IJCRR - 13(12), June, 2021
Pages: 23-26
Date of Publication: 22-Jun-2021
Print Article
Download XML Download PDF
Macrophage Colony-Stimulating Factor (MCSF) and Receptor Activator of NF?B Ligand (RANKL) in Osteoclastogenesis
Author: Prisca Pakan, Abeer Farhan Alanazi, Nicholas Andronicos
Category: Healthcare
Abstract:Introduction: Osteoclasts are defined as bone-resorbing cells which have an important function in skeletal development as well as in bone remodelling in the adult stage. Two significant cytokines stimulate the differentiation of osteoclasts from cells of the monocyte/macrophage; the monocyte/macrophage colony-stimulating factor (M-CSF) and receptor activation of NF-κB ligand (RANKL). For osteoclast precursors to proliferate, MCSF needs to bind to its receptor c-Fms which activates the signalling pathway required. Furthermore, RANKL which is the elementary differentiation factor of osteoclast stimulates the process through the gene expression control by activating its receptor. Aims: This study aims to demonstrate osteoclastogenesis using RAW264.7 line cell in vitro and identify the alterations in gene expression of the progenitor cells that typify osteoclast cells. Methodology: The study was conducted using hematopoietic precursor cells to examine the cellular differentiation into osteoclast under the control of RANKL. Furthermore, microarrays were used to accurately analyze the expression of the genes of interest by exhibiting the gene expressions changes and expected regulations of osteoclasts marker genes such as Glyceraldehyde 3-phosphate dehydrogenase (GAPDH), Tartrate-resistant acid phosphate (TRAP) and c-FMS. Results: We have identified that the upregulation of TRAP mRNA expression indicated the differentiation of osteoclast cells in the progenitor cells were induced by RANKL. Conclusion: The presence of Macrophage Colony-Stimulating Factor (MCSF) stimulates the differentiation of osteoclast progenitors into osteoclasts in the presence of RANKL.
Keywords: Macrophage Colony-Stimulating Factor, Osteoclast, Receptor Activator of NF?B Ligand, RANKL
Full Text:
INTRODUCTION
Macrophages change their physiology in response to environmental changes that may give incremental changes in the cell population. The established pathway of macrophages activation is through the pathway of Interferon-¥-dependent activation by T helper 1. Macrophages function as human’s immune system that eradicates infectious agents, cancer cells and maintaining tissue homeostasis.1 Macrophage colony-stimulating factor (MSCF) is one of the primary cytokines in osteoclast genesis for the proliferation and differentiation of osteoclast precursors.2 MCSF is differentiated into TRAP, which indicated positive osteoclast.3
The osteoclast is defined as cells that developed from macrophages hematopoietic lineage; conforms to the bone matrix and degrade acid as well as lytic enzymes into the extracellular compartment.4,5 Furthermore, osteoclast is an important factor in bone remodelling to regulate and function normally throughout human life. The disproportion of bone remodelling may cause several skeletal muscle diseases such as osteoporosis, periodontal disease, rheumatoid arthritis, myeloma and metastatic cancers.6,7,8 The knowledge of the stimulation and activation of osteoclast cell is required to unfold the process in bone metabolism and remodelling to formulate therapeutic agents for these diseases.7 In addition, Lorenzo (2017) stated that physiological and pathophysiological effects of TNF related cytokines, such as RANKL on bone mass and how it initiates osteoclast genesis, are also important to appreciate.
RANKL and polypeptide growth factor CSF-1 (MCSF) are required to induce the expression of osteoclast; including Tartrate-resistant Acid Phosphate (TRAP), Cathepsin K (CATK), Calcitonin receptor.8 When activated by RANKL, the progenitor cells are subject to make structural alterations to reabsorb bone.9 Lytic enzymes TRAP and pro CATK are then exported into the reabsorption region.10 This process deteriorates the underlying bone and the degraded products are subsequently processed within the osteoclast and discharged into the circulation system.11
In the present study, we propose a method to investigate cell differentiation of macrophages into osteoclasts by inducing RANKL and MCSF and subsequently analyze the gene expression patterns during in vitro differentiation of RAW264.7 cell line. The results will determine whether RANKL and MSCF as primary cytokines can upregulate the genes expression of the encoding receptor of osteoclast such as TRAP.
Materials and METHODs
Harvesting the RAW264.7 cells. To culture the mouse myeloid/macrophage tumour cell line, RAW264.7, a 5 ml of the culture medium in the dish of RAW264.7 cells were gently squirted over the entire inside bottom of the dish to dislodge the cells. This procedure was repeated several times until the hazy layer of the cells was dislodged and suspended in the medium. The cell suspension was span at 1000 rpm for 5 minutes at room temperature. The supernatant was discarded and another 10 ml of complete medium was added to the cell pellet to suspend the cell again.
