IJCRR - 13(13), July, 2021
Pages: 101-107
Date of Publication: 05-Jul-2021
Print Article
Download XML Download PDF
Phytochemical Screening and Evaluation of Cytotoxicity and Acute Toxicity of Ethanolic Leaf Extract of Cayratiaauriculata
Author: Lalitha Surulichamy, Anusha Dakshinamoorthi, Yogesh Kumar Murkunde, Viji Devaanand, K. Maheshkumar
Category: Healthcare
Abstract:Introduction: The Cayratiaauriculata has been used as a folk medicine to treat various pathophysiological conditions. Aim: In the present study, we evaluated the presence of major phytochemicals, cytotoxicity and acute toxicity effect of ethanolic extract of Cayratiaauriculata leaves. Methodology: The phytochemical screening was carried out using chemical methods; gas chromatography-mass spectrometry (GC-MS) analysis was performed to identify the individual phytoconstituents present in it; cytotoxicity assay was performed in A549 cells and acute toxicity test was performed in the zebrafish model. Results: The results of the qualitative analysis revealed the presence of flavonoids, phenols, coumarin, saponins, tannins, terpenoids, steroids and glycosides. C. auriculatawas found to contain a significant amount of total flavonoid and phenol contents in quantitative analysis. Fifteen different phytoconstituents were expressed in GCMS analysis. In the acute toxicity test, the methanolic extract of C.auriculatadid does not cause mortality or any clinical signs of toxicity in zebrafish to the maximum concentration of 100 mg/L.Therefore the LC50 value of extract was found to be ?100 mg/L. Hence it can be considered safe. Conclusion: This paper will help in considering C. auriculate or further pharmacological studies in future.
Keywords: Herbal extract, Cayratia, phytochemicals, Cytotoxicity, Acute toxicity, Zebrafish
Full Text:
INTRODUCTION
Medicinal plants and their derivatives have a long history of treating human diseases. Day by day, these medicinal plants draw the attention of worldwide researchers because of their lesser side effects and good compatibility with the human body .1The active compounds present in plants containing medicinal properties are considered herbal drugs. These active compounds are phytochemicals and also called secondary metabolites. Some of the common secondary metabolites found in plants include alkaloids, flavonoids, terpenoids, glycosides and phenolics.2 Isolated bioactive molecules from plant serve as starting materials for drug development. 3 However, these secondary metabolites are found to be in meagre quantity in plant material. Due to this constraint, the extraction, purification and characterization of secondary metabolites become very crucial in the process of the plant-based drug discovery process. 4 Extraction is a preliminary and critical step in the process of discovery and isolation of bioactive material. Phytochemical analysis of raw plant materials is very significant to detect and quantify the phytoconstituents present in it. 5
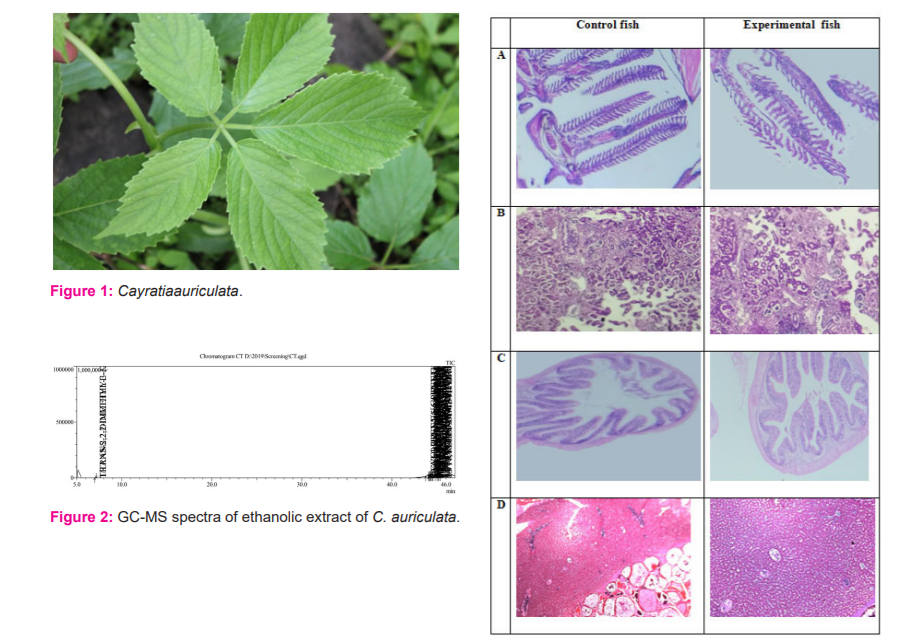
Cayratiaauriculata(C. auriculata) belongs to the Vitaceae family, class Magnoliopsida and phylum Tracheophyta. It is commonly called Jangliangoor and Amarchotioo. 6 Synonyms ofCayratiaauriculataare Cyphostemmaauriculatum (Roxb.), CissusauriculataRoxb., Vitisauriculata (Rob.), and Cayratiaauriculata (Roxb.) Gamble. It has been reported to be distributed in Bangladesh, Bhutan, India, Myanmar, Thailand and Sri Lanka. C. auriculata is a climber with spongy stems, 5-foliate leaves, tetramerous flower and cherry-sized red fruits (Figure 1). Cayratiaauriculata has 2n= 24 chromosomes. Cayratia species has a significant role in the preparations of Ayurvedic medicines, homemade remedies, and natural pesticides as it has a good source of Phytochemicals. 