IJCRR - 13(7), April, 2021
Pages: 209-215
Date of Publication: 12-Apr-2021
Print Article
Download XML Download PDF
The Anticancer Activity of Artemisia Judaica Crude Extract in Human Hepatocellular Carcinoma HepG2 Cells by Induction of Apoptosis and Cell Cycle Arrest
Author: Neima K. Al-Senosy, Naglaa M. Ebeed, Lamiaa M. Salem, Shenouda M. Girgis, Ekram S. Ahmad
Category: Healthcare
Abstract:Introduction: Cancer is one of the major causes of death around the world. Medicinal plants have been investigated across the world to exploit their potential anticancer activity on a large scale of applications. Objective: The present study is focused on the anticancer activity of the methanolic extract of the medicinal plant, Artemisia Judaica, on Human hepatocellular carcinoma (HepG2) and normal liver (THLE2) cell lines, as well as the mechanisms involved were also investigated. Methods: The collected aerial parts of the plant were extracted by maceration with methanol and the crude extract was collected and stored until use. Cytotoxicity and cell proliferation, cell cycle analysis and the expression level of apoptosis-related genes such as p21, CycB1, CDK1, p53, Bcl-2 and Bax in HepG2 cell lines were estimated using the MTT colourimetric, flow cytometry and quantitative real-time PCR (qRT-PCR) assays, respectively. Results: The results showed that A. Judaica extract (IC50 = 33.76\?g/ml) displayed strong cytotoxicity and antiproliferative effect in HepG2 cancer cells. On the other hand, this extract exhibited no cytotoxic activity on the liver normal cell line (THLE2). Flow cytometric analysis of propidium iodide staining revealed that the treatment of HepG2 cells with A. Judaica led to an increase in G2/M phase cell cycle arrest. The qRT-PCR assay revealed that both cyclin B1 (cycB1) and cyclin-dependent kinase (CDK1) genes, as well Bcl-2 showed down-regulation expression levels in HepG2 treated with A. Judaica compared to the untreated cell line. Furthermore, the apoptotic mechanism activated by the plant extract resulted in up-regulation of p53, P21and Bax at mRNA level on HepG2 cell line. Conclusion: These results suggest that A. Judaica could be a promising candidate species as a natural source of anticancer molecules.
Keywords: Artemisia Judaica, Antiproliferation, Apoptosis, Anticancer, HepG2 cell line
Full Text:
INTRODUCTION
Cancer is one of the major causes of death worldwide. It is characterized by genetic alterations of normal cells which become malignant cells. Those are characterized by uncontrolled cell growth, immortality, invasiveness, and the ability to form distant metastasis. It was found that natural products may interfere with the carcinogenesis process by altering the behaviour of tumour cell and targeting cancer cells signalling pathways.1
Among different cancer types, liver cancer is the 4th most common cause of death from cancer worldwide, the incidence of human hepatocellular carcinoma (HCC) is increasing particularly in males in some countries. Hepatitis B and C viruses (HBV and HCV) and dietary aflatoxin intake remain the major causative factors of HCC, where different modes of cancer therapy for HCC have been tried such as surgery, chemotherapy, radiotherapy, as well, tremendous works have been done at the molecular level.2
The use of natural products and supplements of medicinal plants has very increased over the past three decades with more than 80% of people worldwide depend on them for some part of primary healthcare.3 The diet enriched with naturally occurring substances significantly reduces the risk for some cancers. Consequently, many drugs used for the treatment of cancer have been discovered from medicinal plants.4
Biologically active components of the medicinal plants target tumour cells by different mechanisms, resulting in angiogenesis, inhibition of carcinogenesis, cell cycle arrest, oxidative stress, autophagy or differentiation and apoptosis.5-9 Artemisia was found to possess sesquiterpene lactones and other derived phytochemicals as active components. Sesquiterpene lactones were used for their therapeutic and other properties.10 Recently, monoterpenes, sesquiterpenes, sesquiterpene lactones, flavonoids, coumarins, sterols, polyacetylenes have been isolated from Artemisia species.11 Some Artemisia species have shown that it possesses medicinal properties such as anti-bacterial and anti-cancer effects.12 Few species of Artemisia such as Artemisia Judaica, showed promising anticancer activities against the proliferation of cancer cell lines.13 The main effect of its essential oils is attributed to the main constitutes, the thujone, in this species,14 enriched with fraction has potential anticancer activities.15
