IJCRR - 13(11), June, 2021
Pages: 27-34
Date of Publication: 04-Jun-2021
Print Article
Download XML Download PDF
Highlights on Synthetic, Natural, and Hybrid Cholinesterase Inhibitors for Effective Treatment of Alzheimer's Disease: A Review
Author: Kishor Danao, Yogesh Kodape, Debarshi Mahapatra, Sachin Borikar, Nilesh Karande
Category: Healthcare
Abstract:Alzheimer's disease (AD) is a progressive age-related neurodegenerative disorder and is the most common form of dementia among the elderly (65 years). The main pathological hallmarks of AD are the accumulation of amyloid plaques, or senile plaques, containing extracellular deposits of amyloid-\? peptide (A\?) and the presence of intraneuronal neurofibrillary tangles (NFTs), which result from hyperphosphorylated tau-protein. While preparing this review article, a huge collection of published literature from diverse pharmaceutical databases, life science databases, and medical databases such as PubMed, ScienceDirect, Google Scholar, etc. were explored and the literature was classified duly. The article focuses on various cholinesterase inhibitors obtained from diverse sources or categories such as established synthetic inhibitors (Physostigmine, Tacrine, Donepezil, Rivastigmine, Galantamine, and Metrifonate), novel synthetic inhibitors (Phenserine, Tolserine, Tesofensine, and Esolerine), natural inhibitors (Huperzine A, Neferine, Galangin, and Cardanol), synthetic hybrid derivatives (Donepezil and AP2238; Donepezil-Tacrine Hybrid; Tacrine-Ferulic acid Hybrid and Beta Carboline Derivatives; and Tacrine-8-hydroxyquinoline Hybrid), and new experimental synthetic analogues (Phenyl-5,6-dimethoxy-1-oxo-2,3-dihydro-1H-2-indenylmethanone and N-alkyl-7-methoxymatrine). This interesting piece of literature may serve as a readymade reference text material (particularly molecular-weight inhibitors) to the enthusiastic researchers (pharmacologists and medicinal chemists) working delicately in the area of AD. This study will positively open novel opportunities for research and futuristic pharmacotherapeutic application perceptive.
Keywords: Alzheimer’s, Natural, Hybrid, Synthetic, Inhibitor, Cholinesterase’s
Full Text:
INTRODUCTION
Alzheimer's disease (AD) is a progressive age-related neurodegenerative disorder and is the most common form of dementia among the elderly.1 It is generally diagnosed in individuals over the age of 65 years.2 The main pathological hallmarks of AD are the accumulation of amyloid plaques, or senile plaques, containing extracellular deposits of the amyloid-β peptide (Aβ) and the presence of intraneuronal neurofibrillary tangles (NFTs), which result from hyperphosphorylated tau-protein.3 Secondary, pathological hallmark of AD is the oxidation of lipids, proteins, and nucleic acids in neurons.4 The aetiology of AD is still unknown, but several factors have been suggested that appear to reduce the incidence of the disease.5
Three main approaches have been taken:
-
The first involves the reestablishment of neurotransmitters levels, with the inhibition of cholinesterases such as acetylcholinesterase (AChE), butyrylcholinesterase (BChE), and monoamine oxidase (MAO) enzymes.6
-
The second one concerns neuroprotection where oxidative stress is considered to be an early event in the pathological cascade for the disease, suggesting the potential use of antioxidants to limit the effects of free radicals on nerve cells.7
-
The third approach deals with specific aspects related to AD, including the decrease in the production or aggregation of Aβ peptide, and inhibition of γ and β-secretase enzymes which play a critical role in the amyloidogenic and tau protein pathways, among others. Intracellular NFT is made up of paired microtubule-associated protein tau helical filaments, which are abnormally hyperphosphorylated.8 The emphasis on studies that the protein deposition viz. oligomeric Aβ and P-tau.9 Researcher is eagerly viewing the contribution of Aβ and P-tau in AD. Also, they studied the expression of other essential brain proteins, and eventually how they cause neurodegeneration.10 However, to understand the mechanism