IJCRR - 7(20), October, 2015
Pages: 30-36
Date of Publication: 20-Oct-2015
Print Article
Download XML Download PDF
HEALTH EDUCATION REGARDING HUMAN PAPILLOMA VIRUS (HPV VACCINE) FOR PRIMARY PREVENTION OF CERVICAL CANCER FOR PARENTS OF ADOLESCENT GIRLS
Author: Paapa Dasari, Haritha Sagili, Subitha Lakshminarayan
Category: Healthcare
Abstract:Objective: To assess the existing knowledge regarding causation and prevention of carcinoma cervix and to increase their awareness though a health education program addressing prevention especially by HPV vaccination among teachers and parents of school children. Material and Methods: This was a cross sectional interventional study conducted in St. Joseph's of Cluny School, in Puducherry. Health education regarding carcinoma cervix and its prevention was given by an illustrated lecture using powerpoint presentation and Question answer sessions. Pretest and post test questionnaire in Tamil (Local Language) and English was used. Study population was 325 mothers including school teachers. Statistical Analysis: SPSS 16 was used. All categorical variables were presented as percentages and the pre-test and post test questionnaire were compared by using Chi square test. Results: Knowledge of causation by HPV virus improved from 50% to 98% on post test (p< 0.001). There was a significant improvement (80%) regarding dosage, side effects of the vaccine and 75% improvement regarding age of administration of vaccine. There was a significant improvement in knowledge regarding efficacy of the vaccine, place of availability and cost of HPV vaccine. Acceptability if the vaccine is provided through the schools was about 92% after the health education programme.
Conclusion: Though 50% were aware about the disease and its causation the knowledge regarding its prevention by HPV vaccination and its availability was poor at baseline. After the health education programme there was a statistically significant improvement in all aspects.
Keywords: Carcinoma Cervix, Primary prevention, HPV Vaccine, Health education, Parents of adolescent girls
Full Text:
INTRODUCTION
Cervical cancer is the most common cancer among women in India and is also the most common cuase of mortality when deaths due to cancer are considered. Approximately 510,000 new cases are diagnosed and 288,000 deaths are registered per year World wide. In India, 132,000 new cases were diagnosed and 74,000 deaths were accounted due to carcinoma cervix1 . The cause for carcinoma cervix is well established and it is proven beyond doubt that that Human Papilloma Virus (HPV) is the causative agent in more than 95% of cases. There is long latent period as long as 20 years between the infection with pathogenic HPV and the development of cancer. Primary prevention of cervical cancer is being undertaken in developed World by using Quadrivalent or bivalent vaccine2 . Australia was the first country since 2007 to provide free vaccination to the school going children and now more than 110 countries are implementing HPV vaccination . In India it is not yet incorporated in to child or adult national immunization schedule though it was approved by FDA as early as 20061,3 and is available in India. The public aware ness about this modality of prevention is lacking although Indian Academy of Pediatrics Committee on Immunisation (IAPCOI) recommends offering HPV vaccine to all female children who can afford the vaccine (Category 2 of Indian Academy of Paediatrics categorisation of vaccines). In this context, this study aimed to assess the existing knowledge regarding causation and prevention of carcinoma cervix and to increase their awareness and clear misconceptions though a health education programme addressing prevention especially by HPV vaccination. The target population was school teachers and parents of adolescent school girls.
MATERIAL AND METHODS
This was a cross sectional interventional study conducted in one of the best girl’s schools ( St. Joseph’s of Cluny) in Pondicherrycherry after interacting with the principal and teachers. The summary of the importance of the programme was mailed to the Principal. The project was approved by our Institutional Scientific Committee and Ethics committee ( JIP/IEC/SC/2013/3/398). The programme was conducted in 2 sessions after the school hours. The school authorities informed to the parents of students class 6th to 10th to attend and participate voluntarily in the health education programme. However only mother’s of the adolescent girls attended the programme. Though more than 500 mothers attended the programme, 325 were included in the study as per the sample requirement. The sample size was calculated assuming 50% would have knowledge. With 80% power , 5% alpha error and one sided hypothesis testing the sample size was estimated to be 305. All subjects were approached personally by the investigators and oral informed consent was taken for inclusion into the study. They were told that the programme is being conducted to find out the awareness regarding cervical cancer and its prevention among them and information would be given about cause and prevention of the same by an illustrative lecture in English and Tamil and there will be a question and answer session at the end of the session They were also assured that the identity and the information provided by them would be kept confidential. A self administered questionnaire (In English and local language Tamil) was given to all participants (Pre-test) who gave informed consent for taking part in the study.(Annexure-1). An illustrative Lecture on prevention of carcinoma cervix and HPV vaccine administration and side effects was delivered by the principal investigator. Any doubts/queries were cleared to the participant by the investigator and co- investigators. Post test was conducted at the end of the programme. After the post-test activity was over, they were also given a had-out containing information regarding HPV vaccine .
