IJCRR - 13(15), August, 2021
Pages: 112-115
Date of Publication: 10-Aug-2021
Print Article
Download XML Download PDF
A Prospective Study on Assessment of Quality of Life of Patients Receiving Sorafenib for Hepatocellular Carcinoma
Author: Chandran Anand, T.V. Syamaprasad, K Pavithran, Vijayan Meenu
Category: Healthcare
Abstract:Introduction: Hepatocellular carcinoma (HCC) is the leading primary liver malignancy and is a major reason of cancer related death worldwide. Sorafenib is considered as the approved drug of choice in advanced HCC, which is an oral multikinase inhibitor with effective anti-proliferative and antiangiogenic effects. Currently, only limited data is available in India regarding the safety and effectiveness of Sorafenib for the management of HCC. Objectives: To evaluate the clinical profile of Sorafenib for treating advanced HCC. Methods: A prospective study of 66 patients who received Sorafenib for advanced HCC was carried out for a duration of 1 year with a mean follow up of 3 months. Effectiveness was analyzed based on Alpha- Fetoprotein (AFP) levels from laboratory findings. The adverse events (AEs) were graded using the Common Terminology Criteria for Adverse Events (CTCAE), version 4.0. In addition, the quality of life (QoL) was analyzed using: EORTC-QLQ-HCC18 (European organization for research and treatment of cancer- Quality of life group questionnaire) and FACT-Hep (Functional assessment of cancer therapy). Result: The patients' median age was 63 years (range 61-70) with the male to female ratio of 16:1. The major adverse effects observed were fatigue (31.81%), hand foot syndrome (24.24%) and diarrhea (24.24%). A statistically significant decline in Alpha Fetoprotein (AFP) level was observed with Sorafenib treatment (p=< 0.001). The QoL was observed to be better and did not worsen during the therapy. Conclusion: Sorafenib is ideal for patients with advanced HCC, irrespective of baseline status with a good clinical profile.
Keywords: Hepatocellular Carcinoma, Sorafenib, Alpha Fetoprotein, Quality of Life, Adverse event, CTCAE
Full Text:
INTRODUCTION
Globally Hepatocellular Carcinoma is a common primary hepatic malignancy with an increased risk of mortality.1HCC is the eleventh and sixth leading cancer worldwide in women and men, respectively. Majority of the patients with HCC has been observed to have chronic liver diseases as a result of infections due to hepatitis virus.2 HCC accounts for about 85% to 90% of all primary hepatic malignancies. Currently, the overall survival (OS) rate for HCC patients is below par, with only 2% of patients with advanced-stage disease and 26% of those with early-stage disease surviving for over five years.3 Surgical resection offers a cure but is not an option in the majority of the patients due to underlying liver disease and the extent of tumour at the time of diagnosis.4 Sorafenib is an oral multi-targeted tyrosine kinase inhibitor with anti-proliferative and antiangiogenic effects for advanced HCC.1 Serum Alpha-Fetoprotein (AFP), neutrophil-lymphocyte ratio (NLR), vascular endothelial growth factor (VEGF), as well as des-c-carboxy prothrombin (DCP), has been investigated as therapeutic biomarkers of Sorafenib. However, AFP is the most commonly used prognostic factor for Sorafenib due to its ease of measurement.5 Based on the promising results seen in clinical trials, Sorafenib has been favoured as first-line therapy in advanced HCC patients. Apart from its effectiveness, the use of Sorafenib, in turn, can cause some adverse drug reactions particularly hand-foot syndrome, fatigue, diarrhoea, weight loss, hypertension, abdominal pain, decreased appetite, fever.6
In India, clinical studies involving evaluation of effectiveness and safety as well as quality of life (QoL) studies of Sorafenib treated patients are less. On these grounds, a study on the clinical profile of Sorafenib is required for designing a treatment plan for the patients.
MATERIALS AND METHODS
A single-centre prospective study was carried out after the approval of the Institutional Review Board. A total of 66 patients who received Sorafenib were selected for a study period of 1 year.
Inclusion and exclusion criteria
The eligibility criteria for the selection of patients were the following: histologically proven HCC, patients who have received Sorafenib during the study period. The exclusion criteria were as follows: patients allergic to Sorafenib, patients with malignancies other than HCC and patients who had a psychiatric illness.