of neurotoxicity and to ensure successful treatment, it is also important to discern how both Aβ and P-tau communicate.11 Various types of neurons deteriorate through the development of AD, and there is a deep depletion of forebrain cholinergic neurons, which is followed by a gradual decrease in acetylcholine.12 AChE is regulated by both the acetylcholine-synthesizing enzyme choline acetyltransferase (ChAT) and the acetylcholine-hydrolyzing enzyme AChE.13 To a large degree, therapies intended to reverse the cholinergic deficit are focused on the relevance of cholinergic activity in cognition. Despite the general reduction in AChE activity in the AD brain, current AD treatment is primarily based on AChE antagonists, which improve cholinergic transmission but have limited and intermittent therapeutic effects.14 For almost 50 years, it has been well known that the distribution of AChE molecular types in the AD brain is especially disturbed, but the physiopathological importance and consequent effects of these fascinating changes in AChE organisms remain unclear.15 A typical characteristic of AD neuropathology is an improvement in AChE levels around amyloid plaques and NFT, but the importance of this rise remains to be determined.16,17
ADNI's main successes were as follows:18-20
-
Production in a multicenter environment of systematic techniques for clinical studies, positron emission tomography (PET), magnetic resonance imaging (MRI), and cerebrospinal fluid (CSF) biomarkers;
-
Elucidation of trends, imaging transition frequencies, and biomarker measures of CSF in control subjects, patients with MCI, and patients with AD. CSF biomarkers are compatible with β-amyloid cascade and tau-mediated neurodegeneration theories for AD expected disease trajectories, while brain atrophy and hypometabolism levels show predicted trends, but show varying rates of change depending on area and seriousness of disease;
-
The evaluation of alternate medical categorization approaches. The best classifiers currently integrate optimal features from several modalities, including MRI, fluorodeoxyglucose-PET, CSF biomarkers and clinical trials;
-
In mildly symptomatic or even non-symptomatic subjects, β-amyloid, CSF biomarkers, and tau, as well as amyloid PET, can represent the earliest steps in AD pathology and are leading candidates for the detection of AD in its preclinical stages;
-
Improving the quality of clinical trials by recognizing the samples who are more likely to suffer imminent potential clinical deterioration by using more responsive outcome tests to minimize sample sizes. Baseline cognitive and/or MRI tests typically expected a better potential regression than other modalities, whereas the most successful outcome measures were found to be MRI measures of improvement;
-
Confirmation of the CR1, CLU, and PICALM AD risk loci and recognition of new nominee risk loci;
-
Worldwide influence in Europe, Asia, and Australia through the development of ADNI-like programmes;
-
Understanding the biology and pathobiology of normal ageing, MCI, and AD by combining ADNI biomarker data with ADNI clinical data to promote studies to address disputes regarding contradictory theories about AD etiopathogenesis, thus advancing efforts to identify disease-modifying ADNI drugs;
-
The construction of networks to allow all raw and processed data to be exchanged without embargo by interested science investigators worldwide. The ADNI research was expanded by a two-year 2009 Grand Opportunities grant and a renewal of the ADNI (ADNI-2) from October 2010 to 2016, with an additional 550 participants participating in the study.
PATHOPHYSIOLOGY