6,7C. auriculatahas following significant medical application. It has been used to treat ulcers, cough, cold, intestinal worm, rheumatism, hydrocele, ulcer, diarrhoea and abscess. Its leaf decoction was used as a remedy for uterine disorder and fever. 7 The bark of C. auriculata is used to treat burns, boils, wounds and snakebite. 9-12 Apart from this, the shoot and leaves of C. auriculata were also used as vegetables. 7Despite the widely reported therapeutic applications of C.auriculata, there is no research finding reporting its toxicity profile. Moreover, there was no literature available on the phytochemical processing of this plant species. To address these lacunae, in the present study ethanolic extract of C.auriculataleaveswas examined for the qualitative and quantitative phytochemical profile, identification of phytoconstituents through GCMS, cytotoxicity effect and acute toxicity effect in the zebrafish model.
MATERIALS AND METHODS:
Collection and Extraction of Plant Material:
The C. auriculata plant was collected in forest areas of Visakhapatnam district, Andhra Pradesh. The plant was authenticated by DrPadal, Associate Professor, Department of Botany, Andhra University, and Visakhapatnam- 530003. The leaves were washed thrice thoroughly with distilled water to remove the dirt and debris and then dried under shadow till it gets completely dried. The dried leaves were coarsely ground powdered and extracted using a soxhlet apparatus as follows. Briefly, about 20 g of dry leaf powder of C.auriculatawas extracted with 500 ml of ethanol (Finar Ltd.) solvent. The filtered crude plant extract was concentrated using a rotary evaporator (Buchi, Switzerland). The thick extract was obtained and stored under -20°C for further analysis.
Qualitative preliminary phytochemical analysis
The preliminary phytochemical qualitative screening was carried out using an ethanolic extract of C.auriculatasuch as follows
About 5 ml of extract was shaken vigorously with 5 ml of warm distilled water in a test tube. The formation of stable foam was taken as an indication of the presence of saponins.13
About 2 ml of extract was added to 2 ml of acetic and then cooled well in ice. Then Con. H2SO4 was added carefully. A colour change from violet to blue to green indicates the presence of a steroidal nucleus (which is the aglycone portion of glycoside.13
To 2 ml of extract 2 ml of 10% sodium hydroxide was added. The appearance of yellow colour indicates the presence of coumarin. 15
To 2 ml of extract, 2 ml of the con. HCL was added. Then few drops of Mayer’s reagent were added. The presence of green colour or white precipitate indicates the presence of alkaloids. 13
To 5 ml of extract, 3 ml of lead ethanoate solution was added. The formation of buff-coloured precipitate was taken as an indication of the presence of flavonoids. 14
To 1 ml of extract, 2 ml of 5% ferric chloride was added. The formation of green, blue-black, or blue-green indicates the presence of tannins. 15
To 1 ml of extract,2 ml of distilled water followed by a few drops of 10% ferric chloride was added. The formation of blue or green colour indicates the presence of phenols. 15
About 2 ml of extract was dissolved in 2ml of chloroform and evaporated to dryness. 2ml of concentrated sulphuric acid was then added and heated for about 2min. Development of a greyish colour indicates the presence of terpenoids.16
About 2 ml of extract was dissolved in 2ml of chloroform and 2ml concentrated sulphuric acid. A red colour produced in the lower chloroform layer indicates the presence of steroids. 16
