IJCRR - 2nd Wave of COVID-19: Role of Social Awareness, Health and Technology Sector, June, 2021
Pages: 105-108
Date of Publication: 11-Jun-2021
Print Article
Download XML Download PDF
Inconclusive in COVID-19 Reverse Transcriptase-Polymerase Chain Reaction Test: A Diagnostic Dilemma
Author: Sikdar Subhendu, Bhunia Somnath, Majumdar Amit Kumar, Sarkar Soma, Ganguly Bhattacharjee Swagata
Category: Healthcare
Abstract:Introduction: Cases of COVID 19 pandemic in India has been reported since 30th January 2020. Cases are detected by RTqPCR targeting one or more gene (E/S/N/RdRp/ORF1) based on ICMR guidelines. Results are to be reported as 'Inconclusive' if only one target gene is detected in multiplex qPCR. Objective: The present study is aimed to analyze the \"Inconclusive\" results based on the Ct value of the detected target gene, duration of symptoms of cases and by repeat testing with another fresh sample after 3 days. Methods: This cross-sectional observational study was conducted from June 2020 to December 2020 in a tertiary care hospital in Kolkata. Nasopharyngeal and throat swab from suspected cases were collected and sent to our laboratory for RT-qPCR. Results: A total of 94,443 samples were tested in our laboratory for seven months. 8,455 (8.95%) samples were found to be inconclusive. Out of them, 1287 (15.22%) cases became positive and 3858 (45.63%) became negative after retesting with another fresh sample after 3 days. 1540 (18.21%) cases were lost for follow up. All inconclusive results were correlated with Ct value and duration of the disease symptoms. Maximum cases (64.96%) with Ct value 36 became negative on retesting. Most of the cases (28.36%) had symptoms of less than 2 days duration, among positive cases, whereas, 36.86% cases had symptoms of more than 12 days duration among negative cases. 1770 (20.94%) samples were found to be inconclusive again on retesting after 3 days. Most of these cases (46.55%) had a Ct value within 34- 36 on the first test. Conclusion: All inconclusive samples should always be subjected to further testing after 3 days. There is a chance to get positive results from inconclusive cases whose Ct value is less than 34. Quality control and quality assurance of all processes should be done to check any pre-analytical or analytical fallacies. Clinicians and patients both are to be educated about the probable reasons of inconclusive also
Keywords: COVID 19, Ct value, Inconclusive, RT-PCR, Target gene, Retesting
Full Text:
Introduction
An outbreak of pneumonia of unknown aetiology, which was identified first at Wuhan city in Hubei province of China in December 2019, was caused by a new strain of coronavirus named as 2019 novel coronavirus (2019-nCoV) or SARS-CoV 2.1 This virus rapidly spread across the globe, and subsequently a pandemic was declared on March 11, 2020.2 Since its global recognition, the novel coronavirus disease (COVID-19) pandemic has spread to over 200 countries in less than five months.3 In India, the first case of COVID-19 was reported on January 30, 2020.4 As of February 7, 2021, 10,827,314 laboratory-confirmed cases and 1,55,032 deaths were reported from India. The case reporting is based on the testing of individuals by real-time reverse transcription-polymerase chain reaction (RT-qPCR).5 It is performed by taking nasopharyngeal swabs or throat swab or saliva. A range of RNA target gene is used for detection by different manufacturers, mostly targeting the envelope (E), nucleo-capsid (N), spike (S), RNA-dependent RNA polymerase (RdRp), and ORF1 (Open reading frame)genes.6
In COVID-19 infected symptomatic individuals, viral load can be detected early from day one of symptoms and it peaks within the first seven days of onset of symptoms. This viral load can be measured by the cycle threshold (Ct) value, which is the number of replication cycles required to produce a fluorescent signal. Thus lower Ct values representing higher viral RNA loads. The positivity of the samples starts to decline by 3rd week and subsequently becomes undetectable.6 If only one SARS CoV-2 target gene is detected in the test (multiplex SARS CoV-2 RT PCR test) with valid internal control, the result should be interpreted as Inconclusive and repeated the test.7 Inconclusive SARS CoV-2 reverse transcription-polymerase chain reaction (RT-PCR) reports for the detection of infection in symptomatic patients or during the screening of asymptomatic contacts can cause clinical, diagnostic and infection control uncertainty.8
This study aims to analyze inconclusive results of samples tested at our Centre based on Ct value, duration of symptoms at the time of testing and the results of repeat testing.