A. Judaica (Arabic name, Shih Balady) is a perennial fragment shrub that is abundant in North Africa and Middle Eastern countries.16 As well in Saudi Arabia, Yemen and Egypt.17-19 This plant grows abundantly in different parts of the Arabian peninsula such as Saudi Arabia and Yemen,17,18 and has been used in traditional Egyptian medicine for the treatment of gastrointestinal disorders.19 Besides, the Artemisia species have been used in Iranian traditional medicine as an anti-infectious, anti-bacterial, gastric tonic, digestive and stomachic.20 Isolated compounds from A. Judaica have exhibited antiviral, antibacterial, antifungal, and cytoprotective effects,21-23 and for the treatment of hepatitis, cancer and menstrual-related disorders.24 The composition of A. Judaica includes artemisinic acid, sesquiterpene lactones, methyl wormwood, artemisinic alcohol, eucalyptol, Artemisia ketone, camphor, caryophyllene, piperitone and essential oil.25-26 Besides, diverse chemical components, such as flavonoids, coumarins, sterols, polyacetylenes, monoterpenes, polyphenols, sesquiterpenes and sesquiterpenes lactones, have been found in plants from the Artemisia genus.22,24
Sesquiterpene lactone is also a potent apoptotic inducer in cancer cells via multiple pathways. It is readily depleted intracellular glutathione (GSH), disrupts cellular redox balance, depletes intracellular thiols,27 triggers an intracellular reactive oxygen species (ROS),28 decreases anti-apoptotic Bcl-2 protein expression and induce apoptosis.29 Studies of the molecular mechanisms have shown that caspase and p53-independent activation, downregulation of Bcl-2, generation of ROS, reduction of mitochondrial membrane potential, increased amount of Bax protein, arrest of the cell cycle at G2/M-phase, suppression of the Notch 1, and inhibition of NF-κB are important mechanisms for the cytotoxic effects of Artemisia.30 As well, flow cytometric analysis for cell cycle revealed increment of G2/M phase cell cycle arrest after treatment of HepG2 cells with A. herba alba extract. The apoptotic mechanism was also activated by the crude extract of Artemisia included up-regulation of p53 and Bax and downregulation of Bcl-2 expression levels with no cytotoxic effect for normal cell lines exposed to the plant extract.31
Besides, Artemisia extract increased the number of cells in the G2/M phases, followed by caspase 3 upregulation, and apoptosis. Further, it is inhibited cancer cell proliferation and induced apoptosis.32 Therefore; the present study is designed to evaluate the anticancer activity of crude extract of A. Judaica, on human hepatocellular carcinoma (HepG2) cell line. The possible underlying antiproliferation mechanisms were investigated, by studying the effect of A. Judaica on cytotoxicity, cell cycle arrest and apoptosis-related genes in the human hepatocellular carcinoma (HepG2) cell line.
MATERIALS AND METHODS
Plant collection and extract preparation
The aerial parts (leaves and stems) of A. Judaica were collected during the summer season (July 2020) from Wadi Gharandal, South Sina, Egypt. Taxonomical identification was confirmed by a botanist and voucher specimen, were deposited in the Herbarium of the National Research Center, Dokki, Cairo, Egypt. Herbs were washed and shade dried for a week and was milled to fine powder, then it was extracted with methanol at a ratio of 20 g dry powder in 200 ml of ethanol for 48 h using the maceration method. The liquid extract was filtered and concentrated under vacuum by soxhlet extraction, then stored in the dark at 4°C until use.
Assessment of cytotoxic and anti-proliferous activities of A. Judaica
Cell proliferation and viability of the cells were estimated using MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide) colourimetric assay. Human hepatocellular carcinoma (HepG2) and normal liver (THLE2) cell lines in exponential growth phase were seeded at a density of 1 x 104 cells per well (100 μl/well) onto a 96-well plate (Falcon, Franklin Likes, NJ, USA) in DMEM medium (GIBCO, Grand Island, New York, USA; Cat.no.A1049101). The cell density was adjusted by the trypan blue exclusion method. The whole compounds were cultured in different concentrations (ranged from 0 to 200 µg/mL) for 24 hours at 37 °C in a 5% CO2 with a 95% humidity incubator. Besides, different concentrations of cisplatin as a reference chemotherapeutic drug were added and the microplates were incubated for a further 48 hour in DMEM medium (200 µL). The medium was washed gently twice with ice-cold PBS and a volume of 200 μL MTT [3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide, a yellow tetrazole, (Molecular probes, Eugene, Oregon, USA; Cat.no.V-13154)] was added to each well. The microplate was incubated at 37 °C for another 4 hours in a CO2 incubator. About 180 μL medium/MTT was removed and 100 μL of acidified isopropanol were added per well to solubilize the formazan produced. Finally, the microplate was incubated with shaking for 15 minutes. The absorbance of each well was measured at 630 nm using a microplate reader (ELX800, Biokit, Spain). Assays were performed in triplicate on three independent experiments. Sigmoidal and dose-dependent curves were constructed to plot the results of the experiment. The concentration of the compounds inhibiting cancer cell growth by 50% of the control level (IC50) was calculated using this sigmoidal curve.