Alzheimer’s disease occurs due to the formation of senile plaque and neurofibrillary tangle in the brain (mostly hippocampus and cortex region), where the senile plaque is formed at the surface of the neuron and NFT is created inside the neuron.21 A senile plaque is formed by β-amyloid protein and NFT is formed by tau protein due to hyperphosphorylation. Amyloid precursor proteins are present on the surface of the neuron and are normally cut by the enzymes such as β-secretase and γ-secretase enzyme and this cut part is referred to as β-amyloid.22 This is collected in forming a fibre-like structure called senile plaque. NFT is formed and the signal is transferred from soma to the synapse, further, the signal is transferred from a neuron which is made up of microtubules.23 These microtubules are stabilized by tau protein. In Alzheimer’s disease, this tau protein becomes defective and removes from microtubules.24 The defective tau protein is assembling in neurons and formed filament. An abnormal accumulation of tau filament in neuron form NFT. Senile plaque and NFT are deposited at the synaptic junction due to a decrease the released and concentration of acetylcholine.25
ESTABLISHED SYNTHETIC CHOLINESTERASE INHIBITORS
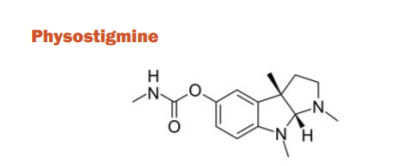
Physostigmine is a carbamate ester and an indole alkaloid that was the first ChE inhibitor investigated for the treatment of AD. It was isolated from the seeds of Physostigma venenosum, a para-sympathomimetic plant alkaloid. The blood-brain barrier (BBB) can move through, has a short half-life, and a small therapeutic index. It also has several side effects such as nausea, vomiting, diarrhoea, dizziness, and headaches. It was previously used for glaucoma treatment and delayed gastric emptying. However, because of the above-listed drawbacks, the drug was not licenced and was discarded for AD use. With a lower side effect profile, the newer medications proved to be more successful.26
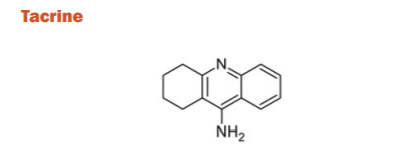
Tacrine is a 1,2,3-triazole derivative that acts both as AchE and BuchE. It was first approved for the treatment of AD in 1993 for its efficacy on the ADAS-Cog and the global measure compared to placebo in phase-II and phase-III clinical trials. For AD subjects, it was found to be quite limited owing to poor tolerance and precipitation of a number of side effects including vomiting, nausea, diarrhea, dizziness, syncope, and seizures. Also, the route of administration and patient compliance were challenging due to 4-times a day dosing regimen as a result of a short half-life. In addition, patients under treatment with this drug are required to have periodic blood monitoring due to hepatotoxicity. Finally, believed to be caused by the preference for BuChE and the availability of less toxic, better-tolerated medications with a simplified dosing schedule, tacrine was discontinued.27
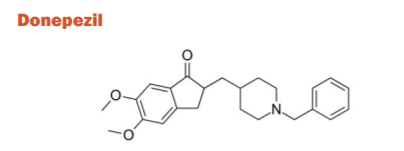
Donepezil is a piperidine derivative that acts as a selective and reversible inhibitor of AchE. Donepezil was approved in 1996 for the treatment of mild-to-moderate AD. A high dose (23mg) formulation was approved for use for moderate to severe AD. It is well absorbed with a relative oral bioavailability of 100% and reaches peak plasma concentrations in 3-4 hrs. The elimination half-life is ~70 hrs and is approximately 96% bound to human plasma proteins. It is primarily metabolized by CYP450 isoenzymes 2D6 and 3A4 and undergoes glucuronidation. Donepezil is known to function not just at the level of the neurotransmitter, but even at the molecular and cellular level in virtually any stage involved in AD pathogenesis.28
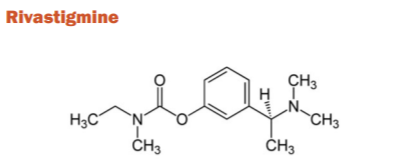
It is a carbamate derivative of reversible cholinesterase inhibitor that is selective for the central nervous system and is used for the treatment of AD. It is a small molecule and has easy BBB permeability which enables fantastic BuChE and AChE inhibitory properties. Rivastigmine was approved for the treatment of mild-to-moderate AD, in the year 2000 and has since gained approval for PAD after a trial with 699 patients. In capsular form, the drug has been assorting with side effects like nausea, vomiting, anorexia, and diarrhoea. In 2007, Rivastigmine was reformulated for delivery through a transdermal patch which resulted in significantly lowered GI side effects compared to the oral capsule. It is minimally metabolized by the CYP450 cytochrome system with weak binding to plasma proteins (~40%). The duration of ChE activity in cerebrospinal fluid is ~10 hrs after a 6-mg oral dose.29
