IJCRR - 13(13), July, 2021
Pages: 154-158
Date of Publication: 05-Jul-2021
Print Article
Download XML Download PDF
To Assess Efficacy and Safety of 0.1% Tacrolimus Ophthalmic Ointment in Refractory Cases of VKC
Author: Agarwal P, Tayal SD, Gautum A
Category: Healthcare
Abstract:Introduction: Vernal kerato-conjunctivitis (VKC) is a chronic, bilateral allergic inflammation of the conjunctiva and cornea. For refractory cases, oral or subtarsal corticosteroids are indicated, immunotherapeutic agents like cyclosporine are also used. Recently Tacrolimus is used for the treatment of refractory VKC. It is an immunomodulator with more potency than cyclosporine. Aim & Objective: To assess efficacy and safety of 0.1% tacrolimus ophthalmic ointment in refractory cases of VKC Material and Methods: Present study was a prospective study carried out on refractory cases of vernal kerato-conjunctivitis. Patients were divided into two groups. Group A Patients received Tacrolimus 0.1% ophthalmic ointment twice daily and anti-allergic medications and Group B patients received only allergic medication. Patients were followed up for up to 6 months. At every follow-up, symptoms and signs of both groups were compared. Results: Mean age of the patients in group A was 14.5\?2.4 years and in the group, B was 15.4\?3.1 years. Males predominated females in both groups. Patients with tacrolimus treatment showed a statistically significant decrease in symptom score and sign score over 6 months. (p< 0.05). Conclusion: Tacrolimus ophthalmic ointment 0.1% is well tolerated and effective in improving objective clinical signs and subjective symptoms of severe VKC refractory to topical antiallergic agents.
Keywords: Tacrolimus, Vernal kerato-conjunctivitis, Refractorycases, Steroid, Cyclosporine
Full Text:
Introduction:
Vernal kerato-conjunctivitis (VKC) is a chronic, bilateral allergic inflammation of the conjunctiva and cornea. Prevalence of VKC is more in the 3-25 years age group with peak incidence at 11-13 years.1 Patient commonly present with itching, slight drooping of the upper lid, ropy discharge, photophobia and blurry vision.2Signs observed in VKC are cobblestone papillae on the upper tarsal conjunctiva, discrete or confluent papillary hypertrophy on limbal conjunctiva, white chalky appearing concretions, called Horner-Granta's dots, corneal erosions and shield ulcer.3Histopathologically it is characterized by a dense inflammatory infiltrate consisting of eosinophils, lymphocytes, basophils, dendritic cells and macrophages within microvessel, outside microvessel and in the epithelium. Lymphocytes are aggregated to form follicles. 4
Treatment modalities included measures to control inflammation. Various pharmacological agents used are topical steroids, mast cell stabilizers and topical Non-steroidal anti-inflammatory drugs.5Most of these treatments are ineffective in refractory VKC. Refractory VKC is defined as symptoms and signs that are persistent after conventional treatment. For refractory cases, oral or subtarsal corticosteroids are indicated, immunotherapeutic agents like cyclosporine are also used,6 but chronic use of topical corticosteroids may increase intraocular pressure (IOP) and susceptibility to opportunistic infections.
Novel treatment therapy for severe allergic ocular diseases with potent anti-inflammatory effects as well as sufficient safety is thus needed. Recently Tacrolimus is used for the treatment of refractory VKC. It is an immunomodulator with more potency than cyclosporine. It suppresses T-cell activation, T helper cell-mediated B-cell proliferation, and formation of cytokines, especially interleukin-2.7 In ophthalmology, tacrolimus has mainly been used to suppress immune reactions in corneal and limbal transplantations, uveitis, and allergic eye disease. 8-10
The good safety profile of 0.1% tacrolimus ophthalmic suspension based on the low blood concentration of tacrolimus, coupled with demonstrated better efficacy, make it an important tool for treating severe allergic conjunctivitis. Therefore we chose 0.1% tacrolimus ointment in this study.
Side effects noted in the use of tacrolimus ointment are burning sensation, activation of herpes simplex dendritic keratitis and development of molluscum contagiosum11,12.
Material & methods: Present study was a prospective study carried out to assess the efficacy of 0.1%tacrolimus ophthalmic ointment in refractory cases of VKC at the Department of Ophthalmology at Saraswathi Institute Of Medical Sciences. Patients were enrolled from November 19 to September 2020. The study population was refractory cases of vernal keratoconjunctivitis.
Inclusion criteria: 1. Patients with VKC refractory to conventional treatment for 3 weeks 2. Patients willing to participate in the study and followup
Exclusion criteria:
1. Patients with trachoma 2. Patients with infectious diseases of eye 3. Patients with hypersensitivity to tacrolimus 4. Patients who had less than 6 months follow up 5.Systemic administration of immunosuppressants within 2 weeks before study.6.pregnant or lactating females7.patients with any cardiac, renal or hepatic disease or diabetes.
