IJCRR - 13(12), June, 2021
Pages: 65-73
Date of Publication: 22-Jun-2021
Print Article
Download XML Download PDF
Lipopolysaccharide-Induced Double Hit on Neurobehaviour and Neurochemistry in the Wistar Kyoto Rat, A Model with Endogenous Depressive-Like Profile
Author: Reshma A. Shetty, Monika Sadananda
Category: Healthcare
Abstract:Introduction: The progenitor Wistar Kyoto (WKY) model, outbred from Wistar rats, demonstrates neurochemical and behav�ioural profiles similar to symptom-presenting depressive patients and may be particularly suitable for teasing out interconnecting phenomena underlying depression and inflammation. Aims and Objectives: To investigate links between brain function and peripheral infection which has gained currency during the current pandemic, there is a need to find suitable models. Methods: Lipopolysaccharide (LPS) was administered to assay neurobehaviours, brain and liver enzyme activity. First, baseline indices of anxiety (elevated plus maze-EPM) and learned helplessness (forced swim test-FST) concerning Wistars was obtained. Results: WKYs demonstrated increased anxiety and inhibitory behaviours in the EPM while risk-taking was reduced. On habitu�ation to the FST, WKY demonstrated reduced immobility, increased swimming and climbing behaviours. In the test, increased adaptive responses were observed. LPS induced a further increase in anxiety levels (EPM) with a concomitant decrease in exploratory behaviour in the novel activity box. Centrally, LPS reduced monoamine oxidase activity in the frontal cortex and hypothalamus while hypothalamic acetylcholinesterase activity was increased. Peripherally, LPS increased liver glutathione activity, with no effect on malondialdehyde at the dose tested. Conclusion: The WKY model could therefore prove a valuable model to test the 'double hit' hypothesis in understanding the neuro-immune link in depression.
Keywords: Wistar Kyoto rat, Lipopolysaccharide, Depression, Behaviour, Neurochemistry
Full Text:
Introduction
Investigating links that exist between peripheral infection and inflammation and brain function and dysfunction has become essential because of the currently ongoing pandemic. However, to tease out this circuitry, there is a need to evolve suitable models. The Wistar-Kyoto (WKY) rat, which is an inbred strain of the progenitor Wistar rat arose as a hypotensive control for the spontaneous hypersensitive rat (SHR) model is one such model as it has since been proposed as a putative model of depression. This is because WKYs demonstrated hormonal and physiological measures and depicted behavioural responses similar to those found in symptom-presenting depressive patients. These include, for instance, hyperreactivity to stress, behavioural inhibition, anxiety-like profile, dysregulation of hypothalamic-pituitary-adrenal (HPA) axis, increased adrenocorticotropic hormone (ACTH) and corticosterone (CORT) levels, neurochemical abnormalities in several systems (dopaminergic, serotonergic and noradrenergic) as well as in peripheral hormones such as thyroid-stimulating hormone (TSH) etc.1-6
So the WKY strain, which exhibits endogenous depressive-like behaviour is thought to be impaired in adaptive capabilities, making it more susceptible to exogenous stressors as it demonstrates hypersensitivity to stress with a propensity to develop stress-induced anxiety-like characteristics. However, the WKY strain harbours heterogeneity not found in other inbred strains, including greater behavioural and genetic variability, which has led to mixed results being obtained earlier from anxiety and learned helplessness measures with differences emerging between inbred strains of WKY.7
Despite this, the WKY rat may be suitable to unravel underlying phenomena that link depression and exogenously-induced stressors or inflammatory states8-11as in the case of a diathesis or double hit model, whether through activation of the HPA system or the immune system that lead to altered physiology. Sickness behaviour is an immunological/inflammatory model of depression or a non-specific reaction to various infectious and pro-inflammatory stimuli, such as LPS administration, which constitutes a well-established experimental approach to study the effects of an acute and transient immune activation on physiology and behaviour.8-12
Acting as a stressor,12 LPS activates common physiological responses (brain cytokine expression, HPA axis activation). How a susceptible WKY strain responds to extraneous immune stimulants would provide a window into understanding vulnerability. LPS elicits a strong immune response leading to the secretion of pro-inflammatory cytokines, which can act on the hypothalamus and other areas of the brain through humoral and nervous routes with profound behavioural deficits like prolonged sleepiness, depression, reduced levels of mobility, anxiety, food and water intake, rearing, grooming.13 These complex behavioural changes such as reduced general activity, reduced social motivation and fever response, collectively termed “sickness behaviour,”14,12include reduced exploration, increased anxiety, cognitive dysfunction, and social withdrawal in rodents.11-16 As WKY demonstrate great heterogeneity, it is essential to carry out baseline tests, assessing anxiety- and depressive-like measures. Towards this, the ideal paradigm is the elevated plus maze (EPM), which introduces a conflict between the animal’s inherent urge to explore new environments, vis-à-vis it's fear of open, brightly lit spaces. It is, therefore, best suited to assess anxiety-like behaviour. Anxiety and depression often demonstrate comorbidity. Typical features or subtypes of the depression syndrome are better coping style or increased resilience,17,18 which are ideally tested out in the forced swim test (FST) in rodents that detects coping strategies in response to stress and is a paradigm for behavioural despair. Immobility or passive behaviour is interpreted as behavioural despair, while swimming is coping with the stressor and climbing is more of defensive behaviour.