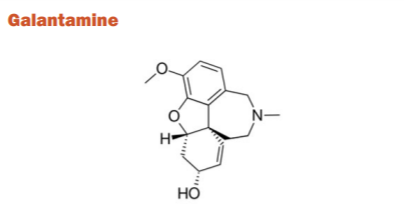
It is a benzazepine derivative obtained from norbelladine. It is found in Galanthus and Other Amaryllidaceae. Galantamine has been medicinal activity due to its sensitising action on nAChRs rather than general cholinergic stimulation due to cholinesterase inhibition. For the treatment of mild-to-moderate AD, it was accepted. A substantial increase in the ADAS-cog score of 3.3 points for the 16 mg/day group and 3.6 points for the 24 mg/day group was correlated with the administration of this medication, as well as a noticeable improvement in the physiological, emotional and functional symptoms of AD compared with placebo.30
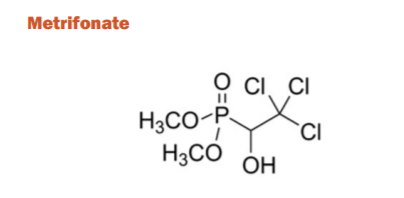
Metrifonate was originally developed for treating schistosomiasis but later found to be a long-acting irreversible ChE inhibitor. Chemically, it is dimethyl phosphonate (phosphonic ester) where the hydrogen atom attached to the phosphorous is substituted by a 2,2,2-trichloro-1-hydroxyethyl group. Although there was a low risk of side effects with short-term use, long-term uses caused respiratory paralysis and neuromuscular transmission dysfunction similar to a myasthenic crisis. As such, the FDA submission was halted and the clinical trials were discontinued during phase-III. However, show a robust therapeutic impact on ADAS-cog and other tests. However, before the further study of the relationship between metrifonate and neuromuscular disease is conducted, metrifonate is currently not an alternative for treatment with AD. Metrifonate is not an authorized for AD medication but demonstrates effectiveness that has been outweighed by safety hazards.31
NOVEL SYNTHETIC CHOLINESTERASE INHIBITORS
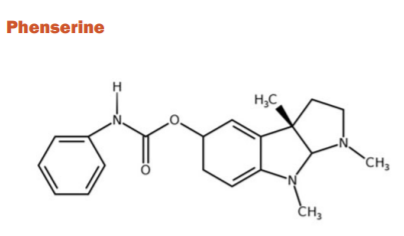
Phenserine is the phenyl carbamate derivation of physostigmine that has a dual effect due to the addition of methyl group on phenyl carbamate moiety by decreasing β-amyloid precursor protein and has a reversible AChE inhibition. It is less toxic than physostigmine and tacrine and has a rapid absorption rate. In elderly rats and dogs, increased learning and memory have also been demonstrated. Another, research on a dog found that phenserine increased learning and memory relative to dogs consuming the placebo group. During 2003-2004, 384 AD patients were enrolled at 10 mg and 15 mg twice daily doses in phase III clinical trial. Phenserine, however, did not substantially boost ADAS-cog scores relative to placebo.32
Tolserine
Tolserine only differs from Phenserine at the 2-methyl substitution on its phenyl carbamoyl moiety. The pre-clinical studies were initiated in the year 2000 and it showed 200-fold more selectivity against AChE vs. BuChE. Tolserine proved to be a highly potent inhibitor of human AChE compared to its structural analogues physostigmine and phenserine.33
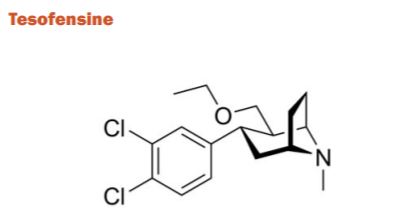
Tesofensine belongs to the phenyl tropane category. It inhibits the presynaptic uptake of serotonin, norepinephrine and dopamine neurotransmitters in both in vitro and in vivo studies. The compound increases the function of the neurotransmitters acetylcholine, noradrenaline, and dopamine, both of which are impaired in patients with AD. A decrease β-amyloid concentration was found in mice after the use of tesofensine which was thought to be a neuroprotective effect. Phase-IIA trials showed significant cognitive improvement in those with mild AD. However, Phase-IIB trials showed limited activity. Currently, tesofensine is being marketed as a drug against obesity.34
Esolerine
Esolerine, a cyclic alkyl carbamate and a tacrine derivative which has been identified as a very strong AChE inhibitor and significantly favoured AChE over BuChE. But, there are no published reports in humans, so it is difficult to identify potential risks and benefits in pre-clinical and clinical models.35
NATURAL DERIVATIVES
