IJCRR - 13(9), May, 2021
Pages: 88-94
Date of Publication: 07-May-2021
Print Article
Download XML Download PDF
Association of Anti-TPO Antibodies with Insulin Resistance and Dyslipidemia in Hashimoto's Thyroiditis: An Observational Study on South Indian Population
Author: Rajarajeswari R, Sumathi S, Asmathulla S, Srinivasan AR, Girija S, Maithili Karpaga Selvi N
Category: Healthcare
Abstract:Introduction: Hashimoto's Thyroiditis (HT) is a frequently observed autoimmune thyroid disease and the commonest cause of hypothyroidism. Although the association of hypothyroidism with cardiovascular risk is a well-documented fact, it is still not clear as to whether thyroid autoimmunity is an independent risk factor for atherosclerosis, a cardiometabolic risk factor.
Objective: In this study, we attempted to elicit the probable association of Anti \?Thyroid peroxidase (Anti-TPO) antibodies with cardiometabolic factors (insulin resistance and dyslipidemia) in patients.
Methods: In this observational study, sixty-five healthy controls and sixty-eight HT patients were enrolled. Serum concentrations of TSH, FT4, FT3, (Anti-TPO), Total Cholesterol (TC), Triglycerides (TG), HDL-Cholesterol (HDL-C), Very Low-Density Lipoprotein (VLDL), Fasting Blood Glucose (FBG) and Fasting Insulin levels were measured. LDL-Cholesterol (LDL-C), HOMA-IR and Atherogenic Index of Plasma (AIP) were calculated.
Results: There was a significant increase in TC, TG, LDL, VLDL, AIP, Fasting Insulin and HOMA-IR among the HT group (P < 0.001) compared to healthy controls. Despite a significant positive correlation between Anti-TPO antibodies and TC, LDL, Fasting Insulin, HOMA-IR, Anti-TPO was found to be independently associated with AIP as revealed in Regression analysis, which unequivocally demonstrates the cardiovascular risk in HT.
Conclusion: The findings of the present study point to the implication of thyroid autoimmunity in insulin resistance and dyslipidemia, independent of thyroid function in patients with HT. Thus, the assessment of lipid profile variables and insulin resistance possess value in the treatment and management of HT patients.
Keywords: Hypothyroidism, Thyroid autoimmunity, Anti-Thyroperoxidase antibodies, Hyperlipidemia, Insulin resistance, Cardiovascular disease risk
Full Text:
Introduction
Hashimoto’s Thyroiditis (HT) is a genetic autoimmune disease, characterized by the destruction of thyroid cells by cell- and antibody-mediated immune responses1 and is regarded as the commonest cause of hypothyroidism. HT has a prevalence rate of 1-4% and found to be more common in women and increases with age.2,3 Elevated circulating antibodies to thyroid antigens is a characteristic feature in HT patients, of which anti-thyroid peroxidase antibodies (anti-TPO) being the most specific and sensitive for the diagnosis of the disease.2 Several studies have found that hypothyroidism is associated with insulin resistance, dyslipidemia and chronic inflammation and in turn, increases the risk for atherosclerosis.4,5 Though the exact mechanism of the atherosclerotic process in hypothyroidism remains to be delineated, few studies suggest the role of thyroid autoimmunity in atherosclerosis, independent of thyroid function.6-8 Inflammation observed in HT may induce alterations in lipid metabolism, contributing to the increased risk of atherosclerosis.9 Few studies have reported insulin resistance in hypothyroid state10,11, whereas, a few other studies failed to demonstrate the relationship.12,13 The mechanism connecting hypothyroidism to insulin resistance is not clear. Mazaheri et al suggested that an imbalance between pro-inflammatory and anti-inflammatory cytokines resulting from chronic inflammation can be one of the mechanisms behind insulin resistance in HT. They had studied in euthyroid HT patients on levothyroxine treatment, which was believed to attenuate the inflammation in HT and might mitigate the study results.14 Thus, in this study we aimed to study the association of thyroid autoimmunity with cardiometabolic factors in newly diagnosed Hashimoto’s Thyroiditis subjects, at a tertiary care hospital from South India, which is, to our knowledge, one among the few studies.