About 2 ml of extract was shaken with 10 ml of benzene and then filtered. And 5 ml of the 10% ammonia solution was then added to the filtrate and thereafter shaken vigorously. The appearance of a pink, red or violet colour in the lower ammonia layer was taken as the presence of free anthraquinones.13
Phytochemical Quantitative Analysis:
The phytochemical quantitative screening was carried out using ethanolic extract of C.auriculatato to estimate the number of total phenolics and flavonoids by the method of. 17
2.3.1.Totalphenolics content
The total phenolics content of ethanolic extract of C.auriculatawas estimated using Folin-Ciocalteau reagent. About 20 µg of the extract was taken and made up to 1 mL with distilled water. Then 500 µL of diluted Folin’s reagent and 2.5 mL of 20% sodium carbonate solution were added. The mixture was shaken well and incubated in dark for 40 min and read spectrophotometrically at 725 nm. A calibration curve of gallic acid was constructed. The results were compared with the gallic acid calibration curve and the total phenolic content of the sample was expressed as mg of gallic acid equivalent (mg GAE/g extract) by using the standard curve.
Total flavonoids content
About 1 mL of extract was diluted with 200 µL of distilled water followed by the addition of 150 µL of 5 % sodium nitrite solution. This mixture was incubated for 5 min and then 150 µL of 10% aluminium chloride solution was added and allowed to stand for 6 min. Then 2 mL of 4% sodium hydroxide solution was added and made up to 5 mL with distilled water. The mixture was shaken well and left it for 15 min at room temperature. The absorbance was read at 510 nm. The total flavonoids content was expressed as rutin equivalent mg RE/g extract using the standard curve.
GCMS Analysis:
Gas chromatography-Mass Spectroscopy analysis was performed to identify the phytoconstituents present ethanolic extract of C.auriculataleaves. It is an effective chemical analysis and also a common confirmation test.18 A Shimadzu GC-2010 Plus gas chromatograph was used for analysis. The sample was introduced by split injection of ratio 10:1. The oven temperature was programmed to increase as follow, 35°C for 2 minutes, then rise by 20°C per minute to reach 450°C and remain at 450°Cfor 5 minutes. The helium is used as carrier gas at a flow rate of 2 ml/minute. The software GCMS solution ver. 2.6 was used for analyses. Identification of the components present in the extract was determined by comparing the name, molecular weight, and structure of the spectrum of known components stored in the library of National Institute Standard and Technology (NIST) library V which was provided by the instrument software.
Acute toxicity study in zebrafish:
60 adult zebrafish of both male & female with a mean body length and weight of 3±0.5 cm and 0.334±0.05 g respectively were procured from Whizbang Bioresearch, Chennai. The acute toxicity study was performed as per OECD 203. After acclimatization, the fish were randomly divided into six groups of 10 fishes each. The test solution was prepared by dissolving the required quantity of the test item in the aquarium habitat water of known quantity. The fishes in Group I, II, III, IV and V was treated with extract at different concentrations during the test period of 96 hours, that is, 100, 50, 25, 12.5, and 6.25 mg/L respectively. The fishes in Group VI were normal control. The exposure solutions were maintained at optimum pH, temperature and dissolved oxygen concentration throughout the study as same as in the acclimation procedure. The test fishes were observed and recorded at 24, 48, 72 and 96 hours for mortality and morbidity. Observations were done at 0, 3, 6, 24, 48, 72 and 96 hours for clinical signs of toxicity. At the end of the test period, fishes were euthanizedinTricaine (MS-222) and subjected to necropsy.