Materials and Methods
The study was carried out over 94,443 suspected symptomatic COVID 19 patients as well as asymptomatic groups, such as high-risk contacts or high-risk healthcare workers, as per ICMR guideline9 from June 2020 to December 2020 at COVID laboratory in the Department of Microbiology of Nil Ratan Sircar Medical College & Hospital, Kolkata.
Sample collection & transport
Nasopharyngeal swab and throat swab were collected in Viral transport media from suspected cases in different wards and designated COVID wards of NRS Medical College & Hospital as per ICMR guidelines10 and sent to our Laboratory.
Samples from other district and rural hospitals were also sent to our laboratory as stated by the West Bengal Department of Health and Family Welfare updated from time to time.
Testing and collection of Data
All the samples were processed in our Laboratory as per standard protocol and tested by quantitative reverse transcriptase PCR test with the kits supplied by ICMR and the state Health Department. Results were analyzed thereafter and all data were collected.
Results
A total of 94,443 samples were tested in the COVID laboratory of Nil Ratan Sircar Medical College & Hospital, Kolkata for a duration of seven months (June 2020 to December 2020). 8,455 samples were found to be inconclusive (Table 1).
All inconclusive samples were subjected to re-testing (from repeat samples) after 3 days. Out of which, 1287 (15.22%) became positive and 3858 (45.63%) became negative [Table 2]. 1770 (20.94%) samples were found to be inconclusive again. In 1540 (18.21%) cases, they didn’t send their samples again to our Laboratory for re-testing (lost to follow up).
Ct value of these inconclusive results was analyzed and found that among positive samples, 64.96% had Ct value <34, whereas, among negative samples, 72.37% had Ct value >36 [Figure 1].
When all the cases were distributed according to the duration of disease, among positive cases, maximum patients (28.36%) had symptoms of less than 2 days’ duration, whereas, among negative cases, 36.86% cases had symptoms of more than 12 days’ duration (Figure 2).
Figure 2: Showing the distribution of cases according to the duration of disease (n= 94,443)
Discussion
During COVID 19 pandemic in India, there had been a spurt in ‘inconclusive’ reports that are leaving patients and clinicians baffled and postponing treatment. There may be various reasons for being only “one target gene” positive starting from sampling error to a technical error in consideration of the dynamics of target genes like N and ORF1ab gene.
We found that 8.95% of COVID RT PCR reports became ‘inconclusive’ in our study. Although It has been noted that up to 5% of COVID RT-PCR reports may be inconclusive [8]. It may be due to low viral load, faulty sample collection and transport and technical issues related to RNA extraction.8 When all inconclusive samples were subjected to re-collection, RNA extraction and re-testing by RT PCR after 3 days, 15.22% cases became positive whereas 45.63% cases turned negative. When these inconclusive reports were analyzed according to Ct value, 64.96% positive cases after retesting had Ct value <34 whereas, 72.37% negative cases had Ct value >36. Again, after comparing these cases with the duration of disease, it was found that among positive cases, maximum patients (28.36%) had symptoms of less than 2 days’ duration, whereas, 36.86% cases had symptoms of more than 12 days’ duration among negative cases.
This can be explained in this way that during the pre-symptomatic phase of infection, when the virus started to replicate and the disease curve started to ascend, the viral load is too low to detect or may detect one target gene, which when tested later after 3 days turned to be positive. Being asymptomatic or pre-symptomatic cases of less than 2 days’ duration, most of the inconclusive reports with Ct value <34, became positive on re-testing. Thus, a lower Ct value (<34 of one detected target gene) in inconclusive results increases the probability of being positive while re-testing after 3 days. This theory may also be implied for the negative results. At the later stage of disease, especially after 10-14 days, when the disease curve is about to touch baseline, one target gene may be detected again due to low viral load or presence of remnants of RNA or degraded RNA which when tested after 3 days become negative. Higher Ct value (>36) here indicating the increased probability of being negative on re-testing.
Out of total inconclusive samples, 1770 (20.94%) samples became inconclusive again after re-testing. When these cases were analyzed according to the duration of disease, most of the patients (24.41%) were asymptomatic or found of having symptoms of <2 days (20.56%), followed by 22.20% of cases with symptoms within >12 days’ duration. Most of the cases (46.55%) having Ct value within 34-36. This may happen due to various reasons such as – (a) beta coronavirus infections which are not due to SARS-COV2, (b) different analytical sensitivity of individual viral gene PCR, especially at low viral load and (c) nonspecific binding of PCR primer or probe during the late phase of PCR cycles (e.g. after 35 cycles).8 It may be due to problems associated with RNA extraction or due to faulty sample collection, storage and transport procedure.8 It may also because descension of the curve towards baseline is delayed due to aberrant or low immunological activity of patients and it persists for a long time beyond 12 days in samples at low concentration.