Neurochemical and behavioural responses are impacted by LPS administration. LPS induces profound cerebral changes in monoamine metabolism. For instance, at 2-4 hrs post LPS administration, monoaminergic transmission is on the rise with the serotonergic system and the HPA axis is activated.19 Brain areas such as the frontal cortex are stressor sensitive, while the hypothalamus is the seat of neuro vegetative symptoms associated with sickness behaviour, so we selected these brain areas for assaying ubiquitous acetylcholinesterase (AChE), monoamine oxidase (MAO) and stress marker neuronal nitric oxide synthase (NOS).
Peripherally, LPS induces inflammation by acting via macrophage TLRs. These activated macrophages initiate a cascade of events culminating in reactive oxygen species (ROS) and generation of free radicals, a reaction that leads to lipid peroxidation, as measured here by liver MDA levels wherein the phospholipid bilayer gets increasingly porous, culminating in necrosis. GSH, the primary non-protein sulfhydryl ubiquitous tripeptide catalyzed by glutamyl cysteinyl synthetase is a potent anti-oxidant scavenging molecule is, which donates its electron to ROS, thus lowering their adverse effect, so liver GSH activity was also measured.
Here, we first established whether the experimental WKY subjects demonstrate anxiety- and depressive-like profiles and then used them for further testing with LPS with behavioural measures being recorded in EPM 2 hours post-injection and a novel activity box 3 hours post-injection. Oxidative stress was quantified by performing biochemical assays of potent anti-oxidant molecules in the liver such as Glutathione (GSH) and levels of malondialdehyde (MDA) were used to assess lipid peroxidation in the liver. Quantification of ubiquitous central acetylcholine degrading enzyme AChE, monoaminergic modulator MAO and gaseous signalling molecule NOS, was carried out. As the immune system is activated in response to mitogens, such as LPS and stressors by inducing an increase in immune cell populations, especially lymphocytes and neutrophils, total and differential leukocyte count [total leukocyte count (TLC), and differential leukocyte count (DLC)] in blood circulation was carried out to detect leucocyte misdistribution. Thus, we screened for anxiety-related behaviours in adult male WKY vis-a-viz Wistar and observed LPS-induced changes in behaviour and neurochemical profiles in WKY.
Materials and Methods
Subjects
90-day old, male Wistar (n=8) and WKY (n=8) rats were procured from the ICMR-National Animal Resource Facility for Biomedical Research (NARFBR), Hyderabad, India and housed in the group under standard laboratory conditions with artificial 12h light/dark cycle (lights on at 7:00 h) at an ambient temperature of 22-24°C with free access to food and water. Animals were maintained in groups of 3-4, and experiments carried out according to the guidelines laid down by the Committee for Control and Supervision of Experiments on Animals, Government of India, as per the ARRIVE guidelines and were permitted by Institutional Animal Ethics Committee (No. IAEC/106/2011). All experiments were conducted in the light cycle (9:00–17:00 h). All behavioural recordings were carried out for 5 minutes under 8-8.5 lux as measured at the base of the arena. Recording done with a CCD camera (WV CP500; Panasonic) and data acquisition and analysis done with Ethovision® 9.0 (Noldus, Netherlands).
Baseline behaviour
Wistars (n=8) and WKY (n=8) rats were subjected to baseline testing in the elevated plus maze (EPM), habituated to the swim apparatus and 24hrs later tested in the forced swim test (FST).
Elevated Plus Maze (EPM)
The EPM test was carried out as described in detail elsewhere.20,21 Briefly open and closed arm time and entries along with locomotion were automatically quantified. Anxiety index was calculated as open arm time and entries about total time and total entries.22 Non-classical anxiety measures such as nose dips, stretch-attend postures 22,23 were also quantified.
Forced Swim Test (FST)
The modified FST protocol was adapted24, 25 and is a well-characterized paradigm to analyze depression-like behaviour in rodents. The FST was carried out as described in detail elsewhere.20,21
Lipopolysaccharide in WKY rats
24hrs later, WKY rats were randomly divided into two groups. N=4 were injected with 1mg/kg body weight of LPS (E.coli, serotype, Sigma) dissolved in saline i.p. with volume made up with saline to 1 ml. Controls (n=4) were injected with the same volume of the vehicle. Two hours after the injection, rats were placed into the EPM as described above.