Statistical Analysis
Data was processed by means of SPSS 16 and presented as proportions. All categorical variables like knowledge regarding Carcinoma cervix , HPV Vaccine were presented as frequencies or percentages The pretest and post test scores were compared by using Chi square test. Differences in the proportions along with 95% confidence intervals is expressed .All statistical analysis was carried out with 5 % level of significance and p value of <0.05 will be considered as significant.
RESULTS
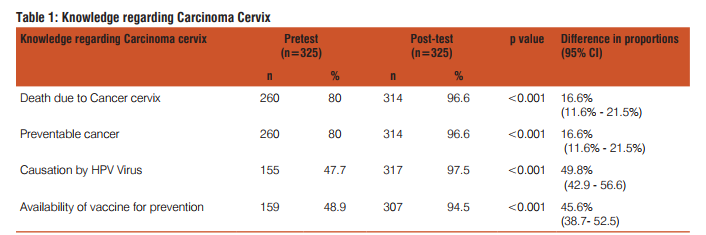
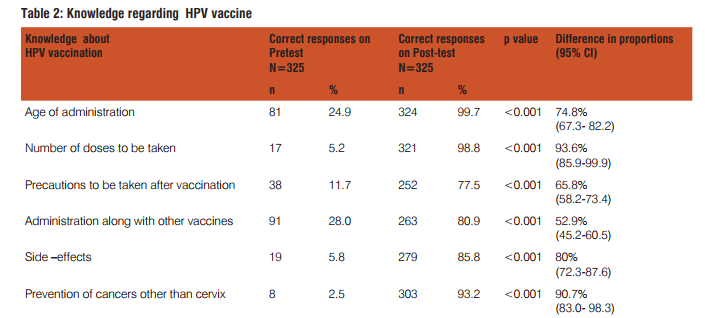
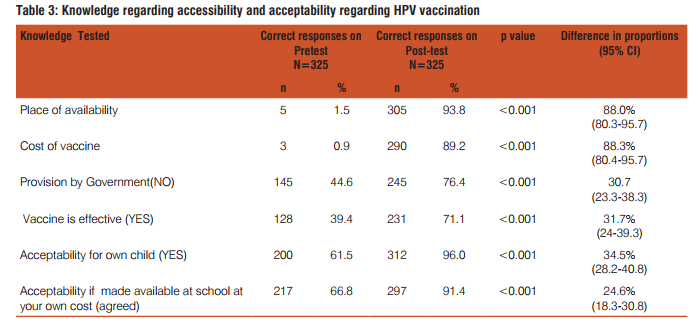
Table 1 shows the awareness of the disease causation and its prevention. Three hundred and twenty five people participated in the study Eighty percent of them know about the disease can cause death and it is preventable. But only 50% knew the causative agent and the vaccine availability to prevent it. After the lecture (health education programme) this proportion increased to 97%. Similarly correct knowledge of causation by HPV virus improved from 50% to 98% on post test (p<0.001). Table 2 shows the knowledge regarding vaccine administration. In pre-test, 20% were aware about the age of administration of vaccine; seven percent knew about the number of doses. Twelve percent had awareness regarding the precautions to be taken when vaccine is administered. Twenty percent responded that it can be administered along with other vaccines and 40% felt that it is very effective. Six percent had correct knowledge regarding side effects and only 2.5 % answered correctly about other advantages of vaccine. After the lecture (post-test) almost all (99.7%) were clear regarding the age of administration of HPV vaccine and 99% were sure of the number of doses and frequency. Ninety three percent came to know about the other advantages of preventing all HPV related cancers. This is as against the pre-test awareness of 2.5%. Eighty one percent were aware that it can safely be administered along with other vaccines and 77% learnt the precautions to be taken when HPV vaccine is administered. Their confidence levels increased by 80% regarding side-affects. Knowledge regarding effectiveness of vaccine is increased by 32%. Knowledge of vaccine and vaccination improved significantly after the health education session. The improvement in knowledge varied from 52% in case of co-administration of other vaccines to a maximum of 94% regarding the number of doses to be administered. There was a significant improvement (80%) side effects of the vaccine and 75% improvement regarding age of administration of vaccine. Knowledge regarding Vaccine availability, acceptability and Cost is shown in Table 3 There was a significant improve-embolism, appendicitis, seizures, syncope, allergic reactions, and anaphylaxis13. A systemic review and metaanalysis on the safety and efficacy of HPV vaccine showed that the vaccine is highly effective in the prevention of HPV associated pre-cancerous lesions with minimal side –effects14 The rationale of efforts to increase the awareness regarding HPV vaccination is further supported by the evidence of increasing HPV etiology among other cancers like oropharengeal, anal and penile. It is reported that approximately 15,000 HPV 16 and 18 cancers occur in women 7,000 HPV 16 and 18 associated cancers occur in men in United states and hence the ACIP recommends HPV vaccination of male children beginning at 9 years and also adults up to 26 years15 The above facts need to be put forward to the policy makers in the department of Health and Family Welfare and also to the Public through the Health Education Programmes especially through NRHM (National Rural Healyh Mission). Once upon a time it was made aware through health education that cancer was curable only if detected at early stage but know we should make the Public aware that it is preventable as well, like Tuberculosis by Primary vaccination. School based vaccination programmes are successful in coverage of all three doses of vaccination and in UK the implementation of HPV vaccination was initiated in Schools16. Parental support/ Mother’s support in school based HPV vaccination is encouraging as most of the mothers have supported such a programme17,18. In the present study also 91% supported school based HPV vaccination. Hence a school based health education pregramme for adolescents and their parents to increase the awareness regarding prevention of