MaterialS and Methods
This observational study was designed to include sixty-eight newly diagnosed and untreated HT patients and sixty-five age and gender-matched healthy controls who had attended the clinics at a tertiary care hospital in South India. The patients who had elevated serum TSH levels, with or without elevated fT3 and fT4, high serum anti-TPO and enlarged rubbery thyroid were categorized as Hashimoto’s thyroiditis.14 Patients with diabetes mellitus, cardiovascular disease, pregnancy, thyroid cancer and any other autoimmune disease such as lupus erythematosus and rheumatoid arthritis were excluded from the study. All the participants were enrolled for the study following written informed consent. The study was conducted following the Declaration of Helsinki and was approved by the institute ethics committee (IEC letter no.: SMVMCH-EC/DO/AL/817/2018).
Anthropometric measurements and laboratory assessments were carried out on all the participants, as per established protocol. Bodyweight and height were measured with a calibrated digital scale and stadiometer respectively. Body Mass Index (BMI) was calculated as weight (kg) divided by height (m) squared. Waist circumference (WC) was measured midway between the lowest rib and iliac crest at umbilicus level and Hip circumference (HC) was measured at the widest girth of the Hip using a measuring tape in centimetres. Waist to Hip Ratio (WHR) was calculated by WC divided by HC. Blood pressure values were measured in the sitting position. Demographic information and family history of diabetes, hypertension and any thyroid disorder were obtained from the participants.
Blood was collected from all the participants in the fasting state and subjected to centrifugation at 3000 rpm. The serum separated was used for the estimation of biochemical parameters. Glucose was measured by Glucose oxidase – peroxidase method, total cholesterol (TC) was measured by cholesterol oxidase method and triglyceride (TG) levels were measured by glycerol kinase- peroxidase method, high-density lipoprotein cholesterol (HDL-c) was measured by divalent cation precipitation method using the reagents adapted to Biolis 50i, Tokyo BoekiMedisys, Japan. Very low-density lipoprotein cholesterol (VLDL) level was computed from triacylglycerol concentration and low-density lipoprotein cholesterol (LDL-C) level was calculated using Friedwald’s formula. Atherogenic Index of Plasma (AIP) is the logarithmically transformed ratio of TG to HDL-C.15
Serum concentrations of fT3, fT4 and TSH were measured using the Chemiluminescence (CLIA) method adapted to Advia Centaur XP, Siemens, USA. Anti-TPO was determined by Medizym Anti-TPOEnzyme Linked Immunosorbent Assay (ELISA) kit, Berlin, Germany. Insulin was measured using DRG Insulin ELISA kit, Germany and Insulin Resistance (IR) was assessed by Homeostasis Model assessment index (HOMA-IR = Fasting Insulin (µIU/mL) x Fasting Glucose (mg/dL)/405.
Statistical Analysis
Statistical Analysis was performed using licenced SPSS software 20.0 version. Data were presented as mean ± standard deviation (SD) or median (inter-quartile range), wherever appropriate. Differences between the two groups were tested using Independent Student ‘t’ test for normally distributed variables and the Mann-Whitney U test for non-normal distributed variables. Spearman’s correlation was used to determine the correlation of TSH and Anti-TPO with other variables. Based on Anti-TPO levels, the HT group was further divided into two groups and the between-groups comparison of continuous variables among the control group and two subgroups of HT was performed by Oneway ANOVA. P-value < 0.05 was considered to be statistically significant.
Results
A total of 133 subjects including sixty-five healthy controls (three males and sixty-two females) and sixty-eight Hashimoto’s Thyroiditis patients (two males and sixty-six females) were enrolled in this study. Anthropometric parameters and Thyroid profile of the two groups were compared in Table 1.
Based on the data obtained, there was a significant difference between the mean values of the HT group and the control group in terms of BMI, WC, WHR, fT3, fT4, TSH and Anti – TPO antibodies (Table 1).
Table 2 represents the comparison of cardiometabolic factors between HT group and healthy controls. Correlation analyses of TSH and Anti-TPO with other biochemical parameters were enabled.TSH was found to be positively correlated with Anti-TPO (r value = 0.813, P value = 0.000), LDL (r value = 0.275, P value = 0.023), TC/HDL (r value = 0.256, P value = 0.035) and LDL/HDL (r value = 0.302, P value = 0.012).