Huperzine-A (HupA) is a lycopodium alkaloid used for memory deficiency isolated from the Chinese medicinal herb Huperzia serrata. The plant is widely grown in China and several other regions of Asia. The famous Chinese Medicine tradition emphasizes this plant and its products in various herbal remedies owing to high selectivity, reversibility, and potent AChE inhibition action. It is often considered to be the drug of choice in China for the treatment of memory disorders. However, it has been noticed that the synthetic racemic mixture of HupA has less AChE inhibitory effect than the HupA of natural origin. For enhancing the potency and efficacy, Tacrine and donepezil have also been hybridised with HupA. Since the donepezil hybrid was less successful, the HupA-tacrine hybrids were called huprines (huprine Y and huprine X) have shown to be more effective in inhibiting the enzyme AChE in vitro than tacrine. Huprine Y and Huprine Z have shown better inhibitory activity than either of the parent drugs as well HupA with a higher oral bioavailability as compared to tacrine and donepezil. The improvement is more noticeable on working memory than reference memory as compared to donepezil and tacrine. In terms of inhibition functions, HupA is also found to be more active than tacrine, rivastigmine, and galantamine, although it has the least amount of BuChE activity.36
Nelumbo nucifera is an aquatic plant with numerous medicinal properties. It has been recently observed that the stamens of this plant drastically improved the maze learning tasks in rats when fed for a specific duration. Few compounds have been identified as neferine, 5-O-methyladenosine, cycloartenol, vanilloloside, a β-cyclogeraniol diglycoside, p-hydroxybenzoic acid, etc. which demonstrated excellent non-competitive inhibition of AChE and BuChE which opened new avenues for treating AD.37
Galangin
Himatanthus lancifolius is a shrub that contains several indole alkaloids having several medicinal properties such as antimicrobial effects, gastroprotection, and the ability to affect the vascular and non-vascular smooth muscle responsiveness. The AChE inhibiting properties of ethyl acetate and dichloromethane extracts (containing 21 different flavonoids) have been in vitro studied where significant inhibition of the target was observed. Because of their free radical scavenging properties, flavonoids have been of considerable importance in AD science and care. Epidemiological data shows that higher flavonoid intake is associated with lower AD incidence. Galangin, a flavonol has demonstrated the highest inhibitory effects on AChE activity. However, it is unknown whether galangin binds to the AChE directly or has the same binding site as the ATCh substrate38.
The cashew nut-shell liquid (CNSL) containing primary non-isoprenoid phenolic lipids (NIPL) components such as cardols, cardanols, anacardic acids, and methylcardols have been recognized as potential templates for the development of AChE inhibitors. The produced derivatives had electrical, hydrophobic, and structural properties that are required to act potentially as AChE molecules. The promising candidates for AD treatment have been identified from cardanols because of the abundance of the raw material source. Although, there are no human studies reported to date.39
SYNTHETIC HYBRID DERIVATIVES
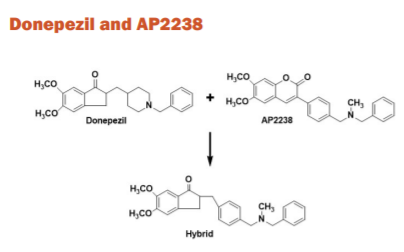
AP2238 is the first published compound to bind with both the anionic sites of AChE. The inhibitory potency against AChE is comparable to donepezil, while its ability to contrast β-amyloid aggregation is higher. A sequence of donepezil and AP2238 hybrids was formed in which the donepezil idanone centre is bound to the AP2238 phenyl-N-methylbenzylamino moiety. A derivative in which the AP2238 phenyl-N-methylbenzylamino moiety was substituted by the AP2243 phenyl-N-methylbenzylamino moiety and a tetralone scaffold was replaced by the idanone ring. The hybrids showed improved inhibition of AChE-induced aggregation vastly when compared to the reference compounds.40
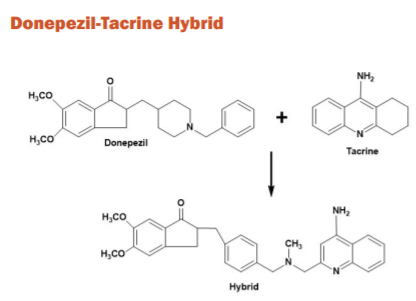
