IJCRR - 13(15), August, 2021
Pages: 133-138
Date of Publication: 10-Aug-2021
Print Article
Download XML Download PDF
Evaluation of the Antibacterial Activity of the Leave Extract of Oroxylum Indicum Against Multidrug-Resistant Bacteria Causing Urinary Tract Infection
Author: Debasmita Dubey, Mahesh Chandra Sahu, Pradeep Kumar Naik
Category: Healthcare
Abstract:Aim: Evaluation of antibacterial efficacy of the methanolic leaf extract of Oroxylum indicum against isolated MDR strains viz. Acinetobacter sp., Citrobacter sp., Enterobacter sp., Escherichia coli, Klebsiella pneumonia, Proteus Vulgaris, Proteus mirabilis, Pseudomonas aeruginosa, Staphylococcus aureus, and Enterococcus fecalis causing urinary tract infections (UTI). Methods: Pathogenic bacteria were isolated and identified using the routine microbiological procedure. All the bacteria were MDR as determined from the antibiotic sensitivity test. Agar well diffusion and microbroth dilution methods were used for determining antibacterial efficacy and the minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) was of O. indicums methanolic leaf extract were determined. Cord blood lymphocytes. were used to determine, the in vitro toxicity of the leaf extracts. Results: The leaf extract of O. indicum exhibited good antibacterial activity against both Gram-positive and Gram-negative MDR bacteria. The MIC value of the plant extract ranges from 0.19 to 0.78 mg/ml and the MBC value ranges from 0.78 to 1.56 mg/ ml with all the isolated bacterial pathogens. Plant extracts were found to be very effective against these MDR strains. The plant extract also revealed no toxicity even at a very high concentration of 1800 mg/l with lymphocytes isolated from the cord blood. Surprisingly, the MIC value was found to be 600 mg/l with lymphocytes. Conclusion: O. indiucm's methanolic leaf extract can be used as a potential antibacterial drug. Similarly, other medicinal plants can also be explored for their possible antibacterial efficacy over the antibiotic resistance problem.
Keywords: UTI (Urinary Tract infection), O. indicum, Antibacterial Activity, Toxicity Study, Alternative Medicine
Full Text:
Introduction
Urinary tract infections (UTI) caused by both “Gram-negative (GN) and Gram-positive (GP) pathogenic bacteria increase the rate of morbidity and mortality throughout the globe. The first antibiotic, penicillin, or 6-aminopenicillins acid (6-APA) controls bacterial infections, however, certain bacteria have developed resistance against it.1,2 Further, pathogenic bacteria have been shown to develop resistance to newly introduced antibiotics.2-4 Thus, multidrug-resistant (MDR)bacteria must be controlled with an iron hand, which has been giving impetus to search for newer antibacterial from conventional and non-conventional sources, apart from a chemical modification of existing antibiotics. Several phytochemicals were reported to have antibacterial and antifungal activities and lend themselves to the preparation of mainstream medicine.5,6
Oroxylumindicum(L.) Vent. is commonly known as shyonaka or sonpatha, is a small to medium-sized deciduous tree of the family Bignoniaceae. It is distributed throughout India, upto an altitude of 1200 M and found mainly in the ravine and moist places in the forests and also in Himalayan foothills, Eastern and the Western Ghats, and North-East India.7,8 Different plant parts of O. indicumare being used in Ayurveda and traditional medicine for the treatment of different ailments such as fevers, cough, cancer, diarrhoea, ulcer, dropsy, jaundice and preventing other respiratory disorders.9 Root extract has been used for ayurvedic preparations like Dashmularisht and Chyawanprash.10 It is also one of the important ingredients in an ayurvedic formulation such as Amartarista, Dantyadyarista, Narayana taila, Dhanawantaraghrita, Brahma Rasayana and Awalwha.11 It possesses antioxidant, antifungal, antimicrobial, anti-inflammatory, antibacterial, anti-arthritic and anti-cancer properties.12 The leaves and stem bark are reported to contain flavonoids namely chrysin, oroxylin-A, scutellarin, baicalein.13,14 Root bark is reported to contain chrysin, baicalein, biochanin-A, and ellagic acid.15 Seeds are reported to use in the perfume industry.9 Baicalein is reported to possess an anti-inflammatory, anti-ulcer, antioxidant, hepatoprotective, and immunomodulatory activity.16-20. Baicalein is also reported to check the proliferation of human breast cancer cell line MDA-MB-435.16-21
In the present study, we have attempted to investigate the antibacterial activity of the leaf extract of O. indicum against both the GP and GN MDR strains of bacteria isolated from the clinical samples of patients suffering from UTI.