Table 3 represents the correlation of anti-TPO with cardiometabolic factors. On correlation analysis, HOMA-IR was found to be correlated only with Anti-TPO. But when HOMA-IR was subjected to Linear Regression analysis, it was found to be independently associated with WHR (Standardized Co-efficient ‘Beta’ = 0.039, P-value < 0.05). On Linear Regression analysis, TSH, TC/HDL and AIP were found to be independently associated with Anti-TPO antibodies (Table 4). Based on Anti-TPO levels, the HT group was subdivided into two (HT Group I: HT patients with Anti-TPO level less than 1000 IU/L and HT Group II: HT patients with Anti-TPO level more than or equal to 1000 IU/L). Patients with Anti TPO antibody levels of more than 1000 IU/ml were classified as having highly positive antibodies. Anthropometric and Biochemical parameters were analyzed among controls, HT Group I and HT Group II, the data of which is shown in Table 5.
Discussion
Studies have demonstrated that HT is the primary cause of non- iatrogenic hypothyroidism. Hypothyroidism has been observed to enhance the risk for cardiovascular disease and atherosclerosis. Though reports are available on metabolic indices in euthyroid subjects with HT, the results are inconsistent and at times paradoxical. Furthermore, it remains to be seen as to whether thyroid autoimmunity is an independent risk factor for atherosclerosis. Thus, in this observational study, we aimed to compare the anthropometric parameters, lipid profile variables and Insulin resistance between the newly diagnosed HT subjects and healthy controls, among the South Indian population.
The findings of the present study depict a higher prevalence of insulin resistance and dyslipidemia in patients with HT when compared to healthy controls. In our study, we found a significant increase in the levels of fasting Insulin as well as Insulin resistance in HT patients when compared to controls. Though this result is in line with previous studies16,17, several other studies have not shown any significant difference between the HT patients and healthy controls.18,19 A study conducted by Mazaheri et al., demonstrated a significant increase in fasting insulin only in HT subjects with highly positive Anti-TPO antibodies, whereas it failed to demonstrate any difference in fasting Insulin and HOMA-IR between patients with and without HT.14 There was no correlation between TSH and HOMA-IR in all the patients with HT. However, Anti-TPO antibody concentrations were found to be positively correlated with HOMA-IR in HT subjects. However, in the present study when HT subjects were subdivided into two groups based on Anti-TPO levels and analyzed, the HT group with highly positive Anti-TPO antibody showed a significant increase in HOMA-IR when compared to the HT group with Anti-TPO concentration less than 1000 IU/L. These findings suggest that thyroid autoimmunity may be associated with insulin resistance independent of thyroid function, as reported earlier in the Iranian and Chinese populations.14,16 The anti-TPO antibodies in all probability might possess a destructive effect on thyrocytes by activating T helper 1 cells, and resultant production of proinflammatory cytokines such as TNF-α and IL-6 resulting in inflammation.20 The monocytes, macrophages, lymphocytes in association with the proinflammatory cytokines – particularly, TNF-α and IL-6, may contribute to insulin resistance and atherosclerotic plaques.21,22 Insulin resistance may impair the PI3-K/AKt pathway, decreasing the bioavailability of Nitric Oxide, which might contribute to endothelial dysfunction.16 The present study also exhibits an association between HOMA-IR and WHR in Linear regression analysis, which would explain the fact that insulin resistance and central adiposity would co-exist14 which is another risk factor for cardiovascular diseases.