Activity Box
Three hours after the injection, the rats were placed in the centre of an activity box which was an open cube of 44x44x44cms. The box was located 50 cms above the ground, and the animal’s behaviour was observed for 5 minutes. The observed parameters were ambulation and rearing.
Differential Leucocyte Counting
Immediate after exposure to the activity box, all animals were deeply anaesthetized, and blood collected for mono- and poly-morphonuclear leucocyte (PMNL) staining and quantification. Briefly, blood was smeared and stained with Leishman’s stain (Himedia) and observed under a Leica DM2500 and quantification were done using Leica Application Suite software.
Enzyme Assays
Liver and brain tissues of LPS-injected rats (n=4) and control rats (n=4) were taken and deep-frozen until further. Brain areas, frontal cortex and hypothalamus, were dissected out, tissues were homogenized, centrifuged and the supernatant used for spectrophotometric estimations of acetylcholinesterase, nitric oxide synthase, and monoamine oxidase. Liver tissues were homogenized, centrifuged and the supernatant used for spectrophotometric estimations of Glutathione (GSH), Lipid peroxidation by Malondialdehyde. Enzyme activity was expressed as specific activity in nanomoles or micromoles of the enzyme per mg of protein. 26-29
Statistical analysis
All data are expressed as group mean ± S.E.M and tested for statistical differences using t-test. Repeated measures t-test was used to detect differences between habituation and test in Wistar and WKY rats. Differences at p values less than 0.05 were considered significant.
Results
WKYs demonstrated increased anxiety levels with reduced open arm time in the EPM but did not demonstrate learned helplessness in the FST test. LPS induced a decrease in all parameters, notably distance moved and centre time when compared to saline-injected controls, though anxiety-related measures showed no difference. Risk assessment in the EPM and exploratory behaviour in the activity box was significantly reduced. These behavioural changes were accompanied by increased hypothalamic acetylcholinesterase activity and the concomitant decrease in MAO activity in both the frontal cortex and the hypothalamus. Activated brain substrates underlying this were prefrontal and piriform cortices.
EPM
As compared to the parent progenitor strain Wistars, WKYs demonstrated reduced open arm time and increased closed-arm time (fig. 1a,b). Though entries into closed and open arms were not significantly different (fig. 1d,e), the cumulative anxiety-related measures translated into increased anxiety levels in WKYs when compared to age-matched Wistars (fig. 1f). WKYs demonstrated significantly (tdf=14 = 4.014; p < 0.0017) reduced ambulation as assessed by distance moved when compared to Wistars. WKYs covered a distance of 9.46 ± 0.97m while Wistars covered close to double the distance at 17.93 ± 1.66m. Latency or time taken to enter the open arm was increased in WKYs (0.32 ± 0.12s) as against Wistars (0.51 ± 0.20s), though the difference was not significant (tdf=14 = 0.73; p > 0.05). Centre time was significantly reduced in WKY (fig. 1c).
Among the ethological measures, rearing behaviour was comparable between strains (fig. 1g). Risk assessment, as measured in stretch-attend postures wherein the animal while remaining in the confines of the closed arm stretches into the open arm, were significantly increased in WKY (fig. 1i). Head dips, indicative of risk-taking, wherein the animal positioned in the open arm dips its head, were significantly reduced in WKY (fig. 1h). For Mean ± SE values of anxiety-related and ethological measures, see fig.1.
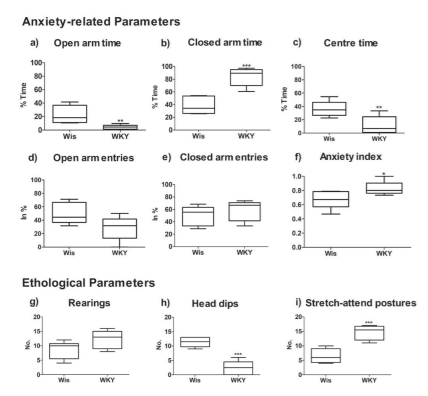
Fig. 1 Anxiety-related and ethological parameters in the EPM of WKYs vs. Wistars. Top panel: Duration spent in the a) open arm (tdf=14=3.35; p=0.0074) was significantly reduced in WKYs with a corresponding increase in b) closed arm time (tdf=14=5.84; p=0.0002), while c) centre time was also significantly reduced in WKYs (tdf=14=3.776; p=0.0026). Middle panel: Entries into the d) open arm (tdf=14=2.412; p=0.05) were significantly reduced in WKYs while those into the e) closed arm were comparable with Wistars (tdf=14=0.7699; p=0.4592). Anxiety index was significantly higher in WKYs than Wistars (tdf=14=2.636; p=0.0249). Lower panel: Ethological parameters: a) Rearing demonstrated a trend (tdf=14=2.073; p=0.0625) while stretch-attend postures (tdf=14=5.302; p=0.0003) were significantly increased in WKY. Head dips from the open arm were significantly reduced (tdf=14=7.571; p<0.0001) in WKYs. Whiskers represent minimum to maximum (variability in the samples); box represents occurrence of majority of the samples (75%; 25%), midline is the median.