Materials and methods
Collection, processing, storage and solvent extract preparation
Leaf of O. indicium was collected from Gandhamardhan, Western Odisha. The plant species were identified with the help of regional flora books (Haines, 1921-25, Saxenaand Brahman, 1994-96). Voucher specimens are deposited in the herbarium of Centre of Excellence in Natural Products and Therapeutics, Dept. of Biotechnology and Bioinformatics, Sambalpur University, Odisha, India. The collected leaf samples were processed and solvent extracts were made and stored as described previously.4
Bacterial Strain isolation, identification and antibiogram
Two GPs, Enterococcus faecalis, and Staphylococcus aureus, as well as 8 GNs viz. Acinetobacter sp, Citrobacter sp, Enterobacter sp, Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis, Proteus vulgaris, and Pseudomonasaeruginosawere isolated from the patients with UTI from a private hospital, Odisha, India. The bacteria were identified based on the conventional colony and biochemical characteristics. Further, Kirby-Bauer’s disk diffusion method was utilized to evaluate the antibiotic sensitivity of the isolated bacteria.22, 23
Evaluation of antibacterial activity and determinations of MIC and MBC of plant
Agarwell diffusion method was used for determining the antibacterial efficacy of the methanolic leaf extract against the isolated UTI MDR bacterial strains.24Further, the microbroth dilution method was determining the MICand MBC values of the methanolic leaf extract, following the protocol described previously.24
Phytochemicals screening
Phytochemical screening tests of the methanolic leaf extract was done according to the protocol described earlier. 24
Evaluation of toxicity of plant extract with lymphocytes
Toxicity of the methanolic leaf extract of O. indicum was evaluated based on in vitro lymphocyte culture isolated from umbilical cord blood as described before. The proliferation oflymphocytes in the presence of graded concentrations of the plant extract was monitored by MTT assay as reported earlier.4The toxicity of plant extracts to isolated lymphocytes was also studied by comet assay as reported earlier.4
Statistical Analysis
The observed percentage of the lethality of the plant extract was determined based on probit analysis.
Results
Antibiotic susceptibility screening of the isolated bacterial strains
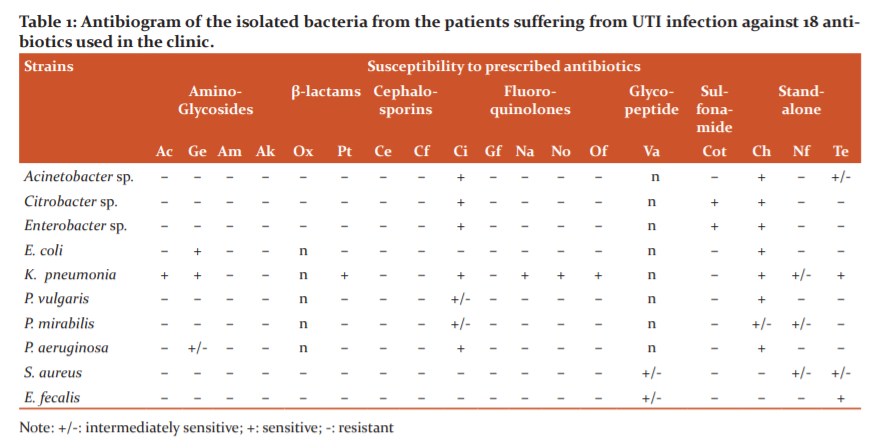
All the isolated bacteria were screened with 18 antibiotics from 6 different groups. The antibiogram of the isolated bacteria is collated in (Table 1). It was found that all the bacterial strains have shown resistance to most of the antibiotics.