Researchers engineered a new sequence of donepezil-tacrine hybrids, interacting concurrently at the periphery, active, and mid gorge binding sites of AChE. They are appealing because both compounds have known effectiveness and BBB permeability with rather distinct MOAs. They were tested for the ability to inhibit BuChE, AChE, and β-amyloid accumulation caused by AChE. The compounds are a mixture of donepezil and 6-chlorotacrine 5,6-dimethoxy-2-[(4-piperidinyl)methyl]-1-idanone molecules. All of the recent hybrids have proved to be incredibly potent inhibitors of hAChE. Both of the new compounds also displayed strong β amyloid aggregation inhibition and were found to be more active than the parent molecules.41
Tacrine-Ferulic acid Hybrid and Beta Carboline Derivatives
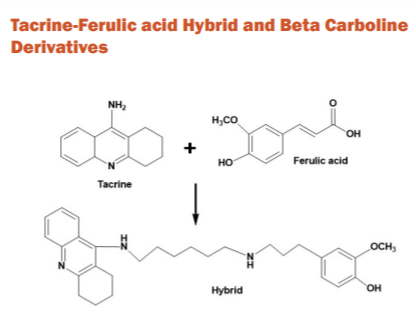
There were 3 β-carboline derivatives and 2 tacrine/ferulic acid combinations tested in vivo in 3-month-old female rats. The tacrine/ferulic acid hybrids exhibited stronger AChE inhibition and similar BuChE inhibition as compared with tacrine in vitro. However, neither compounds showed any therapeutic effects against scopolamine-induced cognitive deficits in vivo. Despite the promising in vitro information, none of the compounds is currently suitable for AChE inhibition against AD.42
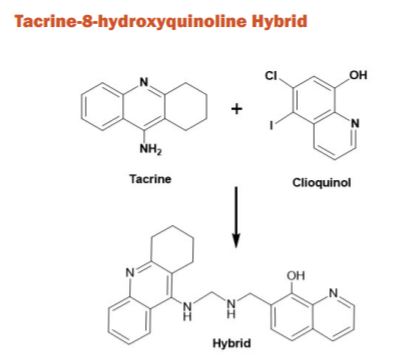
Both tacrine and tacrine 8-hydroxyquinoline derivative are known for ChE inhibition and decreasing β-amyloid concentrations, respectively. Recently, few novel tacrine-8-hdroxyquinoline hybrids have been synthesized and evaluated as possible therapeutic drugs for AD treatment where the hybrids were found to be more effective than tacrine against AChE and BuChE with low cell level toxicity and better BBB penetration.43
NEW EXPERIMENTAL SYNTHETIC ANALOGS
Phenyl-5,6-dimethoxy-1-oxo-2,3-dihydro-1H-2-indenylmethanone
This series of synthetic analogues are currently under development because of targeted pharmacological development, class-specific hepatotoxicity, and known gastrointestinal side effects which may be avoided. The possibility of producing synthetic analogues is that as naturally derived ChEIs they will have the strength and BBB permeability or they may have unanticipated pharmacological properties. This phenyl-5,6-dimethoxy-1-oxo-2,3-dihydro1H-2-indenylmethanone series showed that the majority of the compounds had moderate AChE inhibitory activity.44
N-Alkyl-7-methoxytacrine
Tetrahydro amino acridine (THA) was the first AChEI developed, but dose-dependent but reversible liver toxicity was caused by using the drug. 7-methoxytacrine (7-MEOTA) is a much less toxic THA analogue that is pharmacologically similar to THA. The synthesized 7-MEOTA analogues presented in vitro human recombinant AChE (hAChE) and human plasmatic BuChE (hBuChE) enzyme inhibition which was compared to both THA and 7-MEOTA. The structure-activity relationship findings point at the C6-C7 N-alkyl chains for ChE inhibition.45
CONCLUSION
This vital review content largely underlined the synopsis of various causes of Alzheimer’s disease including the pathogenesis. The article focuses on various cholinesterase inhibitors obtained from diverse sources or categories such as established synthetic inhibitors (Physostigmine, Tacrine, Donepezil, Rivastigmine, Galantamine, and Metrifonate), novel synthetic inhibitors (Phenserine, Tolserine, Tesofensine, and Esolerine), natural inhibitors (Huperzine A, Neferine, Galangin, and Cardanol), synthetic hybrid derivatives (Donepezil and AP2238; Donepezil-Tacrine Hybrid; Tacrine-Ferulic acid Hybrid and Beta Carboline Derivatives; and Tacrine-8-hydroxyquinoline Hybrid), and new experimental synthetic analogues (Phenyl-5,6-dimethoxy-1-oxo-2,3-dihydro-1H-2-indenylmethanone and N-alkyl-7-methoxytacrine). This interesting piece of literature may serve as a readymade reference text material (particularly the low molecular-weight inhibitors) to the enthusiastic researchers (pharmacologists and medicinal chemists) working in the area of Alzheimer’s disease. This study will positively open novel opportunities for research and futuristic pharmacotherapeutic application perceptive.
ACKNOWLEDGMENT
The authors acknowledge the kind support of the college management for providing internet facilities for the literature review.
CONFLICT OF INTEREST
The authors stated No Conflict of Interest for the Publication of this review article in the Journal.