FST
During the habituation exposure, WKY spent 25.05 ± 3.66% of the time in the FST immobile while Wistars were immobile for 36.74 ± 1.88%, the strain difference being significant (tdf=14 = 3.091; p = 0.0112). WKY demonstrated increased mobility or swimming in the FST with 28.79 ± 2.32%, when compared to Wistar who spent 22.15 ± 1.41% of the time swimming, the difference being significant (tdf=14 = 2.57; p = 0.0245). WKY demonstrated increased climbing behaviour in the FST with 42.03 ± 3.47% time spent in climbing, when compared to Wistar who spent 38.15 ± 2.04% time in climbing, the time being comparable (tdf=14 = 0.999; p > 0.05). No differences were observed in latency (time taken) to immobility (Wistar: 43.74 ± 5.54; WKY 49.80 ± 7.05; (tdf=14 = 0.6848; p > 0.05).
During the test exposure, WKY spent 9.08 ± 1.75% of the time in the FST immobile while Wistars spent 21.69 ± 3.92% of the time immobile, the difference being significant (tdf=14 = 2.560; p = 0.0284). WKY demonstrated increased swimming behaviour in the FST with 22.52 ± 0.89%, when compared to Wistar who spent 20.08 ± 1.11% time in swimming, though the difference was not significant (tdf=14 = 1.63; p > 0.05). WKY demonstrated increased climbing behaviour at 64.70 ± 4.10%, when compared to Wistar who spent 51.82 ± 4.96% time in climbing, the difference not being significant (tdf=14 = 1.957; p = 0.07). No differences were observed in latency to immobility (Wistar: 26.71 ± 5.78; WKY 31.73 ± 8.226; (tdf=14 = 0.5105; p > 0.05).
Hab vs Test
Differences between habituation and test are depicted in fig. 2. WKY took a lesser time to become immobile (fig. 2a), demonstrated significantly reduced immobility (fig. 2b) and increased climbing (fig. 2d) behaviour during the test. Swimming behaviour was comparable between habituation and test. Wistars, on the other hand, also demonstrated significantly reduced immobility (fig. 2b) and increased climbing (fig. 2d) behaviour during the test. Their latency to immobility (fig. 2a) and swimming behaviour (fig. 2c) were comparable between the two exposures. The repeated measures t statistic and p values are depicted in fig. 2.
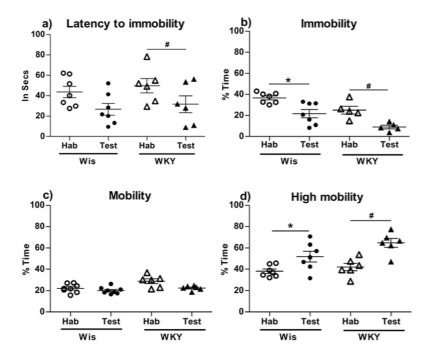
Fig. 2 Forced Swim Test. Differences between habituation and test. a) Latency/time taken to Immobility/become immobile: Wistar - tdf=7=1.231; p=0.273; WKY - tdf=7=2.645; p=0.0457; b) Immobility: Wistar - tdf=7=3.872; p=0.0117; WKY - tdf=7=3.331; p=0.0447; c) Mobility/Swimming behaviour: Wistar - tdf=7=1.291; p=0.2378; WKY - tdf=7=2.282; p=0.0714; d) High mobility/Climbing behaviour - Wistar - tdf=7=2.652; p=0.0380; WKY) - tdf=7=3.940; p=0.0110
LPS in WKY - EPM
LPS treatment reduced locomotory activity by inducing a significant (tdf=6 = 3.344; p = 0.0086) reduction in distance moved (Veh: 1.14 ± 0.10m vs. LPS: 0.59 ± 0.13m). Latency to enter the open arm was comparable, with Vehicle-treated animals taking 40.97 ± 19.36s while LPS-treated rats took 22.03 ± 12.87s to enter the open arm (tdf=6 = 0.8144; p > 0.05). Entries into the open arm were very few (Veh: 0.16 ± 0.02s vs. LPS 0.14 ± 0.08s; tdf=6 = 0.220; p > 0.05) as also closed arm entries (Veh: 0.86 ± 0.33s vs. LPS 0.91 ± 0.06s; tdf=6 = 0.902; p > 0.05).
LPS affected center time which was significantly (tdf=6 = 3.59; p = 0.007) reduced in LPS-treated rats (10.17 ± 2.86s) as compared to controls (34.75 ± 7.37s). Open arm time was significantly reduced in LPS-treated rats (Veh: 4.20 ± 1.18; LPS 0.70 ± 0.47; tdf=6 = 2.305; p = 0.05). Closed arm time was significantly (tdf=6 = 3.752; p = 0.005) higher in LPS-treated rats (287.2 ± 3.85s vs. Veh: 259.8 ± 6.98s). For % open arm time, % centre time and % closed arm time see fig. 3a.