Antibacterial activity of the plant extract
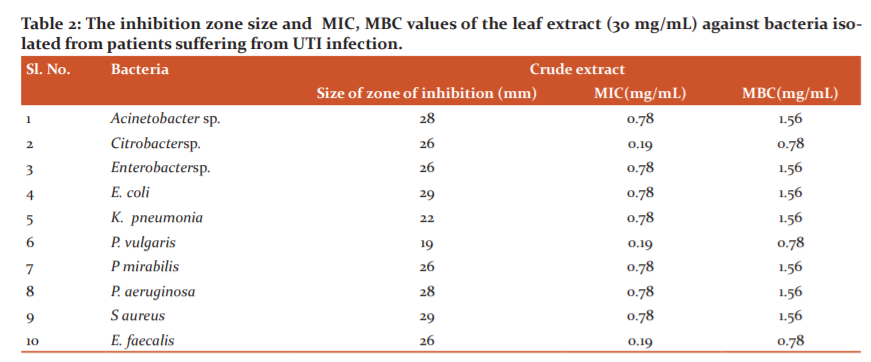
The results of the antibacterial activity of the methanolic leaf extract of O. indicum against isolated MDR strains viz. Acinetobacter sp., Citrobacter sp., Enterobacter sp., E. coli, K. pneumonia, P. vulgaris, P. mirabilis, P. aeruginosa, S. aureus and enterococcus faecalis from patients suffering from UTI infection are presented in (Table 2). The plant extract could effectively inhibit the growth of the bacteria as indicated by the zone of inhibition. The methanolic leaf extract was used for qualitative evaluation of the composition of various secondary metabolites using traditional biochemical characterization.
Toxicity evaluation of the plant extract
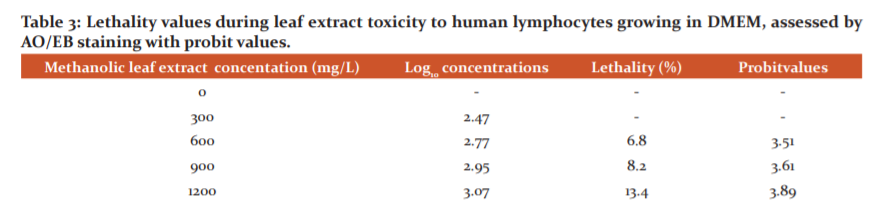
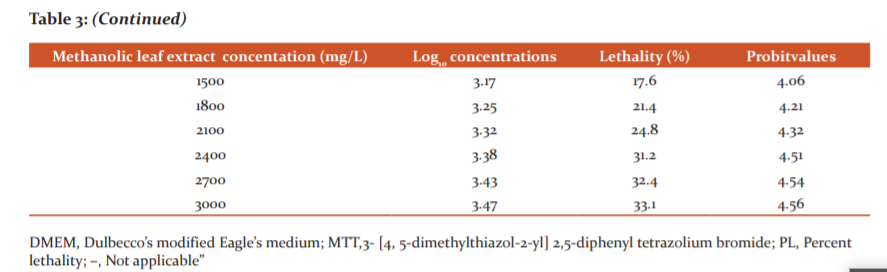
We have used human cord blood lymphocyte culture to evaluate the toxicity of the crude leaf extract. No evidence of toxicity was found even at the concentration of 1800 mg/l leaf extract using comet assay (Figure 1). The experimental MIC value of the crude extract was determined to be 600 mg/l and the computed LC25 value was found to be 1698.24 mg/l during cytotoxicity (Figure 2, Table 3).