carcinoma cervix and other HPV related cancers incorporating HPV Vaccine provision is the need of the hour. WHO position paper on HPV vaccines recommended its introduction in to routine national immunisation schedules as a public health priority19.A systematic review which assessed the knowledge, awareness and acceptability of HPV vaccine in Sub Saharan Africa concluded that there was a high acceptability of the vaccine and low levels of knowledge and awareness. The review identified six countries which met the criteria for GAVI ( Global Alliance for Vaccines and Immunizations) alliance for financial support of the HPV vaccination programme and they have sucessfully introduced free HPV vaccination programme in their country20. The 2 criteria were 1. A DTP3 threshold of 70% national coverage (WHO/UNICEFestimates) and 2. a pilot demonstration of the ability to deliver a complete multi-dose series of vaccines to at least 50% of the target vaccination cohort in an average sized district in a country. India also should implement and provide low cost HPV Vaccination especially to low socioeconomic population as the incidence and mortality of of carcinoma cervix is high1 . Many states in India would meet the eligibility criteria put forward by GAVI alliance.
CONCLUSION
Though 50% of the study populations were aware of the causation of carcinoma cervix and its prevention, the knowledge regarding the HPV vaccine availability, administration and side effects is poor. After the health education programme there is a great improvement in their knowledge in all aspects especially HPV vaccine administration, availability and acceptability. It is very encouraging that more than 90% are willing to vaccinate their children if the vaccine is provided at Schools.
ACKNOWLEDGEMENTS
We would like to express our gratitude to the Principal and staff of St. Joseph of Cluny School, Pondicherry for having given permission to conduct the study at their school, for the arrangements, for audiovisuals to be displayed and for gathering and welcoming all the Mothers of their adolescent school girls. Our thanks are to Mr. Karunakaran, Social worker, department of Obstetrics and Gynaecology, JIPMER for Tamil translation of Questionnaire and also for assissting to conduct the health education programme. We also express our thanks to our postgraduate students Dr. Priyanka Yoga and Dr. Deepthi for helping to distribute and recollect the pre-test and post test forms. We acknowledge the immense help received from the scholars whose articles are cited and included in the references of this manuscript. The authors are also greatful to authors/ editors/publishers of all those articles , journals , books from where the literature for this article has been reviwed and discussed.
ANNEXURE !
QUESTIONNAIRE
1. Are you aware that many women die due to cancer cervix and it is a preventable cancer ? Yes/ No
2. Do you know that cancer cervix is caused by a virus? Yes/ No
3. Are you aware that a vaccine called HPV vaccine is available in India as a preventive measure for cervical cancer? Yes/ No
2. At what age this vaccine is to be given?
3. How many doses need to be taken?/how many times one needs to take?
4. Does it cause any harm? Or serious side effect? YES/NO
5. Any precautions one should take after vaccination?
6. Can be it be given along with all other vaccines? Yes/ No
7. Does it prevent any other cancers or only cancer cervix?
8. Is the Govt providing this vaccine like Polio and DPT etc? Yes/No
9. Do you know where the vaccine is available and how much it costs?
10. Do you think if all the doses of vaccine were taken correctly one does not get cancer cervix in their lifetime/ Yes/ No
11. Would you like your child/your student to receive this vaccine? If yes------ Write why do you think so— If no-------- please write your fears /doubts
12. If the vaccine is made available in school would you like your child to receive it at your own cost? Yes/ No.
Thank you for your participation and the information you have given will be kept confidential and will be anonymised in publication
References:
1. Human Papilloma Virus and related cancers, India. WHO/ICO ,3 red edidtion. Summary report Update June 22,2010.
2. Morbidity Mortality weekly report. Advisory committee on Immunization Practices (ACIP) recommended inmmunisation schedule for persons aged 0 through 18 years and adults aged 19 years and older. United States.2013.www,cdc.gov/mmwr/ preview/mmwrhtml/mm63e0128a1.htm.