The relationship between thyroid function and serum lipids had been investigated in previous studies. In a study conducted by Tamer et al, HT subjects were found to have higher TC, LDL-C, TG and non-HDL-C levels as compared to healthy controls. The same study did not reveal any difference in the levels of TC and LDL-C between overt hypothyroid and euthyroid patients with HT. Though there was no correlation between TSH and any other lipid parameters, Anti-TPO antibodies were found to be positively correlating with TG levels in HT subjects, suggesting the association of thyroid autoimmunity with dyslipidemia, independent of thyroid function.23 In a previous report, the subclinical hypothyroid patients manifested an increase in atherosclerotic cardiovascular disease despite having low TC levels than controls.24 In the present study, serum TC, TG, LDL-C and VLDL levels were found to be significantly elevated in HT subjects when compared to healthy controls. In contrast to the previously reported studies, a positive correlation was found between TSH and LDL – C levels in HT subjects. Anti-TPO antibodies were also correlating positively with TC and LDL – C levels. The result of our study has been augmented by the HUNT study and Tagami et al., where a positive correlation was observed between TSH and lipid levels.25,26
Being an autoimmune disease, HT itself may be a risk factor for the development of cardiovascular disease. According to Volpe’s hypothesis, in HT, due to defective suppressor T cells, the helper T cells are not suppressed leading to a production of various cytokines such as IFN – γ, IL-2 and TNF– α27 which might cause weight gain and hyperlipidemia.28 Taddei et al29 demonstrated that endothelial dysfunction and impaired NO availability were associated with atherosclerosis rather than the thyroid function itself. Tamer et al.23, also reported the association of thyroid autoimmunity with hyperlipidemia, independent of thyroid function. The data from the present study reveals that although TSH demonstrated a positive correlation with LDL-C, it lost its significance on regression analysis. Also, in Linear Regression analysis, Anti-TPO antibodies were found to be independently associated with AIP. Being a surrogate marker of, small dense LDL (sdLDL), the AIP, has been indicated as a superior predictor of cardiovascular risk than the other lipid parameters.30 Since sdLDL is more susceptible to oxidation and glycation and exhibits decreased clearance attributed to the reduced affinity for LDL receptor, the predominance of sdLDL indicates an increased risk for atherogenesis.31,32
To the best of our knowledge, this is among the few studies to show the association of anti-TPO with insulin resistance and dyslipidemia among the south Indian population. The main limitations of the present study are the lack of a euthyroid and hypothyroid group without elevated anti-TPO and small sample size. Further studies, including subjects at different thyroid function levels with and without elevated anti-TPO and larger sample size, may throw more light and provide a deeper insight into the relationship of anti-TPO with hyperlipidemia and insulin resistance - the risk factors for cardiovascular disease. Significance of the study: The study opens up newer vistas in biochemical pharmacology and clinical endocrinology, especially concerning combinatorial drug therapy that could help in the effective and rational management of cardiovascular morbidity, in the light of insulin resistance, thyroid autoimmunity and dyslipidemia.
Conclusion
To conclude, the present study has demonstrated that thyroid autoimmunity might possess effects on insulin resistance and hyperlipidemia, thus increasing the risk for cardiovascular disease. Therefore, it is imperative to objectively determine the lipid levels and insulin resistance that would aid in the management of accompanying cardiovascular disease in patients with Hashimoto’s thyroiditis.