This study was conducted in compliance with the Declaration of Helsinki. The study was approved by the ethical committee of the institute. A valid written consent was taken from patients after explaining the study to them.
Total of 66 patients was included in the study. Out of 66 patients, 6 patients lost to follow up so data of 60 patients was included in the study. Patients were divided into two groups randomly. Group A patients received Tacrolimus0.1%( Talimus-Ajanta Pharmaceuticals, India ) twice daily and anti-allergic medications (sodium cromoglycate eye drop, ketotifen fumarate eye drop) and Group B patients received only antiallergic medications (sodium cromoglicate eye drop, ketotifen fumarate eye drop). All participants were followed up for 6 months.
At every follow-up, symptoms and signs of vernal keratoconjunctivitis were noted. Five symptoms ( itching, discharge, lacrimation, photophobia, foreign body sensation ) were scored (0 to3) depending on severity. Total score for symptoms(0 to 15)was noted at the initiation of treatment (Baseline), 2wk, at one month,3 month and six months.
Slit-lamp examination findings were used to grade (0=none, 1=mild, 2=moderate and 3=severe) each of the eight clinical signs. Score range (0 to 24). (table 1). For each patient, the eye with a higher total score for clinical findings was selected for efficacy assessment. The reduction in total signs and symptoms scores (from baseline) was used as the determinant of efficacy.


To assess the safety and side effects of the treatment, intraocular pressure, lens opacification, secondary infections, or other possible complications were assessed. Data were entered in an excel sheet and analysed by SPSS version 20.
Results:
In our study, we studied 60 patients. Group A patients received Tacrolimus 0.1%Ophthalmic ointment twice daily and anti-allergic medications(sodium cromoglycate eye drop, ketotifen fumarate eye drop). Group B patients received anti-allergic medications(sodium cromoglycate eye drop, ketotifen fumarate eye drop) only.
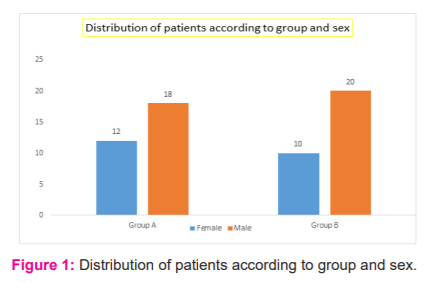
In group A there were 18 males and 12 females and in group B there were 20 males and 10 females. Both the groups were comparable concerning age. The mean age of the patients in group A was 14.5±2.4 years and in the group, B was 15.4±3.1 years. The age of the patient ranged from 1 -27 years.
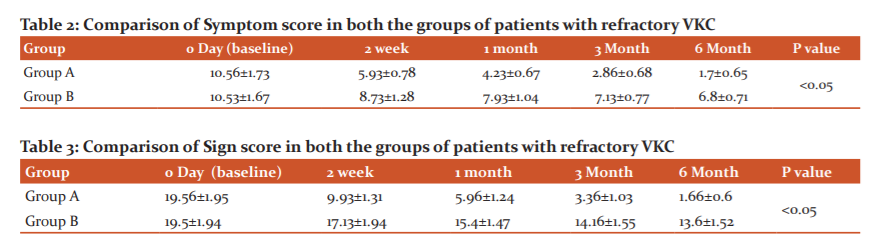
Table 2 shows the comparison score of symptoms in group A and group B. Participants were followed at 2 weeks, 1 month, 3 months and 6 months. Symptom score ranged from 0-15. In the group, A mean score of symptoms at baseline was 10.56±1.73. It decreased over follow up and was found to be 5.93±0.78 (2 weeks), 4.23±0.67 (1 month), 2.86±0.68 (3 months) and 1.7±0.65 (6 months). Thus in the group, A patients showed a significant decrease in symptom score over the follow-up period
In Group B, the mean symptom score at baseline was 10.53±1.67. At 2 weeks it was 8.73±1.28. The score decreased to 7.93±1.04 at 1 month, 7.13±0.77 at 3 months and 6.8±0.71 at 6 months. We observed decreased symptom score in Group B over follow up to 6 months.
In the comparison of both the groups, we found patients in Group A showed a more significant decrease in symptom score as compared to Group B.Resuts were statistically significant (p<0.05)
Table 3 shows comparison score of signs in group A and group B. we assessed the 8 clinical signs with the score ranging from 0-24.