We lost for follow up in 1540 (18.21%) cases, as repeat samples were not sent again for further testing. Probable reasons behind it may be that these cases improved symptomatically or may be expired or samples were sent to another laboratory for re-testing due to changing testing strategy of the State Health Department.
Conclusion
An inconclusive result means the patient may be in the recovery stage or very early (pre-symptomatic) stage of infection. Inconclusive results most probably reflect low levels of virus in the sample but may also be due to sampling error, presence of remnants of RNA or degraded RNA or cross-contamination. In these cases, a second swab should be collected 3 days after the first test and re-tested. If the second test is negative, the patient will be considered as negative for COVID. If the second test is positive, the patient will be considered positive for COVID but If the second test remains inconclusive, interpretation is challenging. This patient should be evaluated with clinical symptoms and CT images and advised accordingly to continue home/hospital isolation. Ct value and duration of the disease of these cases may be a clue whether it is going to be positive or not on re-testing. It is also important that the clinician and various stakeholders (infection control experts) are explained about these possibilities. The counselling of the patient and relatives is also important. 8 Sample collection, RNA extraction or testing should always be done meticulously. Quality control and quality assurance of the whole testing procedure including the pre-and post-analytical parts are also essential.
Acknowledgement: I am sincerely giving thanks to the Head of our Institute for his support and kind cooperation. The authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors/editors/publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
Conflict of interest: Nil
Source of funding: Nil
References:
-
Zhu N, Zhang D, Wang W, Li X, Yang B, Song J. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020; 382:727-33.
-
World Health Organization. WHO Director-General’s opening remarks at the media briefing on COVID-19. Available from: https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-oncovid-19---11-march-2020. Accessed on June 1, 2020.
-
Chatterjee P, Anand T, Singh JK, Rasail R, Singh R, Das S, et al. Healthcare workers & SARS-CoV-2 infection in India: A case-control investigation in the time of COVID-19. Indian J Med Res. 2020;151:459-467.
-
Andrews MA, Areekal B, Rajesh KR, Krishnan J, Suryakala R, Krishnan B, et al. First confirmed case of COVID-19 infection in India: A case report. Indian J Med Res 2020;151:490-2.
-
Murhekar MV, Bhatnagar T, Selvaraju S, Rade K, Saravanakumar V, Vivian W, et al. Prevalence of SARS-CoV-2 infection in India: Findings from the national sero-survey, May-June 2020. Ind J Med Res. 2020;152:48-60.
-
Sethuraman N, Jeremiah SS, Ryo A. Interpreting Diagnostic Tests for SARS-CoV-2. JAMA. 2020:323(22):2249–2251.
-
TaqPath™ COVID-19 Combo Kit and TaqPath™ COVID-19 Combo Kit Advanced Instructions for Use. Interpretation of the results; Analysis and results, Chapter 11: 105-106
-
Bhattacharya S, Vidyadharan A, Joy VM. Inconclusive SARS-COV2 reverse transcription-polymerase chain reaction test reports: Interpretation, clinical and infection control implications. J Acad Clin Microbiol. 2020:22:59-61.
-
Indian Council of Medical Research. Strategy for COVID19 testing in India (Version 4). New Delhi: ICMR; 9 April, 2020.Available from: https://www.icmr.gov.in/ pdf/covid/strategy/ Strategey_for_COVID19_Test_v4_09042020.pdf. Accessed April 30, 2020.
-
Indian Council of Medical Research. Specimen referral form for COVID-19 (SARS-CoV2). New Delhi: ICMR; 2020.Available from: https://www.icmr.gov.in/ pdf/covid/update/SRF_v9.pdf. Accessed April 30, 2020.
-
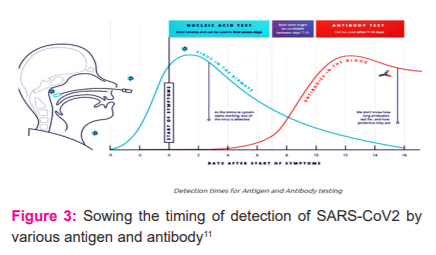
COVID 19 antigen testing. RAMS testing 2021. Available from: https://ramstesting.co.uk/covid-19-antigen-testing/
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License