The occurrence of ADR was determined by direct interview with patients/ bystanders and from electronic medical records and their probability was measured using Naranjo ADR Probability Scale. The ADR severity was assessed by using Common Terminology Criteria for Adverse Events (CTCAE) version 4.0. Basal serum AFP and serum AFP at 6-8 weeks were monitored and recorded to assess the effectiveness of treatment. EORTC-QLQ HCC18 and FACT- Hep questionnaires were used to evaluate the Health-Related Quality of Life (HRQoL).
STATISTICAL ANALYSIS
The information recorded on the data collection forms were uploaded in an excel sheet and data was analyzed using IBM SPSS 20.0 (SPSS Inc, Chicago, USA). The results are given in mean ± SD for all the continuous variables, and as a percentage for categorical variables. Paired t-test was done to test for statistical significance of differences in mean values from baseline to follow-up period and the Wilcoxon Signed Rank test was applied for non-parametric data. The treatment duration was determined (in months), along with the onset of any adverse effects.
RESULTS
The study group included 66 patients with a mean age of 63 years. The present study showed a male predominance (96.6%) with a male to female ratio of 16:1. Alcohol consumption (50%) was observed to be the major cause of HCC development, followed by smoking (29.16%), HBV (18.75%), HCV infection (2.08%). Most of the patients had maintained hepatic function with Child-Pugh B (74.2%) and Child-Pugh A (25.8%).
Safety Profile
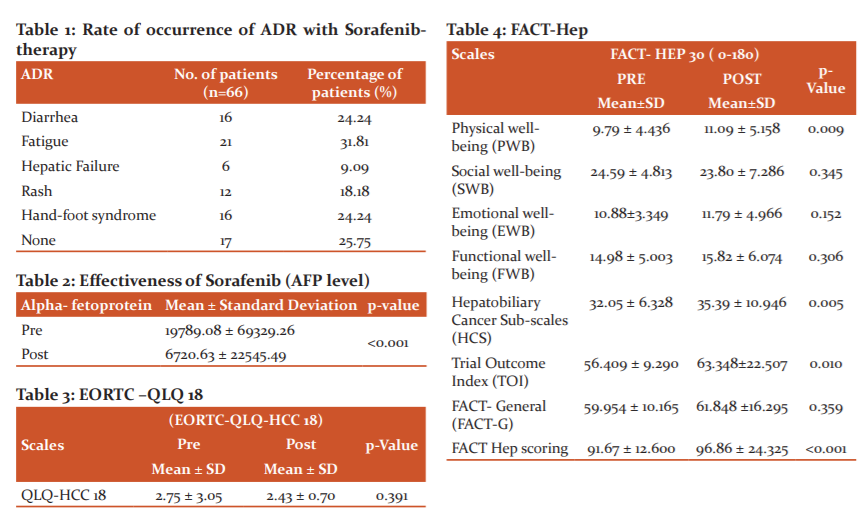
With regards to the safety of treatment, 49 patients experienced several ADR with grades varying from I to IV according to the to CTCAE grading system. The primary adverse reactions were hand-foot syndrome 16 (24.24%), diarrhea 16 (24.24%), fatigue 21 (31.81%), hepatic failure 6 (9.09%) and rash 12 (18.18%) (Table 1).
All of the ADRs were confirmed as Definite (55.1%), Probable (42.85%) and Possible (2%) based on the Naranjo probability scale. According to the CTCAE scale, the incidence of Grade II ADR was 50%, Grade III ADR was 27.02%, Grade I ADR was 21.62% and Grade IV ADR was 1.35%. All these ADRs were managed by dose reduction and drug discontinuation for a short period (4 patients). Dose reduction were done in 13 patients (19.69%) (7 patients from 200mg BD to 200mg OD, 1 patient from 400mg OD to 200mg BD, 1 patient from 200mg TID to 200mg BD, 1 patient from 400mg BD to 400mg OD and 3 patients from 400mg OD to 200mg OD) and the dose escalation were done in 11 patients. Among these 24 patients, 4 subjects ceased and continued the dose for a specific period until the severity of ADRs was reduced.
Effectiveness
Regarding the basal AFP levels, significant differences in survival were observed. Mean AFP was found to be 19789.088 ± 69329.269 at baseline and 6720.637 ± 22545.4962 at final follow-up (<0.001) (Table 2).
Assessment of HRQoL:
HRQoL assessment was done by using EORTC-QLQ HCC18 and FACT- Hep questionnaires.