In Group A, the mean sign score at baseline was 19.56±1.95. At 2 weeks it significantly decreased to 9.93±1.31. The score decreased to 5.96±1.24 at 1 month, 3.36±1.03 at 3 months and 1.66±0.6 at 6 months. Group A patients showed a significant decrease in sign score.
In group, B the mean score of symptoms at baseline was 19.5±1.94. It decreased over follow up and was found to be 17.13±1.94 (2 weeks), 15.4±1.47 (1 month), 14.16±1.55 (3 months) and 13.6±1.52 (6 months). Thus in group B patients showed a decrease in symptom score.
In comparison of both the groups, we found patients in Group A showed a statistically significant decrease in sign score as compared to Group B. (p<0.05)
An assessment of visual acuity, IOP, pupil diameter, and clinical findings for the iris, lens, anterior chamber, and fundus, no abnormal change related to testing drugs was observed. In our study, 8 patients from group A developed side effect (7 had transient burning sensation one patient developed burning sensation)which was managed with the addition of lubricating eye drop.
Discussion:
In our study, we studied 60 patients with refractory VKC for testing the efficacy of tacrolimus 0.1% ointment. Previous studies have used different concentrations. A study by Joseph MA12 used 0.02% concentration while a study by Müller GG used 0.03% concentration13. Kheirkhah A used topical 0.005% tacrolimus eye drop for the treatment of VKC14. The good safety profile of 0.1% tacrolimus ophthalmic suspension based on the low blood concentration of tacrolimus, coupled with demonstrated better efficacy, make it an important tool for treating severe allergic conjunctivitis15. Not many studies have been done on Indian eyes to test the efficacy of tacrolimus 0.1% for the treatment of ocular allergic diseases. Therefore we chose 0.1% tacrolimus ointment in this study
Topical tacrolimus has been used for different durations in previous studies. Duration ranged from 1-7 months. In our study, we followed the patients for 6 months.
The mean age of the patients in group A was 14.5±2.4 years and in group B was 15.4±3.1 years. Males predominated females in both the groups. Both the groups were comparable with respect to age and sex.
In group A mean score of symptoms at baseline was 10.56±1.73. It decreased over follow up and found to be 5.93±0.78 (2 weeks), 4.23±0.67 (1 month), 2.86±0.68 (3 months) and 1.7±0.65 (6 months). In Group B, mean symptom score at baseline was 10.53±1.67. At 2 weeks it was 8.73±1.28. Score decreased to 7.93±1.04 at 1 month, 7.13±0.77 at 3 months and 6.8±0.71 at 6 months.
Patients with tacrolimus treatment showed a statistically significant decrease in symptom score(p<.05)
In Group A, the mean sign score at baseline was 19.56±1.95 and it decreased to 1.66±0.6 at 6 months. In group, B the mean score of symptoms at baseline was 19.5±1.94 which decreased to 13.6±1.52 at 6 months. Patients with tacrolimus treatment showed a statistically significant decrease in sign score over 6 months. (p<0.05).
Similar to our study, in the study by Ohashi Yand others, 0.1% twice-daily dose of tacrolimus showed improvement in symptoms of VKC16.
A study by Miyazaki N and others studied effects of 0.02% Tacrolimus ointment for refractory ocular surface inflammatory diseases and found that there was a lower incidence of elevated intraocular pressure and no adverse side effects during 2–26 months of continuous treatment18.
Muller and Jose 13 reported no difference in the improvement of symptoms or signs in 2 groups of patients with severe VKC treated with tacrolimus and olopatadine or tacrolimus alone.
In contrast to a study by Muller GG,13 Tam PMK 19 reported the additional use of mast cell stabilizers in 2 of 5 patients. A study by Fukushima 20 reported the continued requirement of steroids in nearly half of their patients.
In our study tacrolimus ophthalmic ointment 0.1% was well-tolerated, 8 patients from group A developed side effect(7 had transient burning sensation one patient developed burning sensation) which was managed with the addition of lubricating eye drop. Previous studies have also reported burning sensation upon application of topical tacrolimus.21This study findings suggest the usefulness of topical tacrolimus 0.1% as a steroid-sparing substitute for treating severe allergic conjunctivitis due to its potent immunosuppressive effect,
Conclusion: In this study, we conclude that Tacrolimus ophthalmic ointment 0.1% is well tolerated and effective in improving objective signs and subjective symptoms in cases of severe VKC who are refractory to other topical antiallergic agents.
Conflict of interest-NIL
The Source of the funding-The study was carried out at Saraswathi Institute Of Medical Sciences, Hapur and no extra financial support was required
Ethical Clearance-Ethics committee approval was obtained before the study.IECSIMS/FMT/ETHI/22/19
Acknowledgement-Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of manuscript.
