IJCRR - 13(9), May, 2021
Pages: 174-178
Date of Publication: 07-May-2021
Print Article
Download XML Download PDF
Evaluation and Comparison of Drug Advertisements Published in Medical Journals Using WHO Criteria for Ethical Medicinal Drug Promotion and OPPI Criteria for Drug Advertisements
Author: Dabhade SA, Dabhade SS
Category: Healthcare
Abstract:Background: Advertisements in medical journals is also an important and effective way that is used by pharmaceutical companies to promote their drugs and to reach clinicians. In drug advertisements, misleading and wrong information is seen and sometimes such literature is of poor scientific and educational utility. However, despite the availability of guidelines and self-regulatory codes worldwide, pharmaceutical advertisement in a medical journal are being of poor.
Objective: To evaluate drug promotion advertisement in Indian and Non-Indian medical journals.
Methods: 30 drug advertisements each published in Indian and non-Indian scientific medical journals were selected randomly Drug promotional advertisements that promote allopathy medicines. Each drug advertisements were evaluated for - Compliance with WHO ethical criteria for medicinal drug promotion. Each drug advertisement was also evaluated for - compliance with the OPPI code. The percentage was calculated for compliance to WHO criteria, OPPI code and claims made. An unpaired t-test was used. P< 0.05 was considered significant for comparison.
Results: Adjuvants were mentioned in 56.66% of Indian journals and 86.67% non-Indian journals advertisements. Side effects, warnings, contraindications precautions were significantly more mentioned in non-Indian journal advertisements.
Conclusion: Pharmaceutical companies are involved in establishing a commercial relationship with the treating physicians wherein the educational aspect of scientific information about drugs is compromised. Pharmaceutical companies did not meet all the WHO criteria and OPPI code for ethical drug promotion. Strict compliance to these criteria is necessary for rational drug therapy and the benefit of the patient. Hence critical appraisal of all forms of drug promotion including journal advertisement by the physicians is very crucial. This study recommends that code for drug promotion should be supported by government legislation and strict regulatory implementation.
Keywords: Compliance of drug advertisement, Medical journal, OPPI code, Pharmaceutical industries, Drug advertisements, WHO ethical criteria for medicinal drug promotion
Full Text:
Introduction
According to the World Health Organization (WHO), drug promotion is every informational and persuasive activity of the pharmaceutical companies, which can induce prescription, supply, purchase, and use of medicinal drugs.1 Pharmaceutical industries promote their drugs in many ways, such as direct email advertisements or drug information sheet by medical representatives. Gift articles and free drug samples, sponsorship of scientific and educational activities, seminars, conferences and tours are also provided by pharmaceutical industries. For the doctors, the literature promoting the drugs given by medical representative becomes an important source of information about drugs. It mainly targets physicians.2 Gahalaut et al. said that it may also modulate the prescribing behaviour of physicians without their knowledge.3 Advertisements in medical journals is also an important and effective way which is used by the pharmaceutical companies to promote their drugs and to reach clinicians. Journal advertisements attract physician’s attention because they have appealing graphs and pictures.4 Hence it is necessary that drug advertisements including advertisements in medical journals should provide scientific information. For a better understanding and utilization of drugs in patients, the doctors must observe the research findings and draw conclusions. In drug advertisements, misleading and wrong information is seen and sometimes such literature is of poor scientific and educational utility. This may lead to irrational prescribing. It may also lead to unnecessary adverse effects. Influencing physicians through promotional activities without necessarily benefitting the patients contribute to increased health care costs.5
For drug promotional advertisements, two guidelines are used, one of which is ‘ethical criteria for medicinal drug promotion by WHO. The main objective of ethical criteria for medicinal drug promotion is to support and encourage the improvement of health care through the rational use of medicinal drugs1. Another code is the code of pharmaceutical marketing practices by the International Federation of Pharmaceutical Manufacturers Association (IFPMA).6 In India drug promotional advertisements are not regulated by the governmental agency. In India, promotional activities by pharmaceutical companies are governed by the Organization of Pharmaceutical Producers of India (OPPI). It is a self-regulatory code of pharmaceutical marketing practices.7 OPPI has adopted a self-regulatory code of pharmaceutical marketing suggested by IFPMA.
However, despite the availability of guidelines and self-regulatory codes worldwide, pharmaceutical advertisement in a medical journal is being of poor quality.8In India few studies about the evaluation of drug promotional advertisements published in scientific medical journals have been reported. Drug promotional advertisements found to be not adherent to WHO criteria for medicinal drug promotion.9 Comparison of drug advertisements in medical journals by using WHO criteria and OPPI code of pharmaceutical marketing practices is lacking in India. Hence this study has planned to evaluate and compare the ethical standards of drug advertisements in scientific medical journals using WHO criteria and the OPPI code of pharmaceutical marketing practices. In the present study we aimed to evaluate drug promotion advertisement in Indian and Non-Indian medical journals. Our evaluated and compare the compliance of drug advertisements to WHO criteria for medicinal drug promotion in Indian and non-Indian medical journals, the compliance of drug advertisements to Organization of Pharmaceutical Producers of India (OPPI) code in Indian and non-Indian medical journals and the claims made in drug advertisements in Indian and non-Indian medical journals.