FUNDING INFORMATION
None acknowledged.
AUTHOR CONTRIBUTION
KRD: Supervise the literature survery and drafting and editing of the manuscript
YK: Complete literature survey performed
DKM: Made Chemical Structures, Wrote Structured Abstract, Drawn Graphical Abstract, Set References, Grammatical corrections
SPB: Removed plagiarized contents and attended manuscript revisions
UNM: Final reviewing of this manuscript and provided suggestions
References:
1. Finch CE, Cohen DM. Aging, metabolism, and Alzheimer disease: review and hypotheses. Exp Neurol. 1997;143(1):82-102.
2. Cutler NR, Sramek JJ. Review of the next generation of Alzheimer's disease therapeutics: challenges for drug development. Prog Neuropsychopharmacol Biol Psychiatry. 2001;25(1):27-57.
3. Moraros J. The association of antidepressant drug usage with cognitive impairment or dementia, including Alzheimer disease: A systematic review and meta?analysis. Depress Anxiety. 2017;34(3):217-26.
4. Cummings J. Drug development in Alzheimer’s disease: the path to 2025. Alzheimers Res Ther. 2016;8(1):39.
5. Sramek JJ, Zarotsky V, Cutler NR. Review of drug development and therapeutic role of cholinesterase inhibitors in Alzheimer's disease. Drug Dev Res. 2002;56(3):347-53.
6. Kumar A, Singh A. A review on Alzheimer's disease pathophysiology and its management: an update. Pharmacol Rep. 2015;67(2):195-203.
7. Rosenberg RN. Genomics of Alzheimer disease: a review. JAMA Neurol. 2016;73(7):867-74.
8. Jalbert JJ, Daiello LA, Lapane KL. Dementia of the Alzheimer type. Epidemiol Rev. 2008;30(1):15-34.
9. Christensen H, Griffiths K, MacKinnon A, Jacomb P. A quantitative review of cognitive deficits in depression and Alzheimer-type dementia. J Int Neuropsychol Soc 1997;3(6):631-51.
10. Cazarim MD., Perspectives for treating Alzheimer's disease: a review on promising pharmacological substances. Sao Paulo Med J. 2016;134(4):342-54.
11. Samanta MK, Wilson B, Santhi K, Kumar KS, Suresh B. Alzheimer disease and its management: a review. Am J Therapeut 2006;13(6):516-26.
12. El Kadmiri N. Biomarkers for Alzheimer disease: Classical and novel candidates’ review. Neuroscience 2018;370:181-90.
13. Folch J. Review of the advances in treatment for Alzheimer disease: strategies for combating β-amyloid protein. Neurología. 2018;33(1):47-58.
14. Whitehouse PJ. Neurotransmitter receptor alterations in Alzheimer disease: a review. Alzheimer Dis Assoc Disord. 1987;1(1):9-18.
15. Giacobini E. Invited Review Cholinesterase inhibitors for Alzheimer’s disease therapy: from tacrine to future applications. Neurochem Int. 1998;32(5-6):413-9.
16. Mangialasche F. Alzheimer's disease: clinical trials and drug development. Lancet Neurol. 2010;9(7):702-16.
17. Anand P, Singh B. A review on cholinesterase inhibitors for Alzheimer’s disease. Arch Pharm Res. 2013;36(4):375-99.
18. Bolognesi ML. Melchiorre C. Alzheimer's disease: new approaches to drug discovery. Curr Opin Chem Biol. 2009;13(3):303-8.
19. Kaduszkiewicz H, Cholinesterase inhibitors for patients with Alzheimer's disease: systematic review of randomised clinical trials. Br Med J. 2005;331(7512):321-7.
20. West S, Bhugra P. Emerging drug targets for Aβ and tau in Alzheimer's disease: a systematic review. Br J Clin Pharmacol. 2015;80(2):221-34.
21. Cummings J, Lee G, Ritter A, Zhong K. Alzheimer's disease drug development pipeline: 2018. Alz Dement Transl Res Clin Interv. 2018;4:195-214.
22. Cummings JL, Morstorf T, Zhong K. Alzheimer’s disease drug-development pipeline: few candidates, frequent failures. Alz Res Ther. 2014;6(4):1-7.
23. Ng YP, Or TC, Ip NY. Plant alkaloids as drug leads for Alzheimer's disease. Neurochem Int. 2015;89:260-70.
24. Darvesh S. Inhibition of human cholinesterases by drugs used to treat Alzheimer disease. Alzheimer Dis Assoc Disord. 2003;17(2):117-26.