References:
-
Kumar S. Vernal keratoconjunctivitis: a major review. Acta Ophthalmol. 2009;87:133–147.
-
Abu El-Asrar AM, Tabbara KF, Geboes K, Missotten L, Desmet V. An immunohistochemical study of topical cyclosporine in vernal keratoconjunctivitis. Am J Ophthalmol. 1996;121:156–161.
-
Bielory L, Frohman LP. Allergic and immunologic disorders of the eye. J All Clin Immunol. 1992;89:1–15.
-
Buckley DC, Coldwell DR & Reaves TA (1993): Treatment of vernal keratoconjunctivitis with superfan, a topical non-steroidal anti-inflammatory agent. Invest OphthalmolVis Sci 38: 133–140.
-
Sechhi AG, Tognon MS & Leonard A (1990): Topical use of cyclosporine in the treatment of vernal keratoconjunctivitis. Am J Ophthalmol 110: 641–64.
-
Sawada S, Suzuki G, Kawase Y, Takaku F. Novel immunosuppressive agent, FK506. In vitro effects on the cloned T cell activation. J Immunol 1987; 139(6): 1797–1803.
-
Kobayashi C, Kanai A, Nakajima A, Okumura K. Suppression of corneal graft rejection in rabbits by a new immunosuppressive agent, FK-506. Transplant Proc. 1989; 21(1 Part 3): 3156–3158.
-
Kawashima H, Fujino Y, Mochizuki M. Effects of a new immunosuppressive agent, FK506, on experimental autoimmune uveoretinitis in rats. Invest Ophthalmol Vis Sci. 1988; 29(8): 1265–1271.
-
Iwamoto H, Yoshida H, Yoshida O, Fukushima A, Ueno H. Inhibitory effects of FK506 on the development of experimental allergic/immune-mediated blepharoconjunctivitis in Lewis rats by systemic but not by topical administration. Graefes Arch Clin Exp Ophth. 1999; 237(5):407–414.
-
ZribiH, Descamps V, Hoang-Xuan T, Crickx B, Doan S. Dramatic improvement of atopic keratoconjunctivitis after topical treatment with tacrolimus ointment restricted to the eyelids. J Eur Acad Dermatol Venereol. 2009; 23(4): 489–490.
-
Joseph MA, Kaufman HE, Insler M. Topical tacrolimus ointment for the treatment of refractory anterior segment inflammatory disorders. Cornea. 2005; 24(4): 417–420.
-
Müller GG, José NK, Castro RS. Topical Tacrolimus 0.03% as Sole Therapy in Vernal Keratoconjunctivitis. Eye Contact Lens. 2014;40:79-83.
-
Kheirkhah A, Zavareh MK, Farzbod F, Fukushima A, Kumagai N, Nakagawa Y et al. Topical 0.005% tacrolimus eye drop for refractory vernal keratoconjunctivitis. Eye. 2011;25:872–880.
-
Ebihara N. Blood level of tacrolimus in patients with severe allergic conjunctivitis treated by 0.1% tacrolimus ophthalmic suspension doi: 10.2332/allergolint.11-OA-0349. Epub 2012 Feb 25.
-
Ohashi Y, Ebihara N, Fujishima H, Fukushima A, Kumagai N, Nakagawa Y et al. A randomized, placebo-controlled clinical trial of tacrolimus ophthalmic suspension 0.1% in severe allergic conjunctivitis. J Ocul Pharmacol Ther. 2010;26:165–174.
-
Al-Amri AM. Long-term follow-up of tacrolimus ointment for the treatment of atopic keratoconjunctivitis. Am J Ophthalmol. 2014;157:280–286.
-
Miyazaki D, Tominaga T, Kakimaru- Hasegawa A, Nagata Y. Therapeutic effects of tacrolimus ointment for refractory ocular surface inflammatory diseases. Ophthalmology 2008;115:988–92.
-
Tam PMK, Young AL, Chen LL. Topical tacrolimus 0.03% monotherapy for vernal keratoconjunctivitis—case series. Br J Ophthalmol. 2010;94:1405–1406.
-
Fukushima A, Ohashi Y, Ebihara N. Therapeutic effects of 0.1% tacrolimus eye drops for refractory allergic ocular diseases with a proliferative lesion or corneal involvement. Br J Ophthalmol. 2014;98:1023–1027
-
Kymionis GD, Goldman D, Ide T, Yoo SH. Tacrolimus ointment 0.03% in the eye for treatment of giant papillary conjunctivitis. Cornea. 2008;27:228–229.
-
Rikkers SM, Holland GN, Drayton GE, Michel FK, Torres MF, Takahashi S. Topical tacrolimus treatment of atopic eyelid disease. Am J Ophthalmol. 2003;135:297–302.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License