LPS induced a decrease in the ethological parameters tested such as rearing frequency (Veh: 8.00 ± 0.55; LPS: 4.60 ± 1.29) which were significantly reduced (tdf=6 = 2.429, p = 0.041), as also stretch-attend postures from the safety of the closed arm onto the open arm (Veh: 8.60 ± 1.17; LPS: 3.80 ± 1.16 (tdf=6 = 2.921, p = 0.019).
LPS in WKY - Activity box
3 hours LPS injection, reduced ambulation was observed, with vehicle-treated WKY demonstrating 8.78 ± 0.56m while LPS-treated rats demonstrated reduced locomotor activity at 3.59 ± 0.60m moved; tdf=6 = 6.284; p ? 0.0001). Rearing behaviour was also influenced by LPS: vehicle-treated rats exhibited 7.18 ± 0.91 rears vs. LPS-treated rats which exhibited 3.20 ± 1.16 rears, the difference being significant (tdf=6 = 2.735; p = 0.023). There was no significant difference in centre duration with vehicle-treated rats depicting 2.90 ± 0.98s in the center of the activity box vs. LPS-treated rats which demonstrated 38.30 ± 30.64s, the difference being not significant (tdf=6 = 1.155; p = 0.2750). Neither was there any difference in time spent in the periphery (Veh: 297.3 ± 0.98s vs. LPS: 261.9 ± 30.64s; tdf=6 = 1.155; p > 0.05). For activity box measures of vehicle vs. LPS treated animals three hours following the LPS injection, see fig. 3b.
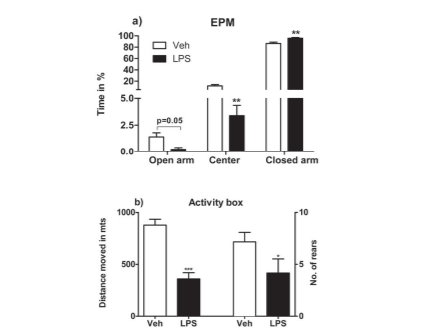
Fig. 3 a): In the EPM at 2 hrs post LPS injection, LPS-induced effects on open arm, centre and closed arm times. There was a significant reduction in centre time of LPS group (tdf=6=3.59; p=0.007), a significant increase in closed arm time (tdf=6=3.752; p=0.005) while open arm time just reached significance (tdf=6=2.305; p=0.05). For time in secs, see text. b) At 3hrs post LPS, distance moved and rearing behavior in the activity box. Distance moved: Veh: 8.783±0.5639m; LPS: 3.593±0.6035 (tdf=6=6.284; p<0.001). Rearing behaviour: Veh: 7.167± 0.91; LPS 3.20 ± 1.16 (tdf=6=2.735; p=0.023).
PMNL vs MNL
Differential leucocyte count in vehicle and LPS-treated groups demonstrated a decrease in Lymphocyte and monocyte counts in LPS-treated animals. The decrease in the lymphocytes (tdf=6 = 6.693; p<0.001) and monocytes (tdf=6=57.00; p = 0.002) was very significant. LPS treatment induced a significant rise in neutrophils (tdf=6 = -10.220; p < 0.001).
 Liver and Brain Enzyme Assays
Liver and Brain Enzyme Assays
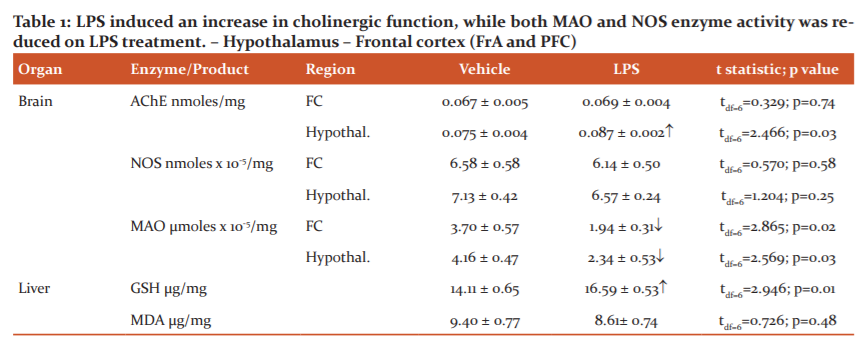
In the brain tissues, a significant increase in AChE levels was observed in the hypothalamus in LPS-treated rats, but no difference was observed in the frontal cortex. NOS levels were reduced on LPS treatment in both frontal cortex and hypothalamic regions, though group differences were not significant. MAO was significantly reduced in both the frontal cortex and hypothalamus (Table 1).