Cell differentiation was performed by incubation in complete medium with RANKL (R) for 1 and 7 days (R1, R7; respectively) and without RANKL (U) for 1 day and 7 days (U1 and U7; respectively). The culture medium was replaced on day 3 and 6 of treatment. In addition, other cell cultures were prepared for differentiation by incubating cell suspension in the tissue culture incubator (95% air/5% CO2); these cells were harvested on the next day for Ribonucleic acid (RNA) preparation.
Meanwhile, the remaining volume in tubes U (without RANKL) and R (with RANKL) was used to set up separate cultures for cell staining on the next two days.
Cell fixation, staining and phenotypic analysis. Both the cell cultures prepared were used. Cell fixation was achieved by using cell fixative and must be performed in a fume hood by gently adding 0.5 mL of cell fixative per well and leave in the fume hood for 30 seconds only and proceed with the preparation of diazotized Tartrate-Resistant Acid Phosphatase (TRAP) stain. A light microscope was used to examine the phenotype of the stained cells.
Cell lysis and RNA extraction. Using Aurum™ Total RNA Mini Kit, the procedure was conducted according to the manufacturer’s protocol.
Digestion of genomic DNA. The purified RNA acquired was treated with RNase-free DNase I to degrade any genomic DNA remaining in the sample.
First-strand cDNA synthesis. To reverse transcribe the mRNA in the samples in obtaining the first-strand cDNA was done using an INVITROGEN SuperScript III First-Strand Synthesis SuperMix-for-RT-PCR kit. This included RNase H digestion of the RNA strand in the cDNA/RNA duplex.
RT-PCR analysis. This assayed the expression level of three genes: (i) glyceraldehyde 3-phosphate dehydrogenase (GAPDH), (ii) tartrate-resistant acid phosphatase (TRAP), and (iii) Fms.
Agarose gel electrophoresis. The electrophoresis was run with 80 voltages for approximately 45 minutes on a 1.5% TAE agarose gel.
RESULTS and discussion Cell fixation, staining and phenotypic analysis
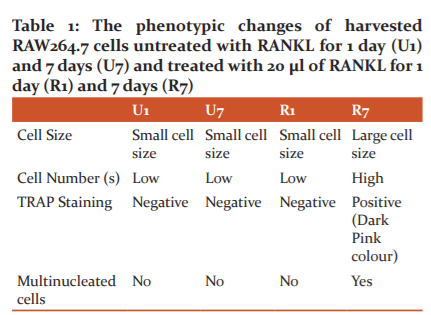
The cell size and population analysis in the untreated cells with RANKL (U1 and U7) showed relatively below the range of cell incubated with RANKL for 7 days (R7). Although R1 cell was treated with RANKL, the treatment was only for 1 day and therefore, the result is similar to the RANKL untreated cells. TRAP staining positive cells were apparent in the 7 days of RANKL treatment (R7). The multinucleated cells were exhibited by R7, as expected due to the cell sizes of R7 which were larger and hence indicated that it had more nuclei (Fig 1,2,3 and Table 1).
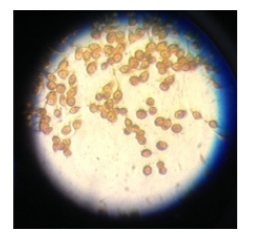
Figure 1. The light microscopic images show the cell population and cell morphological difference of which was not induced by RANKL even after incubated for 7 days (U7)
*U1 = incubation of cells without RANKL treatment for 1 day
*U7= incubation of cells without RANKL treatment for 7 days
*R1= incubation of cells with RANKL treatment for 1 day
*R7= incubation of cells with RANKL treatment for 7 days
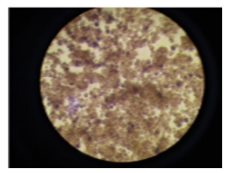
Figure 2. Culture cell induced by 20 µl of RANKL for 7 days (R7). This also represented the TRAP staining activity and the multinucleated cell.
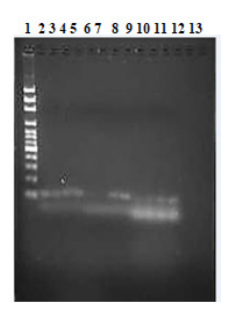
Figure 3. The agarose electrophoresis of the cDNA product
Using gene expression changes to monitor macrophage and osteoclast differentiation
After extracting RNA, the next step was the RT-PCR procedure to see the gene expression of macrophages and osteoclast cell differentiation induced by RANKL. The RT-PCR assay was used to analyse the expression of Glyceraldehyde 3-phosphate dehydrogenase (GAPDH), Tartrate-resistant acid phosphate (TRAP) and c-FMS, which were prepared into 12 tubes and loaded in a certain order.