Discussion
UTI is considered one of the major problems for hospitalized patients. It is Aside from the advancement of UTI from the fecal issue, it is more promptly in females in comparison to males.25,26The bacterial pathogens involved in UTI are mostly Klebsiella, E. coli, Pseudomonas and Enterobacter. Additionally,Candida sp. is also widely recognized as a pathogen that causes UTIs.27,28 In quest of finding bacterial pathogens from the patients suffering from UTI, we have identified 2 GP bacteria, S. aureus and Enterococcus faecalis as well as 8 GN bacteria viz. Acinetobacter sp., Citrobacter sp., Enterobactersp., E coli, K. pneumonia, P. vulgaris, and P mirabilis. Most of these isolated bacterial strains revealed resistance to most of the antibiotics. These isolated bacterial strains were screened for their susceptibility against the methanolic leaf extract of O. indicum, which revealed good antibacterial activity against all the isolated bacterial strains. The MIC value ranges from 0.19 to 0.78 mg/ml and MBC value ranges from 0.78 to 1.56 mg/ml for the crude extract. The antibacterial activity of O. indicum fruit extracts was successfully proven previously against gram-positive bacteria.29Another Indian study also proved the antibacterial efficacy of the stem bark and root extracts against both gram-positive, gram-negative bacteria and pathogenic Candida species.30Similarly, Talari et al, 2013, reported the antibacterial activity of stem bark extracts against clinically important Gram-positive bacteria.31Further, Singh et al., 2002 in his study reported the antibacterial and antifungal efficacy of ethanolic extracts of various Himalayan medicinal plants.32Toxicity evaluation of the methanolic leaf extract of O. indicum failed to reveal any toxicity using lymphocytes isolated from cord blood even at a very high concentration of 1800 mg/l. Parenthetically the MIC value was found to be 600 mg/l with lymphocyte which revealed no severe toxicity. The antibacterial activity of O. Indicum is mainly because of the presence of various phytochemicals in its extract. Bark extract showed the presence of various secondary metabolites in the alcoholic extracts of bark of O. indicum.33Similarly, many other studies revealed plants contains secondary metabolites and many other trace elements, which can be attributed to their antibacterial and other therapeutic efficacy along with a possible treatment option for Covid -19 virus too.34-36
Conclusions
In conclusion, UTI in patients was caused by MDR bacteria, hence cannot be controlled by antibiotics. Methanolic leaf extract of O. indicum proved to be a potent crude drug in this study as it was able to inhibit the growth of all the MDR bacteria. Crude extract failed to reveal any toxicity at very high concentrations. Therefore, it is can be concluded O. indiucm’s methanolic leaf extract can be further used as a potential antibacterial drug. Similarly, other medicinal plants can also be explored for their possible antibacterial efficacy over the antibiotic resistance problem.
Acknowledgements: The authors acknowledge the authors of the article cited in this article.
Funding: The authors would like to thank OHEPEE, Govt. of Odisha for providing financial support through the World Bank under the Centre of Excellence in Natural Products and Therapeutics, Sambalpur University. We are grateful to IMS & Sum Hospital for providing the clinical samples.
Conflicts of Interest: None
Authors Contribution: D Dubeyand MC Sahu conducted the experiments, collected the data and drafted the manuscript. PK Naik is finalized the data and manuscript.
Ethical Issues: This paper includes no human or animal subjects, hence no ethical approval is required.
References:
-
Chambers HF, Deleo FR. Waves of resistance: Staphylococcus aureus in the antibiotic era. Nat Rev Microbiol. 2009;7(9):629-41.
-
Cantón R, Morosini MI. Emergence and spread of antibiotic resistance following exposure to antibiotics. FEMS Microbiol Rev. 2011;35(5):977-91.
-
Davies J, Davies D. Origins and evolution of antibiotic resistance. Microbiol Mol Biol Rev. 2010;74(3):417-433.
-
Sahu MC, Patnaik R, Padhy RN.In vitro combinational efficacy of ceftriaxone and leaf extract of Combretum albidum G. Don against multidrug-resistant Pseudomonas aeruginosa and host-toxicity testing with lymphocytes from human cord blood. J Acute Med. 2014; 4: 26–37.