MATERIALS AND METHDOS
This is an observational and crosses?sectional study conducted in tertiary care hospital, Pune. Thirty drug advertisements each published in Indian and non-Indian scientific medical journals were selected randomly out of 50 literature each using a random number generator.
Inclusion criteria:
-
Drug promotional advertisements that promote allopathy medicines,
-
Drug promotional advertisements that contain at least one therapeutic claim was included for the analysis in this study.
Exclusion Criteria:
-
Those advertisements promoting medicinal equipment’s (e.g., insulin pump, etc.) and devices and orthopaedic prosthesis, product monographs,
-
Ayurvedic and Siddha medicines, reminder advertisements were all excluded from this study.
Each drug advertisements were evaluated for - Compliance to WHO criteria for medicinal drug promotion which includes 1) INN of the active substance (i.e., generic name) 2) Brand name and content (per dose) 3) Name of other ingredients 4) Approved use 5) Regimen 6) Side effects 7) Warnings 8) Contraindications 9) Precautions 10) Interactions 11) Name and address of manufacturer 12) references.
Each drug advertisement was also evaluated for - compliance to OPPI code which includes
-
Brand name.
-
The name and address of the drug industry.
-
The active ingredients, using approved generic names.
-
Date of production of the advertisement.
-
Approved indication or indications for use, together with the dosage and method of use; and a statement of the contraindications, precautions, and side effects.
One mark is given for the presence of criteria/claim in journal advertisement and zero marks is given for the absence of criteria/claim, the percentage was calculated for compliance to WHO criteria, OPPI code and claims made. An unpaired t-test was used. P< 0.05 was considered significant for comparison. The percentage of claims made in Indian and non-Indian medical journals were calculated.
Results
Table 1 and Figure 1 showed that 93.33% of Indian journals and 100% non-Indian journals had mentioned generic name in the advertisements. Both the journal gave the brand name in 100% advertisements. Adjuvants were mentioned in 56.66% of Indian journals and 86.67% non-Indian journals advertisements. Mentioning adjuvants were significantly more in non-Indian journals. Approved use of drug in of the advertised drug was mentioned in 90% Indian journal and 93.33% non-Indian journals advertisements respectively.
Regimens of the advertised drug were mentioned in 76.66% Indian journal and 93.33% non-Indian journals advertisements. Side effects of the advertised drug were mentioned in 86.67% Indian journal and 98% non-Indian journals advertisements. Warnings were mentioned in 46.67% of Indian journal and 80% non-Indian journal advertisements. Mentioning of warnings were significantly more in non-Indian journals. Contraindications were mentioned in 60% of Indian journal and 76.67% non-Indian journal advertisements. Precautions were mentioned in 66% of Indian journal and 90% non-Indian journal advertisements. Drug interactions were mentioned in 40% of Indian journal and 76.67% non-Indian journals advertisements. Mentioning of drug interactions were significantly more in non-Indian journals. The name and address of the manufacturer were mentioned in 90% of Indian journal and 93.33% non-Indian journals advertisements. References were mentioned in 53.33% of Indian journal and 83.33% non-Indian journals advertisements. The mentioning of references were significantly more in non-Indian journals.
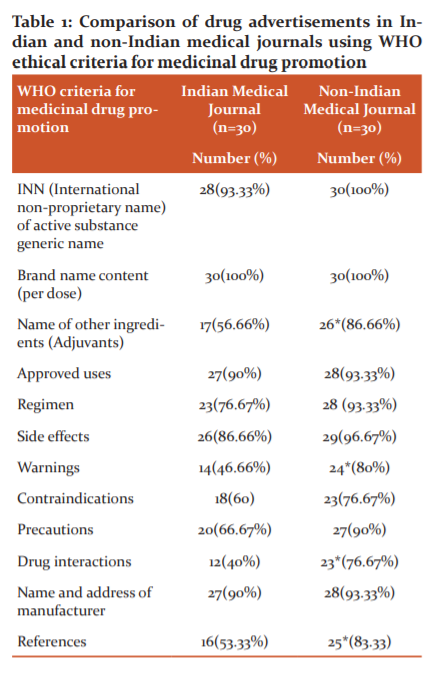
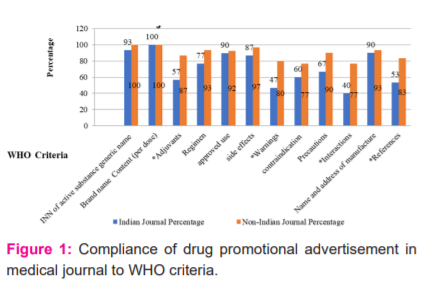
Table 2 and Figure 2 showed that 100% Indian and non-Indian journals were mentioned.