EORTC-QLQ HCC18: Since the p-value is 0.391 which is greater than 0.005 (Table 3) obtained using Wilcoxon Signed Ranks Test, it is proven that there is no notable difference between Pre and Post EORTC-QLQ HCC 18 scores. There is no significant improvement in QOL of the patients, assessed through EORTC- QLQ HCC 18.
FACT- Hep: We calculated the FACT-Hep scales and analyzed concerning variation in scores. Paired sample t-test was carried out from baseline to 5 months follow-up for all FACT-Hep subscales. A significant increment was found on PWB, HCS, TOI and FACT Hep scoring. The increase in these scores is likely to indicate the effectiveness of the drug. It was also seen that there was a decrement in scores of SWB, EWB, FWB and FACT-G. As the p-value obtained from Wilcoxon Signed Ranks test is <0.001 (Table 4), it is proven that there is a significant difference between Pre and Post FACT - Hep scores, i.e., there is betterment in the quality of life of the patients.
While comparing data of the EORTC- QLQ HCC 18 and FACT- Hep, it was seen that values of the former did not show any difference or improvement in QOL but the latter had improvements in some domains.
DISCUSSION
Despite the latest advancements in the management of advanced HCC, its prognosis continues to be below par. HCC is unaffected by cytotoxic chemotherapy because of the excessive rate of expression of drug-resistant genes. Sorafenib is recommended for unresectable advanced HCC and notably ameliorate progression-free and overall survival. Studies assessing its clinical profile, especially in the Indian population are less.
Most of the patients included in our study were in the age group of 61-70 years with a mean age of 62.48 ± 9.71 years and the median age of 63.5 years. These observations were similar to a study conducted by Abou-Alfa GK et al.7
The number of males was higher (96.6%) than females (3.4%). This could be mainly due to the consumption of alcohol and smoking status in males.
Among our study subjects, the main etiologies were found to be alcohol consumption (48.97%), Smoking (28.57%), HBV (18.36%), HCV (2.04%) and HEV (2.04%). According to the study conducted by (Bai et al., 2013)8, it was seen that Hepatitis B virus constituted an incidence rate of 2.41% (0.05-4.76) in a 95% confidence interval.
In the study, the subject’s mainly dosing regimens used were 200mg OD, 200mg BD, 200mg TID and 400mg OD. They were given to the patients subsequently; 51 (77.27%), 9 (13.63%), 2 (3.03%) and 4 (6.06%) respectively. Among our study population, there was an increase in dose in 11 patients among which 5 patients from 200mg OD to 200mg BD, 1 patient from 200mg BD to 400mg BD, 4 patients from 200mg OD to 400mg OD and 1 patient from 200mg OD to 400mg BD. Owing to the ADRs, reduction of dose was done in 13 patients in which 7 patients from 200mg BD to 200mg OD, 1 patient from 400mg OD to 200mg BD, 1 patient from 200mg TID to 200mg BD, 1 patient from 400mg BD to 400mg OD and 3 patients from 400mg OD to 200mg OD. Among these 24 patients, 4 subjects ceased and continued the dose for a specific period until the severity of ADRs was reduced. Apart from these, 42 patients exhibited good compliance with the therapy.
The adverse reactions observed during Sorafenib therapy were Hand-foot skin reaction 16 (24.24%), Diarrhea 16 (24.24%), Fatigue 21 (31.81%), Hepatic failure 6 (9.09%) and Rash 12 (18.18%). The most frequent ADRs found in our study were Fatigue, Diarrhea and Hand Foot Skin Reaction which were consistent with a study conducted by Llovet et al.9 Grade II drug-related adverse events are reported in our study. This is similar to a study by Giovan et al.10 where Grade I and II Adverse events were commonly reported. Sorafenib related adverse events led to dose reduction or temporary discontinuation. The main cause of reduction in the dosage or temporary discontinuation of the drug is due to diarrhoea and hand-foot skin reaction.
The effectiveness of Sorafenib analyzed using Alpha-fetoprotein, found that there is a significant decrease in AFP levels from baseline (before treatment) to final follow-up i.e., 3 months. A study conducted by Personeni et al.11investigated the prognostic usefulness of a serum AFP response, defined as a > 20% decrease in AFP during 8 weeks of treatment with Sorafenib. Statistical analysis revealed that AFP response is the only prognostic factor of survival.
HRQoL assessment done using EORTC-QLQ HCC18 showed no significant difference between Pre and Post EORTC-QLQ HCC 18 scores (p-value=0.391) indicating no overall improvement in QOL of the patients.