Histology of zebrafish:
For the histopathology analysis, the fish was fixed in 10% neutral buffered for 48 h. Then the fish were processed in graded concentrations of alcohol, xylene and impregnate in paraffin. Processed tissues were embedded in paraffin block and whole body sagittal sections were prepared at 5- micron thickness mounted on slides and stained with Haematoxylin and Eosin stain. The analysis of slides was performed under a light microscope [Optoscope] and photographed with the camera (digital). Slides were scored as per the method described based on the severity of histological changes 19
RESULTS & DISCUSSION:
In the present study, we established the phytochemical and toxicity profile of the ethanolic extract of C. auriculata leaves for the first time. No studies were carried out in this species before. Therefore, the further comparison was made on the same plant genus. As C. auriculata has been used as a crude extract in folk medicine, we have used the crude ethanolic extract of C. auriculate or all our experiments. Moreover, the crude extract will contain a mixture of bioactive compounds. Though the plant-based medications are often considered to be safe and have no side effects, 20 it is essential to derive the safety profile of the particular plant extract to determine the dose level for the examination of the therapeutic index of drugs through subsequent pharmacological studies.Hence we scrutinized the cytotoxic potential and acute toxic potential of C. auriculata in a zebrafish model.
Phytochemical preliminary Qualitative Analysis:
The preliminary phytochemical screening using chemical methodsconducted on the ethanolic extract revealed the presence of various phytochemicals like flavonoids, phenols, coumarin, saponins, tannins, terpenoids, steroids and glycosides. The qualitative analysis for the C.auriculata extract is shown in Table 1. In another study, phytochemical screening of Cayratiapedata(Lam.) Gagnep. var. glabraGamble has been reported to contain carbohydrates, proteins, amino acids, alkaloids, anthraquinones, flavonoids, glycosides, phenols and tannins, steroids and sterols, triterpenoids and volatile oil. 21 Cayratiatrifoliawas found to contain kaempferol, myricetin, quercetin, triterpenes and epifriedelanol, steroids, terpenoids, flavonoids, tannins, hydrocyanic acid and delphinidin. 22 The leaf and stem of Cayratiagracilisshowed the presence of carbohydrates, tannins, saponins, flavonoids, balsams, resins, terpenes, alkaloids and sterols. 23Ethanol extract of galing stem (C. trifoliaDomin.) shows the presence of alkaloids, saponins, terpenoids, tannins, and flavonoids. 24 These results show that all Cayratia plants consist of the following common phytochemicals, flavonoids, glycosides, phenols, tannins, steroids, terpenoids, saponins, and alkaloids. These phytochemicals have certain pharmacological properties. For example, phenolics compound act as a reducing agent, hydrogen donor, metal chelator25 and has anticancer and cardioprotective activity . 21 Flavonoids act as an antioxidant .23, 24, 25 Tannins have astringent and anti-diarrhoea activity. Saponins are known to have activity against gastro-intestinal infections and cardiovascular diseases . 23
3.2. Phytochemical Quantitative Analysis:
The phytochemicals present in the extracts was quantitatively determined by standard procedure. The total phenolic and flavonoid content in the ethanolic extract of C. auriculata was estimated to be 111.36 mg GAE/g and 26.32 mg RE/g extract. In another study, the stem ethanolic extract of C.trifoliawas estimated to contain total phenol, tannin, alkaloid, flavonoid and saponin contents as 34.97 ± 0.4, 54.52 ± 0.3, 33.74 ± 0.68, 26.07 ± 0.40, and 39.52 ± 0.50 mg/g respectively.25 The ethanol extract of C. pedatavar. glabrawas found to contain 131.7 ± 3.6 and 52.8 ± 12.9 mg TAE/g extract of Total phenolics and Tannin respectively.21 C. pedateandC.auriculatacontain a large amount of phenolic content.