The name of the product (normally the brand name) in the advertisements 90% of Indian and 98%non-Indian journals were mentioned the name and address of the pharmaceutical company or its agent responsible for marketing the product. 93.33% Indian and 98%non-Indian journals were mentioned the active ingredients, using approved names where they exist. 30% Indian and 83.33%non-Indian journals were mentioned the date of production of the advertisement. date of production of the advertisement was significantly lower in Indian journals. Approved indication or indications for use together with the dosage and method of use; and a statement of the contraindications, precautions, and side-effects were mentioned by 73.33% Indian journal and 98% non-Indian journal advertisements. This was significantly more mentioned in non-Indian journal advertisements.
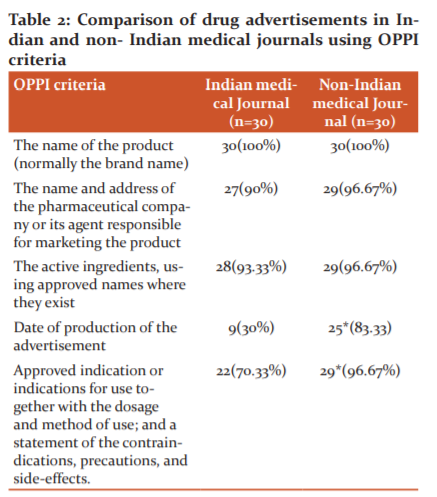
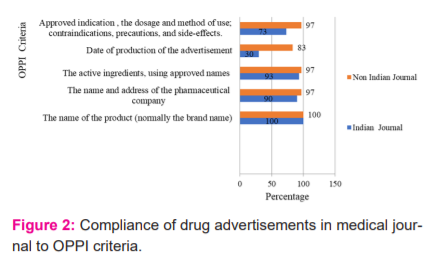
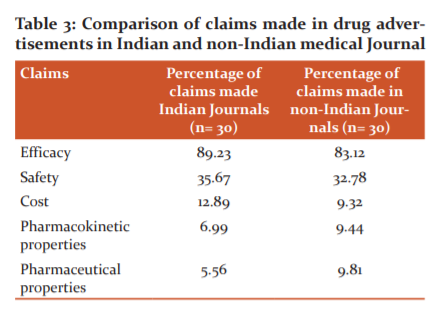
Table 3 showed the claims made in the journal advertisements. Efficacy was mentioned in 89.33% of Indian journal and 83.12% non-Indian journal advertisements. Safety was mentioned in 32.78% non-Indian journal and 35.67% of Indian journal advertisements. The cost was mentioned in 9.32% non-Indian journal and 12.89% of Indian journal advertisements. PK properties were mentioned in 9.44 % of non- Indian journal and 6.99% Indian journal advertisements. Pharmaceutical properties were mentioned in 9.81 % of non- Indian journal and 5.56% of Indian journal advertisements.
Discussion
Marketing d0rugs through drug advertisements is one of the strategies adopted by pharmaceutical companies. Pharmaceutical companies are involved in establishing a commercial relationship with the treating physicians wherein the educational aspect about scientific information about drugs is compromised.10 It has been observed in this study that pharmaceutical companies did not meet all the WHO criteria and OPPI code for ethical drug promotion. In this study, generic name, brand name and address of manufacturer were seen in >90% of drug advertisements in both Indian and non-Indian journals. Less than 60% of drug advertisements in Indian journals are compliant with WHO criteria like the name of adjuvant, warnings, interactions, references. Whereas more than 80% of drug advertisements in non-Indian journals were compliant with to above WHO criteria. These findings are statistically significant and are similar to Jadhav et al.11 and Ganashree et al.12
It has been observed that the majority of drug advertisements in Indian and non -Indian medical journals are compliant with dosage schedule and therapeutic indications. But drug advertisements in Indian medical journals are less compliant to mentioning adverse drug reactions, precautions, contraindications, and interactions. Strict compliance to these criteria is necessary for rational drug therapy and the benefit of the patient. Hence critical appraisal of all forms of drug promotion including journal advertisement by the physicians is very crucial. In this study, it is observed that Indian journals are significantly less compliant to OPPI criteria as compared to non-Indian journals in mentioning of approved indications, dosage, method of use, contraindications, precautions, and side-effects are considered. Indian medical journals are significantly less compliant in mentioning the date of production of that advertisement as required by the OPPI criteria.