But there was a significant increment in PWB, HCS, TOI and FACT Hep scoring. The increase in the scores is likely to be the effectiveness of the drug. It was also seen that there was a decrement in scores of SWB, EWB, FWB and FACT-G. As the p-value obtained is 0.077 which is > 0.05, it is proven that there is no significant difference between Pre and Post FACT - Hep scores, i.e., there is no improvement in the quality of life of the patients.
Significant relationships were observed between differences in the AFP levels from baseline to 3-month follow-up on the PWB, HepCS and TOI subscales. For all of these subscales, the improvement in AFP was linked with a slight negative change in HRQoL. When AFP was reduced from baseline to 3 months, patients reported a considerable increment in HRQoL. One explanation of this finding could be associated with the fact that those patients who responded to treatment also suffered greater adverse effects, thereby resulting in poorer HRQoL i.e., SWB, EWB, FWB, FACT- G and FACT-Hep.
CONCLUSION
This study emphasizes the superiority of Sorafenib in improving the clinical status and Quality of life in advanced HCC patients. Our study shows that the change in AFP level despite adverse reactions is an important parameter in measuring the effectiveness of Sorafenib. Thus, proving that in addition to its diagnostic and tumour staging function, AFP can also be used as a predictive factor of early response to Sorafenib in HCC patients.
Conflict of interest: The authors have no conflicts of interest, financial or otherwise.
ACKNOWLEDGEMENT
We express our respect and thanks to Amma, the Chancellor of Amrita Vishwa Vidyapeetham; Dr. Sabitha. M, Principal, Amrita School of Pharmacy, Kochi; Dr. Prem Nair, Medical Director, AIMS; Dr. Shantikumar Nair, Dean of Research, AIMS. We extend our sincere thanks to Dr KR Sundaram, HOD, Department of Biostatistics, AIMS for helping us in the statistical works related to the dissertation.
Source of Funding: None.
Authors’ Contribution:
References:
1. Balogh J, Victor D 3rd, Asham EH, Burroughs SG, Boktour M, Saharia A, et al. Hepatocellular carcinoma: a review. J Hepatocell Carcinoma. 2016;3:41–53.
2. Furuse J. Sorafenib for the treatment of unresectable hepatocellular carcinoma. Biolog. 2008;2:779–88.
3. Mittal S, El-Serag HB. Epidemiology of hepatocellular carcinoma: Consider the population. J Clin Gastroent. 2013;47:S2–6.
4. Tinkle CL, Haas-Kogan D. Hepatocellular carcinoma: natural history, current management, and emerging tools. Biologics. 2012;6:207–19.
5. Shiozawa K, Watanabe M, Ikehara T, Shimizu R, Shinohara M, Igarashi Y, et al. Evaluation of sorafenib for advanced hepatocellular carcinoma with low α-fetoprotein in arrival time parametric imaging using contrast-enhanced ultrasonography. J Med Ultrason. 2017;44:101–7.
6. He AR, Goldenberg AS. Treating hepatocellular carcinoma progression following first-line sorafenib: therapeutic options and clinical observations. Therap Adv Gastroent. 2013;6:447–58.
7. Abou-Alfa GK, Schwartz L, Ricci S, Amadori D, Santoro A, Figer A, et al. Phase II study of sorafenib in patients with advanced hepatocellular carcinoma. J Clin Oncol 2006;24:4293– 300.
8. Bai W, Wang YJ, Zhao Y, Qi XS, Yin ZX, He CY, et al. Sorafenib in combination with transarterial chemoembolization improves the survival of patients with unresectable hepatocellular carcinoma: A propensity score matching study: Sorafenib and TACE for HCC. J Dig Dis. 2013;14:181–90.
9. Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc J-F, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378–90.
10. Grignani G, Palmerini E, Ferraresi V, D’Ambrosio L, Bertulli R, Asaftei SD, et al. Sorafenib and everolimus for patients with unresectable high-grade osteosarcoma progressing after standard treatment: a non-randomised phase 2 clinical trial. Lancet Oncol. 2015;16:98–107.
11. Personeni N, Bozzarelli S, Pressiani T, Rimassa L, Tronconi MC, Sclafani F, et al. Usefulness of alpha-fetoprotein response in patients treated with Sorafenib for advanced hepatocellular carcinoma. J Hepatol. 2012;57:101–7.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License