The result of agarose gel electrophoresis of the PCR products shown in Fig 3 showed relatively precise expected amplifications from the genes. It can be seen that the lanes of control (GAPDH) were evenly and constantly expressed while TRAP and c-FMS lanes were also expressed but in different intensity. This means that RANKL were able to induce the expression of TRAP mRNA.
DISCUSSION
MCSF (Macrophage Colony Stimulating Factor) is expressed constitutively in the bone microenvironment in response to the incremental change of PTH and inflammatory molecules such TNF or RANKL.12, In this study, osteoclast cells were differentiated in vitro using murine stem cell bone marrow. Due to the presence of MCSF, macrophages were differentiated and RANKL activated TRAP which is the marker for osteoclast. Consequently, the mononucleosis progenitor cells altered the motif of their gene expression. These occurred by cells migrating and fusing and thus formed cells with multiple nuclear. As cells continue fusing, the cells get larger and become multinucleated. On day 7 of incubation (Fig 2), it had reached sufficient time for the RANKL concentration, the huge circular-shaped cells continue to fused into large cytoplasmic masses, therefore the cells density continue to increase.13
GAPDH
Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as a comparison to gene expressions data due to its constitutively expressed gene or as referred to as a “housekeeping” gene.14 It is also asserted that although several other “housekeeping” genes might be expressed differently between body tissues, it is proven that GAPDH can be used as the internal control for the normalisation in gene expression data, particularly during apoptosis. As housekeeping genes, a constant level of expression in the tissue is supposedly preserved. This is depicted in the result in both Fig 3 which in the lane 2, 3, 4, and 5 the stains were amplified in a stabilized manner. Therefore, these amplifications are not related to whether osteoclasts are associated with this effect or not, but GAPDH is used as the internal control.
TRAP
TRAP is a cytochemical marker of osteoclasts and its biochemical concentration in serum can be used to analyze osteoclast function and levels of bone resorption because TRAP is secreted in large amount by osteoclasts.15 Based on the ground of this, lane 6, 7, 8 and 9 (Fig 3) there were gene expressions on these, however, they are expressed in a different degree. Lane 6 and 7 (Fig 3) were untreated RANKL cells on day 1 and day 7 (U1 and U7), therefore, the gene expression of TRAP was not as much as in lane 8 and 9 (R1 and R7) which were treated with RANKL. The overexpression in lane 8 and 9 are most likely due to the existence of RANKL.
C-FMS
C-FMS is a precursor for the mature and late stage of osteoclastogenesis.16 As shown in Fig 3, although lane 10 until 13 were expressed, it can be seen that lane 13 which is RANKL induced for 7 days exhibited a more vivid stain, which indicated a greater expression.
CONCLUSIONS
RANKL were able to activate the expression of TRAP mRNA. TRAP is a cytochemical marker of osteoclasts; therefore, the positive assay in the culture cell of day 7 treatment with RANKL emphasizes the cellular differentiation of osteoclasts cells. Thus, the presence of Macrophage Colony-Stimulating Factor (MCSF) may stimulate the differentiation of osteoclast progenitors into osteoclasts in the presence of RANKL.
ACKNOWLEDGEMENT
This research was fully supported and funded by the University of New England, Australia. There was no conflict of interest in this research.
Authors contribution: Prisca Pakan and Nicholas Andronicos both designed the project and protocols. Prisca carried out the whole experiment, supervised by Nicholas. Prisca performed the data analysis and wrote the manuscript. Nicholas was responsible for the directing and planning of the research.
References:
1. Um Y, Eo HJ, Kim HJ, Kim K, Jeon KS, Jeong JB. Wild simulated ginseng activates mouse macrophage, RAW264.7 cells through TRL2/4-dependent activation of MAPK, NF-κB and PI3K/AKT pathways. J Ethnopharmacol. 2020 Dec 5;263:113218.
2. Hodge JM, Collier FM, Pavlos NJ, Kirkland MA, Nicholson GC. M-CSF Potently Augments RANKL-Induced Resorption Activation in Mature Human Osteoclasts. Laudet V, editor. PLoS One [Internet]. 2011 Jun 29 [cited 2020 Oct 12];6(6):e21462. Available from: https://dx.plos.org/10.1371/journal.pone.0021462
3. Ruef N, Dolder S, Aeberli D, Seitz M, Balani D, Hofstetter W. Granulocyte-macrophage colony-stimulating factor-dependent CD11c-positive cells differentiate into active osteoclasts. Bone. 2017 Apr 1;97:267–77.