Acknowledgement: The authors would like to acknowledge Mr. Srinivasan for extending his technical support in conducting this study.
Financial support and Sponsorship: None declared.
Conflict of interest: Nil
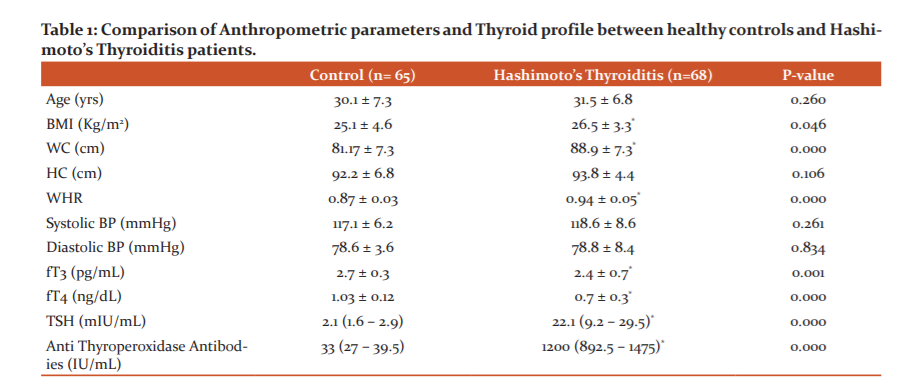
BMI : Body Mass Index, WC: Waist Circumference, HC : Hip Circumference, WHR : Waist Hip Ratio, TSH : Thyroid Stimulating Hormone
The data are represented as mean ± S.D / Median (inter-quartile range)
P value calculated by Independent ‘t’ test for normal distributed parameters and Mann-Whitney U test for Non-normal distributed parameters


TC : Total Cholesterol, TG : Triglyceride, HDL : High Density Lipoprotein, LDL : Low Density Lipoprotein, VLDL : Very Low Density Lipoprotein, HOMA-IR : Homeostatic Model Assessment of Insulin Resistance, AIP : Atherogenic Index of Plasma
The data are represented as mean ± S.D
P value calculated by Independent ‘t’ test for normal distributed parameters and Mann-Whitney U test for Non-normal distributed parameters

TC : Total Cholesterol, HDL : High Density Lipoprotein, LDL : Low Density Lipoprotein, TSH : Thyroid Stimulating Hormone, HOMA-IR : Homeostatic Model Assessment of Insulin Resistance
P value calculated by Spearman correlation method


HT Group I : Anti-TPO < 1000 IU/mL, HT Group II : Anti-TPO ≥ 1000 IU/mL
P value less than 0.05 = ‘*’ :in comparison to Controls , ‘†’ : in comparison to HT Group II
References:
1. Yang M, Du C, Yinpingwang, Liu J. CD19+CD24hiCD38hi regulatory B cells are associated with insulin resistance in type I Hashimoto's thyroiditis in Chinese females. Exp Therap Med 2017;14:3887-3893.
2. Cunha CA, Neves C, Neves J, Oliveira SC, Sokhatska O, Dias C, et al. Cardiovascular risk factors in patients with autoimmune thyroiditis. Rev Port Endocrinol Diabetes Metab 2017;12:133-141
3. Sood N, Nigam JS. Correlation of fine needle aspiration cytology findings with thyroid function test in cases of lymphocytic thyroiditis. J Thyroid Res 2014;2014:430510.
4. Yang N, Yao Z, Miao L, Liu J, Gao X, Fan H, et al. Novel clinical evidence of an association between homocysteine and insulin resistance in patients with hypothyroidism or subclinical hypothyroidism. PLoS One 2015;10(5):e0125922.
5. Lu M, Yang CB, Gao L, Zhao JJ. Mechanism of subclinical hypothyroidism accelerating endothelial dysfunction (review). Exp Ther Med 2015;9:3–10.
6. Bastenie PA, Vanhaelst L, Neve P. Coronary artery disease in hypothyroidism. Lancet 1967; 2:1221-1222.
7. Bastenie PA, Vanhaelst L, Bonnyns M, Neve P, Staquet M. Preclinicalhypothyroidism: a risk factor for coronary heart disease. Lancet 1971;1:203-204.
8. Bastenie PA, Vanhaelst L, Golstein J, Smets P. Asymptomatic autoimmune thyroiditis and coronary heart disease. Cross-sectional and prospective studies. Lancet 1977;2:155-158.
9. van Diepen JA, Berbee JF, Havekes LM, Rensen PC. Interactions between inflammation and lipid metabolism: relevance for the efficacy of anti?inflammatory drugs in the treatment of atherosclerosis. Atherosclerosis 2013;228:306-315.
10. Rochon C, Tauveron I, Dejax C, Benoit P, Capitan P, Fabricio A, et al. Response of glucose disposal to hyperinsulinaemia in human hypothyroidism and hyperthyroidism. Clin Sci (Lond). 2003;104:7–15.
11.Stanicka S, Vondra K, Pelikanova T, Vlcek P, Hill M, ZamrazilV.Insulin sensitivity and counter-regulatory hormones in hypothyroidism and during thyroid hormone replacement therapy. Clin Chem Lab Med 2005;43:715–720.
12. Owecki M, Nikisch E, Sowinski J. Hypothyroidism has no impact on insulin sensitivity assessed with HOMA-IR in totally thyroidectomized patients. Acta Clin Belg 2006;61:69–73.
13. Harris PE, Walker M, Clark F, Home PD, Alberti KG. Forearm muscle metabolism in primary hypothyroidism. Eur J Clin Invest 1993;23:585–8.
14. Mazaheri T, Sharifi F, Kamali K.Insulin resistance in hypothyroid patients underLevothyroxine therapy: a comparison between those with and without thyroid autoimmunity. J Diab Metab Disor 2014;13:103.