The liver tissues of LPS-treated rats demonstrated a significant increase in GSH activity when compared with vehicle-treated controls. Malondialdehyde levels were not affected by the LPS injections and remained at comparable levels in both treated and control groups. The effects of LPS on AChE, MAO and NOS enzyme activity in brain and liver antioxidant enzyme activity and lipid peroxidation are summarized in Table 1.
Discussion
It was essential to establish an anxiety- and depressive-like profile in WKY before using the WKY for the further challenge using LPS. Elevated plus-maze was used to screen for anxiety levels and risk assessment behaviours. WKY depicted increased anxiety-like behaviour in the EPM when compared to age-matched Wistars which was evident from the reduced time spent and reduced number of entries into aversive areas. These together with increased closed-arm time and despite comparable closed arm entries translated into increased anxiety in WKY as compared to age-matched Wistars. The number of closed arm entries which is the best measure of locomotor activity,28 was comparable indicating that the other behaviours were not confounded by a difference in locomotion.
Though the EPM is conventionally used to assess anxiety-like behaviour,30 it also enables assessment of some other complementary, ethological parameters that offer insights into behavioural and physiological effects in rodents, such as risk-taking vide head/nose dips, risk assessment vide stretch-attend postures etc. 31,32 The latter indicates that the animal is hesitant to move from its present location to a new position and thus a high frequency of these postures as was observed in the WKY indicates a higher level of anxiety. The reduced head/nose dips, indicative of reduced risk-taking, arose as a result of the reduced open arm entries.
In the FST, used to assess depressive-like behaviour, WKY demonstrated reduced immobility when compared to Wistars, though both strains did not demonstrate learned helplessness, the construct of the FST. This is at variance with another study on adult WKY study which showed increased immobility as indicative of helplessness with a propensity to develop stress-induced anxiety-like characteristics.33 Moreover, both strains demonstrated similar variations between the habituation and test exposures, with reduced immobility and increased struggling behaviour manifested in the test, though others suggest that strain-specific behavioural differences emerge between habituation and test.34 Here, sample size may have been short to bring out strain-specific differences, particularly in life-death situations, where both strains demonstrated comparable coping strategies as in swimming and comparable struggling as a defensive behaviour.
The similarities could be due to a modified protocol being used, or the size of the FST apparatus. It has also been suggested that the FST does not mimic the causal or phenomenological features of major depression35 and that FST measures define behavioural depression only operationally. Differences also may be less marked when WKYs are compared to a single strain (Wistars) as here, or other strains such as SD, SHR, FSL.1,36-39 The WKY rats may have been sensitized as latency to immobility is reduced during the test or may have been able to adapt to the aversive situation by demonstrating increased struggling or defensive behaviour. Better coping style or increased resilience is also considered typical features or subtypes of depression syndrome.References:
-
Paré WP. “Behavioral despair” test predicts stress ulcer in WKY rats. Physi Behav. 1989 Sep 1;46(3):483-7.
-
Will CC, Aird F, Redei EE. Selectively bred Wistar–Kyoto rats: an animal model of depression and hyper-responsiveness to antidepressants. Mol Psych. 2003 Nov;8(11):925-32.
-
Braw Y, Malkesman O, Merenlender A, Bercovich A, Dagan M, Overstreet DH, Weller A. Withdrawal emotional-regulation in infant rats from genetic animal models of depression. Behav Bra Res. 2008 Nov 3;193(1):94-100.
-
Paré WP, Tejani-Butt S, Kluczynski J. The emergence test: effects of psychotropic drugs on neophobic disposition in Wistar Kyoto (WKY) and Sprague Dawley rats. Progress Neuro-psychopharm Biol Psych. 2001 Nov.
-
Renoir T, Pang TY, Lanfumey L. Drug withdrawal-induced depression: serotonergic and plasticity changes in animal models. Neurosci Biobeh Rev. 2012 Jan 1;36(1):696-726.
-
Rao RM, Sadananda M. Strain-and context-based 50 kHz ultrasonic vocalizations and anxiety behaviour in the Wistar-Kyoto rat. J Biosci. 2015 Sep 1;40(3):561-70.
-
Mehta NS, Wang L, Redei EE. Sex differences in depressive, anxious behaviours and hippocampal transcript levels in a genetic rat model. Genes, Bra Beha. 2013 Oct;12(7):695-704.
-
Yirmiya R. Behavioral and psychological effects of immune activation: implications for'depression due to a general medical condition. Curr Opin Psych. 1997 Nov 1;10(6):470-6.
-
Yirmiya R. Topics in Review: Depression in medical illness: The role of the immune system. West J Med. 2000 Nov;173(5):333.
-
Dantzer R. Cytokine-induced sickness behavior: where do we stand?. Brai Behav Immu. 2001 Mar 1;15(1):7-24.
-
Dantzer R, O'Connor JC, Freund GG, Johnson RW, Kelley KW. From inflammation to sickness and depression: when the immune system subjugates the brain. Natu Revi neuroscience. 2008 Jan;9(1):46-56.