4. Akbar MA, Nardo D, Chen MJ, Elshikha AS, Ahamed R, Elsayed EM, et al. α-1 antitrypsin inhibits RANKL-induced osteoclast formation and functions. Mol Med [Internet]. 2017 Mar 15 [cited 2020 Oct 6];23(1):57–69.
5. Butola LK, Anjanker A, Vagga A, Kaple MN. Endogenous factor and pathophysiology of rheumatoid arthritis: An autoimmune disease from decades. Int J Curr Res Rev. 2020;12(22):34–40.
6. Lou Z, Peng Z, Wang B, Li X, Li X, Zhang X. miR-142-5p promotes the osteoclast differentiation of bone marrow-derived macrophages via PTEN/PI3K/AKT/FoxO1 pathway. J Bone Miner Metab [Internet]. 2019 Sep 17
7. Rath, K., Mohanty, B., B., Kumar, S., Nayak, P., Sahoo J. Do Statins Have Potential As Anti Osteoporotic Drugs and Can They Be Used for Prevention or Targeting Osteoporosis. Int J Current Res Rev. 2013;05(04):73–81.
8. Darshan, D.D. Saqib H., Anir, K. Vijay, K, C N. Role of Drugs in Orthodontic Tooth Movement. Int J Current Res Rev. 2012;04(04):59–62.
9. Song C, Yang X, Lei Y, Zhang Z, Smith W, Yan J, et al. Evaluation of efficacy on RANKL induced osteoclast from RAW264.7 cells. J Cell Physiol [Internet]. 2019 Jul 1 [cited 2020 Oct 6];234(7):11969–75. Available from: https://pubmed.ncbi.nlm.nih.gov/30515780/
10. Xu F, Teitelbaum SL. Osteoclasts: New Insights [Internet]. Vol. 1, Bone Research. Sichuan University; 2013 [cited 2020 Oct 12]. p. 11–26. Available from: /pmc/articles/PMC4472093/?report=abstract
11. Ono T, Nakashima T. Recent advances in osteoclast biology [Internet]. Vol. 149, Histochemistry and Cell Biology. Springer Verlag; 2018 [cited 2020 Oct 6]. p. 325–41. Available from: https://pubmed.ncbi.nlm.nih.gov/29392395/
12. Kimura K, Kitaura H, Ishida M, Hakami Z, Saeed J, Sugisawa H, et al. Effect of macrophage colony-stimulating factor receptor c-Fms antibody on lipopolysaccharide-induced pathological osteoclastogenesis and bone resorption. In: Interface Oral Health Science 2014: Innovative Research on Biosis-Abiosis Intelligent Interface [Internet]. Springer Japan; 2015 [cited 2020 Oct 12]. p. 259–67. Available from: https://link.springer.com/chapter/10.1007/978-4-431-55192-8_22
13. Lorenzo J. The many ways of osteoclast activation [Internet]. Vol. 127, Journal of Clinical Investigation. American Society for Clinical Investigation; 2017 [cited 2020 Oct 6]. p. 2530–2. Available from: https://pubmed.ncbi.nlm.nih.gov/28530641/
14. Qu X, Mei J, Yu Z, Zhai Z, Qiao H, Dai K. Lenalidomide regulates osteocytes fate and related osteoclastogenesis via IL-1β/NF-κB/RANKL signaling. Biochem Biophys Res Commun. 2018 Jun 22;501(2):547–55.
15. Thummuri D, Jeengar MK, Shrivastava S, Nemani H, Ramavat RN, Chaudhari P, et al. Thymoquinone prevents RANKL-induced osteoclastogenesis activation and osteolysis in an in vivo model of inflammation by suppressing NF-KB and MAPK Signalling. Pharmacol Res. 2015 Jun 11;99:63–73.
16. Zhou C, Liu W, He W, Wang H, Chen Q, Song H. Saikosaponin a inhibits RANKL-induced osteoclastogenesis by suppressing NF-κB and MAPK pathways. Int Immunopharmacol. 2015 Mar 1;25(1):49–54.
17. Nishimura K, Shindo S, Movila A, Kayal R, Abdullah A, Savitri IJ, et al. TRAP-positive osteoclast precursors mediate ROS/NO-dependent bactericidal activity via TLR4. Free Radic Biol Med. 2016 Aug 1;97:330–41.
18. Yeo CE, Kang WY, Seong SJ, Cho S, Lee HW, Yoon YR, et al. Neuromedin B and its receptor silencing suppresses osteoclast generation by modulating precursor proliferation via M-CSF/c-Fms/D-type cyclins. Exp Cell Res. 2017 Oct 1;359(1):112–9.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License