-magnesium. Clin Chem 1979;25:560e4.
-
Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of LDL-C in plasma without use of the preparative ultracentrifuge. Clin Chem 1972;18:499e502.
-
Steele CJ, Schöttker B, Marshall AH, Kouvonen A, O'Doherty MG, Mons U, et al. Education achievement and type 2 diabetes-what mediates the relationship in older adults? Data from the ESTHER study: a population-based cohort study. BMJ Open 2017;7(4):e013569.
-
Hsu CC, Lee CH, Wahlqvist ML, Huang HL, Chang HY, Chen L, et al. Poverty increases type 2 diabetes incidence and inequality of care despite universal health coverage. Diabetes Care 2012;35(11):2286e92.
-
Yassin MM, Abu Mustafa AM, Abujami SM, Jabber EA. Leptin status and biochemical parameters in type 2 diabetic males from Gaza strip. AMBS 2017;3(1): 4-10.
-
Parkkola A, Turtinen M, Härkönen T, Ilonen J, Knip M; Finnish Pediatric Diabetes Register. Family history of type 2 diabetes and characteristics of children with newly diagnosed type 1 diabetes. Diabetologia 2021;64(3):581-90.
-
Amor A, Jimenez A, Moize V, Ibarzabal A, Flores L, Lacy AM, et al. Weight loss independently predicts urinary albumin excretion normalization in morbidly obese type 2 diabetic patients undergoing bariatric surgery. Surg Endosc 2013;27(6):2046-51.
-
Raja P, Maxwell AP, Brazil DP. The potential of albuminuria as a biomarker of diabetic complications. Cardiovasc Drugs Ther 2020. https://doi.org/10.1007/s10557-020-07035-4.
-
Abu Mustafa AM, Yassin MM. Clinical and biochemical aspects associated with diabetic nephropathy among type 2 diabetic males in Gaza strip. Int J Med 2017;5(1):142-8.
-
Yassin MM, Lubbad AH, Abu Taha AJ, Saadallah NM. Homocysteine and hematological indices in hemodialysis patients. Ibnosina J Med BS 2014;6(4):173-9.
-
Singla H, Panag K, Batta A, Goyal G. Homocysteine levels in patients of type 2 diabetes mellitus with diabetic nephropathy and its clinical implications. Int J Med Res Rev 2015;3(9):1070-6.
-
Macisaac RJ, Ekinci EI, Jerums G. Markers of and risk factors for the development and progression of diabetic kidney disease. Am J Kidney Dis 2014;63(2 Suppl 2): S39-62.
-
Yassin MM, Altibi HI, El Shanti AF. Clinical and biochemical features of type 2 diabetic patients in Gaza Governorate, Gaza Strip. West Afr J Med 2011;30(1):51-6.
-
Rai S, Prajna K, Rai T. Lipid profile in type 2 diabetes mellitus and in diabetic nephropathy. Int J Clin Biochem Res 2017;4(4):379-82.
-
Russo G, Piscitelli P, Giandalia A, Viazzi F, Pontremoli R, Fioretto P, et al. Atherogenic dyslipidemia and diabetic nephropathy. J Nephrol 2020;33(5):1001-8.
-
Ma N, Xu N, Yin D, Liu W, Wu M, Cheng X. Relationship between plasma total homocysteine and the severity of renal function in Chinese patients with type 2 diabetes mellitus aged ≥75 years. Medicine (Baltimore) 2020;99(27):e20737.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License