-
Cowan MM. Plant products as antimicrobial agents, Clin MicrobiolRev 1999; 12: 564–582.
-
Vázquez-Sánchez D, Cabo ML, Rodríguez-Herrera JJ. Antimicrobial activity of essential oils against Staphylococcus aureus biofilms. Food Sci Technol Int. 2015 Dec;21(8):559-70.
-
Radhika LG, Meena CV, Peter S, Rajesh KS, Rosamma MP. Phytochemical and antimicrobial study of Oroxylum Indicum, Anc. Sci Life. 2011; 30: 114–120.
-
Jayaram K, Prasad MN. Genetic diversity in Oroxylum Indicum(L.) Vent. (Bignoniaceae), a vulnerable medicinal plant by random amplified polymorphic DNA marker, Africa. J Biotech. 2008; 7: 254–262.
-
Harminder VS, Chaudhary AK. A Review on the Taxonomy, Ethnobotany, Chemistry and Pharmacology of Oroxylum Indicum Vent, Indian J Pharm Sci. 2011; 73: 483–490.
-
Yasodha R, Ghosh M, Santan B, Gurumurthi K, Importance of biotechnological research in tree species of Dashmula. Indian Fore. 2004; 130: 79–88.
-
Dinda D, SilSarma I, Dinda, Rudrapaul MP.Oroxylum indicum (L.) Kurz, an important Asian traditional medicine: from traditional uses to scientific data for its commercial exploitation. J Ethnopharm 2015; 161:255–278.
-
Deka DC, Kumar V, Prasad C, Kumar K, Gogoi BJ, Singh L, Srivastava RB. Oroxylum Indicum –a medicinal plant of North East India: An overview of its nutritional, remedial, and prophylactic properties. J App Pharm Sci. 2013; 3 (S1): 104-112.
-
Sankara S, Nair AG, Flavanoid of stem bark of Oroxylum Indicum.Curr Sci. 1972; 41: 62–63.
-
Chen LJ, Song H, Lan XQ, Games DE, Sutherland IA. Comparison of high-speed counter-current chromatography instruments for the separation of the extracts of the seeds of Oroxylum Indicum. J Chromatograph A. 2005;1063(1-2):241-245
-
Zaveri M, Khandhar A, Jain S. Quantification of Baicalein, Chrysin, Biochanin-A and Ellagic Acid in Root Bark of Oroxylum Indicum by RP-HPLC with UV Detection. Eurasian J Analytical Chem. 2008;3(2):245-257.
-
Zhong X, Surh YJ, Do SG, Shin E, Shim KS, Lee CK, Na HK. Baicalein Inhibits Dextran Sulfate Sodium-induced Mouse Colitis. J Cancer Prev. 2019;24(2):129-138.
-
Khennouf S, Benabdallah H, Gharzouli K, Amira S, Ito H, Kim TH, Yoshida T, Gharzouli A. Effect of tannins from Quercus suber and Quercus coccifera leaves on ethanol-induced gastric lesions in mice. J Agric Food Chem. 2003 Feb 26;51(5):1469-73.
-
Ng TB, Liu F, Wang ZT. Antioxidative activity of natural products from plants.Life Sci. 2000; 66: 709–723.
-
Niedworok J, Jankowstia B, Kowalczy E, Okroj W. A comparative investigation of hepatoprotective effects of baicalein and sylimarol.Herb Pol. 1999; 45:199–205.
-
Chiang LC, Ng LT, Chiang W, Chang MY, Lin CC, Immunomodulatory activities of flavonoids, monoterpenoids, triterpenoids, iridoid glycosides and phenolic compounds of Plantago species.Planta Med. 2003; 69: 600–604.