Indian journals are significantly less compliant with the OPPI code, while Non-Indian journals are significantly more compliant with the IFPMA code.13 This study endorses this finding. It needs to be noted that the OPPI code is derived from the IFPMA code hence we can say that Indian journals are less compliant with the IFPMA code. This means that Indian journals are significantly less compliant to voluntary code proposed by associations of pharmaceutical industries (OPPI, IFPMA). In this study, claims made in the advertisements in medical journals were evaluated and compared. It is found that efficacy claims are present in 89.23% and 83.12% in Indian and non-Indian medical journals respectively. Safety claims are present in 30.67% and 32.78% in Indian and non-Indian medical journals respectively. Cost and convenience claims are more in non-Indian journals as compared to Indian journals, though it is not significant. These finding are similar to Mali et al.10 where efficacy claims were made in 92% of drug advertisements and safety claims in 37.8% of drug advertisements.
In this study, pharmacokinetic claims and pharmaceutical claims (e.g. sustained-release preparation, dispersible tablets etc) were seen in 5.44% and 5.61% of advertisements respectively in Indian and non-Indian journals. Mali et al. mention 16.4% claims on pharmacokinetic properties and 29.6%. claims on pharmaceutical properties in drug advertisements. Thus, from the above findings, it is seen that pharmaceutical companies highlight the positive aspects of their products while omitted the negative aspects.
Conclusion
Drug advertisements in medical journals are an important source of providing information about drugs to doctors. Drug advertisements in Indian journals are not adequately compliant with the WHO and OPPI code. For further improvement in the quality of drug advertisements in Indian journals, this study recommends that code for the drug promotion should be supported by government legislation and strict regulatory implementation. Medical practitioners and medical students need to be educated to critically evaluate the drug promotional activities and increase their ability to recognize misleading promotional advertisements. Doctors should be encouraged to report any violation of the ethical code of drug promotion to the regulatory authorities.
Acknowledgement: Authors would like to acknowledge librarian Dr.D.Y.Patil medical college, Pimpri, Pune for making the medical journals available.
Authors’ Contribution: The first author has given the idea of the topic and written the article. The second author has done the data collection and statistical part.
Conflict of Interest: None
Source of Funding: None
References:
-
Ethical Criteria for Medicinal Drug Promotion 1988. WHO Geneva Available from http://www.apps.who.int/medicinedocs/documents/whozip08e/pdf. [Accessed on 2020 Nov 30].
-
Mikhael EM. Evaluating the reliability and accuracy of the promotional brochures for the pharmaceutical companies in Iraq using WHO guidelines. J Pharma Bio Sci 2015;7:65-8
-
Gahalaut P, Chauhan S, Mishra N, Rastogi M, Thakur R. Drug advertisements in two Drmatology journals: A critical comparison of IJDVL and JAAD. Indian J Dermatol Venereol Leprol 2014;80(2):115.
-
Politics of Medicines: Analysing Pharmaceutical Advertisements in Medical Journals. http://www.politicsofmedicines.org/articles/analysing-pharmaceutical-advertisements-in-medical-journals. Accessed 2020, Nov. 15.
-
Saxena JD, Yadav P. Drug promotional literature distributed by pharmaceutical companies: Do they provide enough information to ascertain their validity? J Pharmacol Pharmacother 2011;2(3):192–194.
-
IFPMA code of practice – IFPMA. Available from: ifpma.org/resource-centre/ifpma-code-of-practice/. [Accessed 2020, June12].
-
OPPI Code of Pharmaceutical Practices. The organisation of Pharmaceutical Producers of India 2012. Mumbai: 2012. Available from: http://www.ifpma.org/fileadmin/content/About%20us/2%20Members/Associations/Code India/OPPI_Code_of_Pharmaceutical_Practices__-_2012.pdf.
-
Oshikoya KA, Senbanjo IO, Soipe A. Adequacy of pharmacological information provided in pharmaceutical drug advertisements in African medical journals. Pharm Practi 2009;7(2):100.
-
Charan J, Yadav P, Saxena D, Kantharia ND. Drug advertisements published in Indian Medical Journals: Are they ethical? J Pharm Bioallied Sci 2011 Jul;3(3):403-6.
-
Mali SN, Dudhgaonkar S, Bachewar NP. Evaluation of rationality of promotional drug literature using World Health Organization guidelines. Indian J Pharmacol 2010;42:267–72.
-
Jadhav SS, Dumatar CB, Dikshit RK. Drug promotional literature (DPLs) evaluation as per World Health Organization (WHO) criteria. J Appl Pharmac Sci 2014;4(06):084-088.
-
Ganashree P, Bhuvana K, Sarala N. A critical review of drug promotional literature using the World Health Organization guidelines. J Res Pharm Pract 2016;5(3):162–165
-
Vachhani PM, Solanki MN, Desai MK. An evaluation of drug promotional literature published in scientific medical journals. J Pharm Bioallied Sci 2016;8(3):248–252.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License