-
Arakawa H, Blandino Jr P, Deak T. Central infusion of interleukin-1 receptor antagonist blocks the reduction in social behavior produced by prior stressor exposure. Physi Beha. 2009 Aug 4;98(1-2):139-46.
-
Kelley KW, Bluthé RM, Dantzer R, Zhou JH, Shen WH, Johnson RW, Broussard SR. Cytokine-induced sickness behaviour. Brain, Beha Immu. 2003 Feb 15;17(1):112-8.
-
Kent S, Bluthé RM, Kelley KW, Dantzer R. Sickness behaviour as a new target for drug development. Tre Pharmacol Sci. 1992 Jan 1;13:24-8.
-
Benson S, Engler H, Schedlowski M, Elsenbruch S. Experimental endotoxemia as a model to study neuroimmune mechanisms in human visceral pain. Annals of the New York. Aca Sci. 2012 Jul;1262(1):108-17.
-
Cunningham C, Campion S, Lunnon K, Murray CL, Woods JF, Deacon RM, Rawlins JN, Perry VH. Systemic inflammation induces acute behavioural and cognitive changes and accelerates neurodegenerative disease. Bio Psyc. 2009 Feb 15;65(4):304-12.
-
Russo SJ, Murrough JW, Han MH, Charney DS, Nestler EJ. Neurobiology of resilience. Nat Neurosci. 2012 Nov;15(11):1475-84.
-
Krishnan V, Han MH, Graham DL, Berton O, Renthal W, Russo SJ, LaPlant Q, Graham A, Lutter M, Lagace DC, Ghose S. Molecular adaptations underlying susceptibility and resistance to social defeat in brain reward regions. Cell. 2007 Oct 19;131(2):391-404.
-
Pitychoutis PM, Nakamura K, Tsonis PA, Papadopoulou-Daifoti Z. Neurochemical and behavioural alterations in an inflammatory model of depression: sex differences exposed. Neurosci. 2009 Apr 10;159(4):1216-32.
-
D'Souza D, Sadananda M. Anxiety-and depressive-like profiles during early-and mid-adolescence in the female Wistar Kyoto rat. Int J Devp Neurosci. 2017 Feb 1;56:18-26.
-
Shetty RA, Sadananda M. Immediate and delayed anxiety-and depression-like profiles in the adolescent Wistar-Kyoto rat model of endogenous depression following postweaning social isolation. Behavi Brai Rese. 2017 Mar 1;320:323-32.
-
Cohen H, Matar MA, Buskila D, Kaplan Z, Zohar J. Early post-stressor intervention with high-dose corticosterone attenuates posttraumatic stress response in an animal model of posttraumatic stress disorder. Biolog Psych. 2008 Oct 15;64(8):708-17.
-
Melo TG, Izídio GS, Ferreira LS, Sousa DS, Macedo PT, Cabral A, Ribeiro AM, Silva RH. Antidepressants differentially modify the extinction of an aversive memory task in female rats. Prog Neuro-Psychopharmacol Bio Psych. 2012 Apr 27;37(1):33-40.
-
Porsolt RD, Anton G, Blavet N, Jalfre M. Behavioural despair in rats: a new model sensitive to antidepressant treatments. Euro J Pharmac. 1978 Feb 15;47(4):379-91.
-
Detke MJ, Rickels M, Lucki I. Active behaviors in the rat forced swimming test differentially produced by serotonergic and noradrenergic antidepressants. Psychopharmac. 1995 Sep 1;121(1):66-72.
-
Ellman GL, Courtney KD, Andres Jr V, Featherstone RM. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmac. 1961 Jul 1;7(2):88-95.
-
Dawson TM, Bredt DS, Fotuhi M, Hwang PM, Snyder SH. Nitric oxide synthase and neuronal NADPH diaphorase are identical in the brain and peripheral tissues. Procee Nati Acad Sci. 1991 Sep 1;88(17):7797-801.
-
Dawson GR, Crawford SP, Stanhope KJ, Iversen SD, Tricklebank MD. One-trial tolerance to the effects of chlordiazepoxide on the elevated plus-maze may be due to locomotor habituation, not repeated drug exposure. Psychoph. 1994 Jan 1;113(3-4):570-2.
-
Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by the thiobarbituric acid reaction. Analy Biochem. 1979 Jun 1;95(2):351-8.
-
Walf AA, Frye CA. The use of the elevated plus-maze as an assay of anxiety-related behaviour in rodents. Nature Proto. 2007 Feb;2(2):322-8.
-
Pellow S, Chopin P, File SE, Briley M. Validation of open: closed arm entries in an elevated plus-maze as a measure of anxiety in the rat. J Neurosci Meth. 1985 Aug 1;14(3):149-67.
-
File SE, Zharkovsky A, Hitchcott PK. Effects of nitrendipine, chlordiazepoxide, flumazenil and baclofen on the increased anxiety resulting from alcohol withdrawal. Progr Neuro-Psychopharmac Bio Psych. 1992 Jan 1;16(1): IN3-93.