-
Lambertini E, Piva R, Khan MT, Lampronti I, Bianchi N, Borgatti M, Gambari R. Effects of extracts from Bangladeshi medicinal plants on in vitro proliferation of human breast cancer cell lines and expression of estrogen receptor alpha gene. Int J Oncol. 2004 Feb;24(2):419-23.
-
CLSI, Performance standard for antimicrobial susceptibility testing: twenty-first informational supplement. Wayne (PA): Clinical and Laboratory Standards Institute, 2019.
-
Sahu MC, Swain SK. Surveillance of antibiotic sensitivity pattern in chronic suppurative otitis media of an Indian teaching hospital. World J Otorhin Head Neck Surg. 2019; 5: 88–94.
-
Sahu MC, Debata NK, Padhy RN. Antibacterial activity of Argemone mexicana L. against multidrug-resistant Pseudomonas aeruginosa, isolated from clinical samples. Asian Pacific J Trop Biomed. 2012; 2: S800–S807.
-
Iosifidis E, Antachopoulos C, Tsivitanidou M, Katragkou A, Farmaki E, Tsiakou M, Kyriazi T, Sofianou D, Roilides E. Differential correlation between rates of antimicrobial drug consumption and prevalence of antimicrobial resistance in a tertiary care hospital in Greece. Infect Contr Hospit Epidem. 2008;29(7):615-622.
-
Hsu LY, Tan TY, Tam VH, Kwa A, Fisher DA, Koh TH; Network for Antimicrobial Resistance Surveillance (Singapore). Surveillance and correlation of antibiotic prescription and resistance of Gram-negative bacteria in Singaporean hospitals. Antim Agents Chemother. 2010;54(3):1173-8.
-
Jaggi N, Sissodia P, Sharma L. Control of multidrug-resistant bacteria in a tertiary care hospital in India. Antim Resist Infect Contr. 2012;1(1):23.
-
Chopra I, Schofield C, Everett M, O’Neill A, Miller K, Wilcox M, Frere JM, Dawson M, Czaplewski L, Urleb U, Courvalin P. Treatment of health-care-associated infections caused by Gram-negative bacteria: a consensus statement. Lancet Infectious Dis. 2008;8(2):133-139.
-
Sithisarn P, Nantateerapong P, Rojsanga P, Sithisarn P. Screening for Antibacterial and Antioxidant Activities and Phytochemical Analysis of Oroxylum Indicum Fruit Extracts. Molec. 2016 Apr 7;21(4):446.
-
Mat Ali R, Houghton PJ, Raman A, Hoult JR. Antimicrobial and anti-inflammatory activities of extracts and constituents of Oroxylum Indicum (L.) Vent. Phytomed. 1998 Oct;5(5):375-81.
-
Talari S, Sampath A, SujathaK, Nanna RS. Antibacterial activity of stem bark extracts of Oroxylum Indicum and endangered ethnomedicinal forest tree. J Pharm Biol Sci. 2013; 7: 24-28.
-
Singh HB, Prasad P, Rai LK Folk medicinal plants in the Sikkim Himalayas of India, Asian Fol Stud. 2002; 61: 295–310.
-
Dev LR,Ranjeeta P, Anurag M, Rajiv G. Pharmacognostic and phytochemical studies of the bark of Oroxylum Indicum.Pharmacogno J. 2010; 2: 297–303.
-
Samatha T, Srinivas P, Shyamsundarachary R, Rajinikanth M, Swamy NR, Phytochemical analysis of seeds, stem bark, and root of an endangered medicinal forest tree Oroxylum Indicum(L) Kurz.IntJ Pharm Bio Sci. 2012; 3:1063–1075.
-
Balakrishna M, Seetharam P. Qualitative and quantitative evaluation of trace elements in Amaranthaceae family medicinal plant using ICP-MS. Int J Cur Res Rev. 2021; 13 (5); 58-63.
-
Devi SK, Brundha MP, Smiline Girija. Structural Basis of SARS COV 2 3CL PRO Drug And anticovid-19 discovery from medicinal plants - A review. Int J Cur Res Rev. 2020; 12 (21): S101-105.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License