GCMS Analysis:
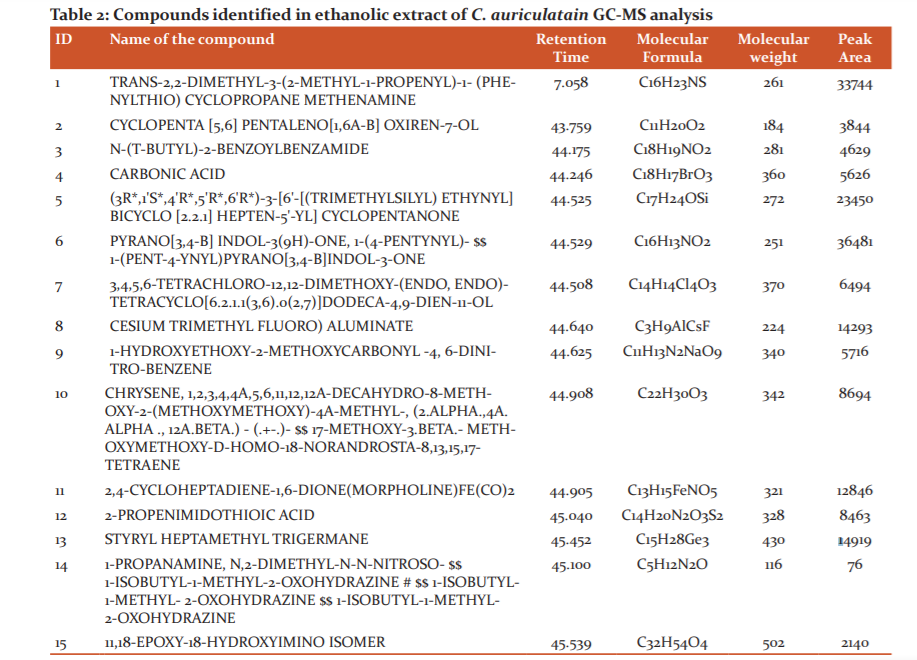
GC-MS chromatogram analysis of the ethanolic extract of C. auriculata indicating the presence of fifteen different phytochemical constituents by comparing their retention times, molecular formula and molecular weight (MW) and mass spectra [Figure 2]. GC-MS analysis for biomolecules in plant extract provides deep insight into the medicinal properties of the plant. 18 The various compounds detected by GC-MS analysis in C. auriculatais shown in (Table 2). In which, the carbonic acid was identified to have an essential role in nitrogen base protonation in blood serum. 26 Further study on predicted biomolecules will help identify the pharmacological activity of each compound.
GC-MS analysis on ethanolic extract of C. trifoliastem exhibited 20phytoconstituents. In which the following compounds are found to present in higher concentration, hexadecanoic acid, ethyl ester, phytol, tetratriacontane, stigmasterol, nonacosane and octadecane.27
Acute toxicity study of the extract in zebrafish:
Zebrafish has around 70% of homologous genes to that of humans. It has become an efficient model vertebrate in toxicity and pharmacology studies .32-34 Hence, we presumed to evaluate the acute toxic potential of C. auriculatain in the zebrafish model. The test conducted to determine the LC50 value of extract in 96 hrs. of exposure. The results show that there were no morbidity, mortality or clinical signs of toxicity were observed in all the experimental groups throughout the study. 28-31All the test fish were found to be normal when compared to the control. There were no treatment-related gross pathological changes were visualized across different test groups in comparison with the control group. These data show that the LC50 of ethanolic extract of C. auriculatawas found to be greater than 100 mg/L under the tested experimental conditions in the present study. As per Organization for Economic Co-operation and Development (OECD) and European Chemicals Bureau (ECB), the pollutants are categorized as harmful to zebrafish if., 10 mg / L < LC 50 < 100 mg / L. 35
Based on this categorization, ethanolic extract of C. auriculatawas considered to be safe. An acute toxicity study of ethyl acetate extract of C. trifolia was performed as per OECD guideline No. 420 in female Wistar rats. The result reveals that C. trifoliawas found to be safe up to the dose of 2000 mg/kg .36This result is in line with our study that Cayratiaspecies shows no toxicity.
Histopathology of zebrafish:
Histopathological investigations were carried out to find out any changes in the cellular morphology and architecture in test fish when compared with control fish. Acute toxicity studies on different doses of C. auriculata leaf extracts showed no discrete histopathological changes in the gills, kidney, liver, and intestine, heart, and muscle tissue of the test fishes in comparison with control group fishes (Figure 4). There was normal cellular architecture observed in all the experimental groups.
CONCLUSION:
To conclude based on the above results, it was found that the ethanolic extract of C. auriculata holds more phytochemicals and contains various phytoconstituents which was detected through GCMS. Future studies on these phytoconstituents may be useful in identifying their pharmacological efficacy. The extract shows a cytotoxic effect against A549 cells in comparison with control. Acute toxicity test results show that the extract is safe in testing with zebrafish. Thus C.auriculatacanbe further studied for its pharmacological activity in future.