15. Dobiásová M, Frohlich J. The plasma parameter log (TG/HDL-C) as an atherogenic index: correlation with lipoprotein particle size and esterification rate in apoB-lipoprotein-depleted plasma (FER(HDL)). Clin Biochem 2001;34(7):583-588.
16. Liu J, Duan Y, Fu J, Wang G. Association between thyroid hormones, thyroid antibodies and cardiometabolic factors in non-obese individuals with normal thyroid function. Front Endocrinol 2018;9:130.
17. Dimitriadis G, Mitrou P, Lambadiari V, Boutati E, Maratou E, Panagiotakos DB, et al. Insulin action in adipose tissue and muscle in hypothyroidism. J Clin Endocrinol Metab 2006;91: 4930?4937.
18. Mousa U, Bozku? Y, Kut A, Demir CC, Tutuncu NB. Fat distribution and metabolic profile in subjects with Hashimoto’s Thyroiditis. Acta Endocrinologica (Buc). 2018;14(1):105-112.
19. Amouzegara A, Kazemiana E, Gharibzadeha S, Mehran L, Tohidib M, Azizia F. Association between thyroid hormones, thyroid antibodies and insulin resistance in euthyroid individuals: A population-based cohort. Diabetes Metab. 2015. Front Endocrinol (Lausanne) 2018;9: 130.
20. Nielsen CH, Brix TH, Leslie RQ, Hegedus LA. Role for autoantibodies in the enhancement of pro-inflammatory cytokine responses to a self-antigen, thyroid peroxidase. Clin Immunol 2009;133(2):218–227.
21. Wei Y, Chen K, Whaley-Connell AT, Stump CS, Ibdah JA, Sowers JR. Skeletal muscle insulin resistance: role of inflammatory cytokines and reactive oxygen species. Am J Physiol Regul Integr Comp Physiol 2008;294:673–680.
22. Wilson HM, Barker RN, Erwig LP. Macrophages: promising targets for the treatment of atherosclerosis. Curr Vasc Pharmacol 2009;7:234–243.
23. Tamer G, Mert M, Tamer I, Messi B, DamlaK?l?c, Ar?k S. Effects of thyroid autoimmunity on abdominal obesity and hyperlipidaemia. Polish J Endocrinol 2011;62:421-428.
24. Hak AE, Pols HA, Visser TJ, Drexhage HA, Hofman A, Witteman JC. Subclinical hypothyroidism is an independent risk factor for atherosclerosis and myocardial infarction in elderly women: The Rotterdam Study. Ann Intern Med. 2000;132: 270-278.
25. Asvold BO, Vatten LJ, Nilsen TI, Bjoro T. The association between TSH within the reference range and serum lipid concentrations in a population-based study. The HUNT study. Eur J Endocrinol 2007;156:181-186.
26. Tagami T, Tamanaha T, Shimazu S. Lipid profiles in untreated patients with Hashimoto’s thyroiditis and the effects of thyroxine treatment on subclinical hypothyroidism with Hashimoto’s thyroiditis. Endocrine J 2010;57:253-258.
27. Volpe R. Autoimmune Thyroiditis. Braverman LE, Utiger RD (ed). Werner and Ingbar’s. The Thyroid. JB Lippincott Co, Philadelphia.1991; 921-941.
28. Sultan A, Strodthoff D, Robertson AK. T cell-mediated inflammation in adipose tissue does not cause insulin resistance in hyperlipidemic mice. Circ Res 2009;104:961-968.
29. Taddei S, Caraccio N, Virdis A, Dardano A. Low-grade systemic inflammation causes endothelial dysfunction in patients with Hashimoto’s thyroiditis. J Clin Endocrinol Metab 2006; 91:5076-5082
30. Baliarsingh S, Sharma N, Mukherjee R. Serum uric acid: a marker for atherosclerosis as it is positively associated with "atherogenic index of plasma. Arch Physiol Biochem 2013;119:27-31.
31. Berneis KK, Kraus RM. Metabolic origins and clinical significance of LDL heterogeneity. J Lipid Res 2002;43:1363–1379.
32. Austin MA, King MC, Vranizan KM, Krauss RM. Atherogenic lipoprotein phenotype. A proposed genetic marker for coronary heart disease risk. Circulation 1990;82:495–506.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License