25. Rosini M., Rational approach to discover multipotent anti-Alzheimer drugs. J Med Chem. 2005;48(2):360-3.
26. Batiha GE. Physostigmine: A plant alkaloid isolated from Physostigma venenosum: A review on pharmacokinetics, pharmacological and toxicological activities. J Drug Deliv Therapeut. 2020;10(1-s):187-90.
27. Sameem B, Saeedi M, Mahdavi M, Shafiee A. A review on tacrine-based scaffolds as multi-target drugs (MTDLs) for Alzheimer's disease. Eur J Med Chem. 2017;128:332-45.
28. Greig SL. Memantine ER., donepezil: a review in Alzheimer’s disease. CNS drugs. 2015;29(11):963-70.
29. Schneider LS. Systematic review of the efficacy of rivastigmine for patients with Alzheimer's disease. Int J Geriatr Psychopharmacol. 1998;1(1):S26-34.
30. Corey-Bloom J. Galantamine: a review of its use in Alzheimer's disease and vascular dementia. Int J Clin Pract. 2003;57(3):219-23.
31. Ringman JM, Cummings JL. Metrifonate: update on a new antidementia agent. J Clin Psychiatry. 1999;60(11):776-82.
32. Winblad B. Phenserine efficacy in Alzheimer's disease. J Alzheimers Dis. 2010;22(4):1201-8.
33. Kamal MA, Greig NH, Alhomida AS, Al-Jafari AA. Kinetics of human acetylcholinesterase inhibition by the novel experimental Alzheimer therapeutic agent, tolserine. Biochem Pharmacol. 2000;60(4):561-70.
34. Lehr T. Quantitative pharmacology approach in Alzheimer’s disease: efficacy modeling of early clinical data to predict clinical outcome of tesofensine. AAPS J. 2010;12(2):117-29.
35. Habtemariam S. Natural products in Alzheimer’s disease therapy: would old therapeutic approaches fix the broken promise of modern medicines? Molecules. 2019;24(8):1519.
36. Bai D. Development of huperzine A and B for treatment of Alzheimer's disease. Pure Appl Chem. 2007;79(4):469-79.
37. Jung HA. Selective cholinesterase inhibitory activities of a new monoterpene diglycoside and other constituents from Nelumbo nucifera stamens. Biol Pharm Bull. 2010;33(2):267-72.
38. Seidl C. Acetylcholinesterase inhibitory activity of uleine from Himatanthus lancifolius. Zeitschrift für Naturforschung C. 2010;65(7-8):440-4.
39. Stasiuk M. Inhibitory effect of some natural and semisynthetic phenolic lipids upon acetylcholinesterase activity. Food Chem. 2008;108(3):996-1001.
40. Rizzo S, Bartolini M, Ceccarini L, Piazzi L, Gobbi S, Cavalli A, et al. Targeting Alzheimer’s disease: Novel indanone hybrids bearing a pharmacophoric fragment of AP2238. Bioorg Med Chem. 2010;18(5):1749-60.
41. Alonso D, Medina M. Donepezil–tacrine hybrid related derivatives as new dual binding site inhibitors of AChE. Bioorg Med Chem. 2005;13(24):6588-97.
42. Fleck C. Investigation into the in vivo effects of five novel tacrine/ferulic acid and β-carboline derivatives on scopolamine-induced cognitive impairment in rats using radial maze paradigm. Arzneimittelforschung. 2010;60(06):299-306.
43. Ferna?ndez-Bachiller MI. Novel tacrine− 8-hydroxyquinoline hybrids as multifunctional agents for the treatment of Alzheimer’s disease, with neuroprotective, cholinergic, antioxidant, and copper-complexing properties. J Med Chem. 2010;53(13):4927-37.
44. Ali MA. Design, synthesis and evaluation of novel 5, 6-dimethoxy-1-oxo-2, 3-dihydro-1H-2-indenyl-3, 4-substituted phenyl methanone analogues. Bioorg Med Chem Lett. 2009;19(17):5075-7.
45. Korabecny J. Synthesis and in vitro evaluation of N-alkyl-7-methoxytacrine hydrochlorides as potential cholinesterase inhibitors in Alzheimer disease. Bioorg Med Chem Lett. 2010;20(20):6093-5.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License