Acknowledgement:
The authors would like to acknowledge the unstinted support by the management of Sri Ramachandra Medical College and Research Institute, SRIHER (DU), Chennai.
Conflict of interest:
Nil
Source of Funding
Nil



References:
1. Oladeji O. The Characteristics and Roles of Medicinal Plants: Some Important Medicinal Plants in Nigeria. Nat Prod Ind J. 2016;12(3):102;
2. Egamberdieva D, Mamedov N, Ovidi E, Tiezzi A, Craker L. Phytochemical and Pharmacological Properties of Medicinal Plants from Uzbekistan: A Review. J Med Act Plants. 2017;5(2):59–75.
3. Dhanani T, Shah S, Gajbhiye NA, Kumar S. Effect of extraction methods on yield, phytochemical constituents and antioxidant activity of Withaniasomnifera. Arab J Chem. 2017; 10:S1193–9.
4. Brusotti G, Cesari I, Dentamaro A, Caccialanza G, Massolini G. Isolation and characterization of bioactive compounds from plant resources: The role of analysis in the ethnopharmacological approach. J Pharm Biomed Anal. 2014;87:218–28.
5. Quamar MF, Bera SK. Ethno-medico-botanical studies of plant resources of Hoshangabad district, Madhya Pradesh, India: retrospect and prospects. J Plant Sci. Res. 2014;1(1):101-5.
6. Trias-Blasi A, Chayamarit K, Parnell JAN. Cyphostemmaauriculatum (Roxb.) P. Singh & B. V. Shetty (Vitaceae): typification and a new generic record confirmed for Thailand. Kew Bull.2015;70(2):20.
7. Tetali S, Karkamkar SP. Cayratiasps. of family Vitaceae for utilization in the grape improvement programme. J Plant Sci. Res. 2016;(2):287-290.
8. Sujata T, Karkamkar SP. Cayratiasps. of family Vitaceae for utilization in the grape improvement programme. Int J Minor Fruits, Med Arom Plants. 2016;2(2):14-21.
9. Kamble SY, Patil SR, Sawant PS, Sawant S, Pawar SG. Studies on plants used in traditional medicine by Bhilla tribe of Maharashtra. 2010;9(3):8.
10. Upasani MS, Upasani SV, Beldar VG, Beldar CG, Gujarathi PP. Infrequent use of medicinal plants from India in snakebite treatment. Integr Med Res. 2018;7(1):9–26.
11. Oguegbulu EN, Nwadibia H. Comparative Investigation of Physico-Chemical Properties of Two Varieties of Palm Kernel Oil in Use in Ethno Medicine. Int J Cur Res Rev. 2020;12(12):11.
12. Jain A, Katewa SS, Galav P, Nag A. Some therapeutic uses of biodiversity among the tribals of Rajasthan.Indian J TraditKnowl. 2008;7(2): 256-62
13. Sofowora A. Screening plants for bioactive agents in medicinal plants and traditional medicine in Africa. 2nd Ed. House S, editor. Spectrum books Ltd. 1993. 134–156 p.
14. Evans WC. Trease and Evans Pharmacognosy.15th edition. Edinburgh, Saunders. 2002.
15. Roghini R, Vijayalakshmi K. Phytochemical screening, quantitative analysis of flavonoids and minerals in ethanolic extract of Citrus paradisi. Int J Pharm Sci Res. 2018;9(11):4859-64.
16. Gul R, Jan SU, Faridullah S, Sherani S, Jahan N. Preliminary Phytochemical Screening, Quantitative Analysis of Alkaloids, and Antioxidant Activity of Crude Plant Extracts from Ephedra intermedia Indigenous to Balochistan. Sci World J. 2017;2017:1–7.
17. Senguttuvan J, Paulsamy S, Karthika K. Phytochemical analysis and evaluation of leaf and root parts of the medicinal herb, Hypochaerisradicata L. for in vitro antioxidant activities. Asian Pac J Trop Biomed. 2014;4:S359–67.
18. Rukshana. Phytochemical Screening and GC-MS Analysis of Leaf Extract of Pergulariadaemia (Forssk) Chiov. 2017;7.
19. Zodrow JM, Stegeman JJ, Tanguay RL. Histological analysis of acute toxicity of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) in zebrafish. AquatToxicol. 2004;66(1):25–38.