3. Food and Drug Administration. Product approval-prescribing information [package insert]. Gardasil [human papillomavirus quadrivalent (types 6, 11, 16, and 18) vaccine, recombinant], Merck and Co, Inc: Food and Drug Administration 2009. Available at http://www.fda.gov/biologicsbloodvaccines/vaccines/approvedproducts/ucm094042.htm. Accessed May 25, 2010.
4. Barry D, Increasing knowledge about HPV and HPV Vaccine amongest adolescents and adults through a school based setting:A Capstone Project.2013. http://scholarworks.umass.edu/nursing_dnp_capstone/31
5. Pandey D. Vanya V, Bhagat S, Binu VS, Shetty J. Awareness and attitude towards Human papilloma Virus (HPV) vaccine among medical students in a Premier Medical School in India. PLoS ONE 7(7): e40619. doi:10.1371/journal.pone.0040619
6. Allen JD, Othus MKD, Shelton RC, Norman N, Tom L, del Carmen MG.Parental decision making about the HPV vaccine. Cancer Epidemiol Biomarkers Prev; 2010; 19(9) : 2187–98.
7. Bair, R.M., Mays, R.M., Sturm, L.A., and Zimet, G.D. Acceptability of the human papillomavirus vaccine among Latina mothers. Journal of Pediatric and AdolescentGynecology, 2008 ;21(6): 329-334. doi:10.1016/j.jpag.2008.02.007
8. Diwaker H. Knowledge and awareness about health seeking behaviour and acceptability of cervical cancer vaccine in Urban women in comparision with school students. J South Asian Feder Obstet Gynae. 2012;4 :47-53.
9. Do kyung Young and Wong Yi Ker.Awareness and accepatability of human papilloma virus vaccine: an application of the instrumental variables bivariate prohibit model.BMC Public health 2012;12:31
10. Bernard, D.M., Cooper, S.C., McCaffery, K.J., Scott, C.M., Skinner S.R. The domino effect: Adolescent girls’ response to human papillomavirus vaccination. Medical Journal of Australia 2011;194(6):297-300. Retrieved from https://www.mja.com.au/ journal/2011/194.
11. Kennedy, A., Sapsis, K., Stokley, S., Curtis, C.R., and Gust, D. Parental attitudes toward human papillomavirus vaccination: Evaluations of an educational intervention, 2008. Journal of Health Communication, 2011;16, 300-313. doi:10.1080/10810 730.2010.532296
12. Kjaer SK, Sigurdsson K, Iversen OE, et al. A pooled analysis of continued prophylactic efficacy of quadrivalent human papillomavirus (types 6/11/16/18) vaccine against high-grade cervical and external genital lesions. Cancer Prev Res (Phila) 2009;2:868–78
13. Gee J, Naleway A, Shui I, et al. Monitoring the safety of quadrivalent human papillomavirus vaccine: Findings from the Vaccine Safety Datalink. Vaccine 2011;29;8279–84
14. Lu, B., Kimar, A., Castellsague, X., and Giuliano, A.R. . Efficacy and safety of prophylacticvaccines against cervical HPV infection and disease among women: A systematic reviewand metaanalysis. BioMed Central Infectious Disease,2014 11(13), 1-16. doi:10.1186/1471-2334-11-13
15. CDC. FDA licensure of quadrivalent human papillomavirus vaccine (HPV4, Gardasil) for use in males and guidance from the Advisory Committee on Immunization Practices (ACIP). MMWR 2010;59:630–2.
16. Sheridan A. and White, J.. Annual HPV vaccine coverage in England in2009/2010 [pdf]. .http://data.parliament.uk/DepositedPapers/Files/DEP2012-1386/PQ119371-2.pdf
17. Kadis, J.A. McRee, A., Gottlieb, S.L., Lee, M.R., Reiter, P.L., Dittus, P.J., Brewer, N.T.. Mothers’ support for voluntary provision of HPV vaccine in schools. Vaccine, 2011 29,2542-2547. doi:10.1016/j.vaccine.2011.01.067 .
18. Kelminson, K., Saville, A., Seewald, L., Stokley, S., Dickinson, L.M., Daley, M.F., Kempe, A. Parental views of school-located delivery of adolescent vaccines. Journal ofAdolescent Health, 2012; 51, 190-196. doi:10.1016/j.jadohealth.2011.11.016
19. WHO Human papillomavirus vaccines. WHO position paper. 2009;118–131
20. Pealman S et al. Knowledge and Awareness of HPV Vaccine and acceptability to vaccinate in Sub-Saharan Africa: A systematic Review. PLoS ONE 9(3): e90912. doi:10.1371/journal. pone.0090912
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License