-
McAuley JD, Stewart AL, Webber ES, Cromwell HC, Servatius RJ, Pang KC. Wistar–Kyoto rats as an animal model of anxiety vulnerability: Support for a hypervigilance hypothesis. Behav Bra Res. 2009 Dec 1;204(1):162-8.
-
Armario A, Nadal R. Individual differences and the characterization of animal models of psychopathology: a strong challenge and a good opportunity. Fronti Pharmac. 2013 Nov 8;4:137.
-
Drolet G, Proulx K, Pearson D, Rochford J, Deschepper CF. Comparisons of behavioural and neurochemical characteristics between WKY, WKHA, and Wistar rat strains. Neuropsychophar. 2002 Sep 1;27(3):400-9.
-
Ducottet C, Belzung C. Correlations between behaviours in the elevated plus-maze and sensitivity to unpredictable subchronic mild stress: evidence from inbred strains of mice. Behavl Bra Res. 2005 Jan 6;156(1):153-62.
-
Ferguson SA, Gray EP. Ageing effects on elevated plus-maze behaviour in spontaneously hypertensive, Wistar–Kyoto and Sprague–Dawley male and female rats. Physi Beha. 2005 Aug 7;85(5):621-8.
-
Bison S, Carboni L, Arban R, Bate S, Gerrard PA, Razzoli M. Differential behavioural, physiological, and hormonal sensitivity to LPS challenge in rats. Int J Interf, Cytoki and Mediator Res. 2008 Dec 18;1:1-3.
-
Rauhut AS, Zentner IJ, Mardekian SK, Tanenbaum JB. Wistar Kyoto and Wistar rats differ in the affective and locomotor effects of nicotine. Physio Behav. 2008 Jan 28;93(1-2):177-88.
-
Sah SP, Tirkey N, Kuhad A, Chopra K. Effect of quercetin on lipopolysaccharide induced-sickness behaviour and oxidative stress in rats. Ind J Pharmac. 2011 Apr;43(2):192-96.
-
Maier SF, Watkins LR. Cytokines for psychologists: implications of bidirectional immune-to-brain communication for understanding behaviour, mood, and cognition. Psych Rev. 1998 Jan;105(1):83.
-
Maier SF, Watkins LR. Bidirectional communication between the brain and the immune system: implications for behaviour. Anim Behav. 1999 Apr 1;57(4):741-51.
-
Shetty RA, Sadananda M. Brief social isolation in the adolescent Wistar-Kyoto rat model of endogenous depression alters corticosterone and regional monoamine concentrations. Neurochem Res. 2017 May 1;42(5):1470-7.
-
Bassi GS, Kanashiro A, Santin FM, de Souza GE, Nobre MJ, Coimbra NC. Lipopolysaccharide?induced sickness behaviour evaluated in different models of anxiety and innate fear in rats. Bas Clinpharmacology Toxic. 2012 Apr;110(4):359-69.
-
Walker AK, Nakamura T, Byrne RJ, Naicker S, Tynan RJ, Hunter M, Hodgson DM. Neonatal lipopolysaccharide and adult stress exposure predisposes rats to anxiety-like behaviour and blunted corticosterone responses: implications for the double-hit hypothesis. Psychoneuroend. 2009 Nov 1;34(10):1515-25.
-
Tyagi E, Agrawal R, Nath C, Shukla R. Influence of LPS-induced neuroinflammation on acetylcholinesterase activity in rat brain. J Neuroimm. 2008 Dec 15;205(1-2):51-6.
-
Tyagi E, Agrawal R, Nath C, Shukla R. Effect of melatonin on neuroinflammation and acetylcholinesterase activity induced by LPS in rat brain. Euro J Pharmac. 2010 Aug 25;640(1-3):206-10.
-
De La Garza II R, Mahoney III JJ. A distinct neurochemical profile in WKY rats at baseline and in response to acute stress: implications for animal models of anxiety and depression. Brain Res. 2004 Sep 24;1021(2):209-18.
-
Singh AK, Jiang Y. How do peripheral lipopolysaccharide induce gene expression in the brain of rats?. Toxicology. 2004 Sep 1;201(1-3):197-207.
-
Chen Z, Jalabi W, Shpargel KB, Farabaugh KT, Dutta R, Yin X, Kidd GJ, Bergmann CC, Stohlman SA, Trapp BD. Lipopolysaccharide-induced microglial activation and neuroprotection against experimental brain injury are independent of hematogenous TLR4. Journal of Neuroscience. 2012 Aug 22;32(34):11706-15.
-
Sun S, Zhang H, Xue B, Wu Y, Wang J, Yin Z, Luo L. Protective effect of glutathione against lipopolysaccharide-induced inflammation and mortality in rats. Inflammation Research. 2006 Nov 1;55(11):504-10.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License