20. Kasthuri O R, Ramesh B. Toxicity Studies on Leaf Extracts of Alternantherabrasiliana (L.) Kuntze and Alternantherabettzickiana (Regel) Voss. J Appl Pharm Sci. 2018;8(10):82–9.
21. Sharmila S, Kalaichelvi K, Dhivya SM. Pharmacognostical and phytochemical analysis of Cayratiapedata var. glabra—A vitaceae member. Int J Pharm Sci Res. 2018;9:218-26.
22. Kumar D, Gupta J, Gupta A, Kumar S, Arya R. A review on chemical and biological properties of Cayratiatrifolia Linn. (Vitaceae).Pharmacogn Rev. 2011;5(10):184.
23. Henry E, Jemilat I, Kudirat M, Uche E, Samuel O. Phytochemical, Pharmacognostic and Elemental Analysis of Cayratiagracilis (Guill. and Perr.) Suesseng. J Appl Pharm Sci. 2015;048–52.
24. Yusuf MI, Wahyuni W W, Sri Susanty S, Ruslan R R, Fawwaz M. Antioxidant and Antidiabetic Potential of Galing Stem Extract (CayratiatrifoliaDomin). Pharmacogn J. 2018 ;10(4):686–90.
25. Sowmya S, Perumal Pc, Anusooriya P, Vidya B, Pratibha P, GopalakrishnanVk. Quantitative Analysis And In Vitro Free Radical Scavenging Activity Of CayratiaTrifolia. World J Pharm Res.2019; 3(6):16.
26. https://en.wikipedia.org/wiki/Carbonic_acid#cite_note-8.
27. Sowmya S, Perumal PC, Gopalakrishnan VK. Chromatographic And Spectrophotometric Analysis Of Bioactive Compounds From CayratiaTrifolia (L.) STEM. World J Pharm Res.2018; 8(6):110.
28. He Y, Zhu Q, Chen M, Huang Q, Wang W, Li Q, Huang Y, Di W. The changing 50% inhibitory concentration (IC50) of cisplatin: A pilot study on the artefacts of the MTT assay and the precise measurement of density-dependent chemoresistance in ovarian cancer. Oncotarget. 2016;7(43):70803.
29. J. AnbuJebaSunilson, G. Rejitha, K. Anandarajagopal, Amitava Das MM and PP. Cytotoxic Effect of CayratiacarnosaLeaves on Human Breast Cancer Cell Lines. Int J Cancer Res. 2009;5(3):115–22.
30. DS, Keo S, Cheng SK, Oh H, Kim YC. Protective effects of Cambodian medicinal plants on tert?butyl hydroperoxide?induced hepatotoxicity via Nrf2?mediated heme oxygenase. Mol Med Rep. 2017;15(1):451-9.
31. Perumal PC, Sowmya S, Velmurugan D, Sivaraman T, Gopalakrishnan VK. Assessment of dual inhibitory activity of epifriedelanol isolated from Cayratiatrifolia against ovarian cancer. Bangladesh J Pharm. 2016;11(2):545.
32. Zakaria F, Ibrahim WN, Ismail IS, Ahmad H, Manshoor N, Ismail N, Zainal Z, Shaari K. LCMS/MS Metabolite Profiling and Analysis of Acute Toxicity Effect of the Ethanolic Extract of Centellaasiatica on Zebrafish Model. Pert J Sci Techn. 2019;27(2): 971 - 989.
33. Khudoley V V. Use of aquarium fish, Daniorerio and Poeciliareticulata, as test species for evaluation of nitrosamine carcinogenicity. Natl Cancer InstMonogr. 1984;
34. Wibowo I, Permadi K, Hartati R, Damayanti S. Ethanolic extract of pomegranate (Punicagranatum L) peel: acute toxicity tests on zebrafish (Daniorerio) embryos and its toxicity prediction by in silico. J Appl Pharm Sci. 2018; 8(06):082-6.
35. Batra S, Batra N, Nagori BP. Preliminary phytochemical studies and evaluation of the antidiabetic activity of roots of Cayratiatrifolia (L.) Domin in alloxan-induced diabetic albino rats. J Applied Pharm Sci. 2013;3(3):97.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License