Cell cycle analysis by flow cytometry
To evaluate the effect of A. Judaica crude extract on the cancerous cell division of HepG2, the cell cycle analysis was carried out using the protocol of Applied Bio-system, USA. Cells were digested with warm Trypsin-EDTA + warm PBS-EDTA (0.25%) (500 µl + 500 µl) with incubation for 10 minutes at 37°C. The mixture was centrifuged at 450 rpm for 5 min, and then the supernatant was carefully removed. The mixture was washed twice in warm PBS and the cell pellet was re-suspended in 500 µl warm PBS, centrifuged and the supernatant was removed. A volume of 150 µl PBS + 350 µl ice-cold 70% ethanol was added and incubated at 4°C for 1 hour to fix the cells. To remove ethanol, the mixture was centrifuged at 350 rpm for 10 minutes and then the supernatant was carefully removed. The mixture was washed twice in warm PBS and the cells were re-suspended in 500 µl warm PBS, centrifuged and the supernatant was removed. The cells were re-suspended in 100 µl PBS and stored at 4° for up to 4 days. In the darkness, the cells were stained with 100 µl of propidium iodide (PI) solution + 50 µl RNase A solution (100 µg/ml) and incubated in darkness for 30-60 min. The stained cells were read in Attune flow cytometer (Applied Bio-system, USA).
Determination of the expression levels of apoptosis-regulatory genes by quantitative real-time PCR
RNA extraction was carried out utilizing an RNeasy Mini kit (Qiagen, Inc., Valencia, CA, USA) for HepG2 cell lines, or treated with IC50 of A. Judaica extract for 24 h. The procedure was performed according to the manufacturer’s instructions. The cDNA was synthesized from purified RNA with an RT2 First Strand Kit (Qiagen, Inc., Valencia, CA, USA) according to the manufacturer’s guidelines as the template for RT-qPCR. Corbett Rotor-Gene 6000 (Qiagen, Inc., Valencia, CA, USA) was used to perform quantitative real-time reverse transcriptase PCR (qRT-PCR). A final volume of 25 µl pre-mix was prepared to contain 12.5 µl of RT2 SYBR R Green ROXTM FAST master mix (Qiagen, Inc., Valencia, CA, USA), 1 µl of primers (RT2 qPCR Primer Assays, Qiagen, Inc., Valencia, CA, USA), 1 µl of cDNA, and 10.5 µl RNase-free water to make the final volume. The following primer pairs for target genes and the housekeeping β-actin gene was chosen from the Primer Bank website.
The default PCR conditions were as follows: the PCR plate was run at 95?C for 10 min to activate the enzyme, 40 cycles of 15 s at 95?C (denaturation) followed by 30 s at 60?C (annealing and synthesis). Finally, the dissociation curve was constructed immediately after the PCR run to check and verify results. The housekeeping gene β-actin was used as a reference to calculate fold change in target gene expression.
Statistical Analysis
Data were expressed as means ± standard error (SEM). One way ANOVA using SPSS 18.0 software was used to detect the significant difference. Values were considered statistically significant when P≤0.05. Comparison of means was carried out with Tukey’s HSD test.
RESULTS
Cytotoxic activity of A. Judaica crude extract using MTT assay
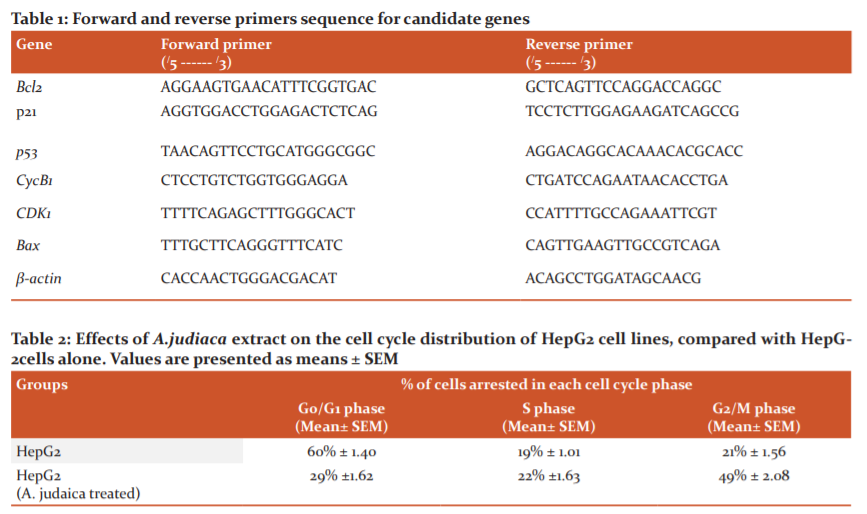
The cytotoxic activity of A. Judaica crude extract was screened on human hepatocellular carcinoma cell line (HepG2) by MTT assay (Fig. 1). The results revealed that A. Judaica extract (IC50 = 33.76μg/ml), inhibited the proliferation and exhibited increasing in cytotoxic activity to HepG2 cell line in a dose-dependent manner, where, the percentage of cell viability declined up to 20% by A. Judaica dose increment, while the crude extract of A. Judaica didn’t induce growth inhibitory effect on normal liver cell line (THLE2) (IC50 = 1041μg/ml), where, a very low cytotoxic activity was noticed by A. Judaica against normal liver cell line.
Effect of A. Judaica crude extract on cell cycle arrest using flow cytometry
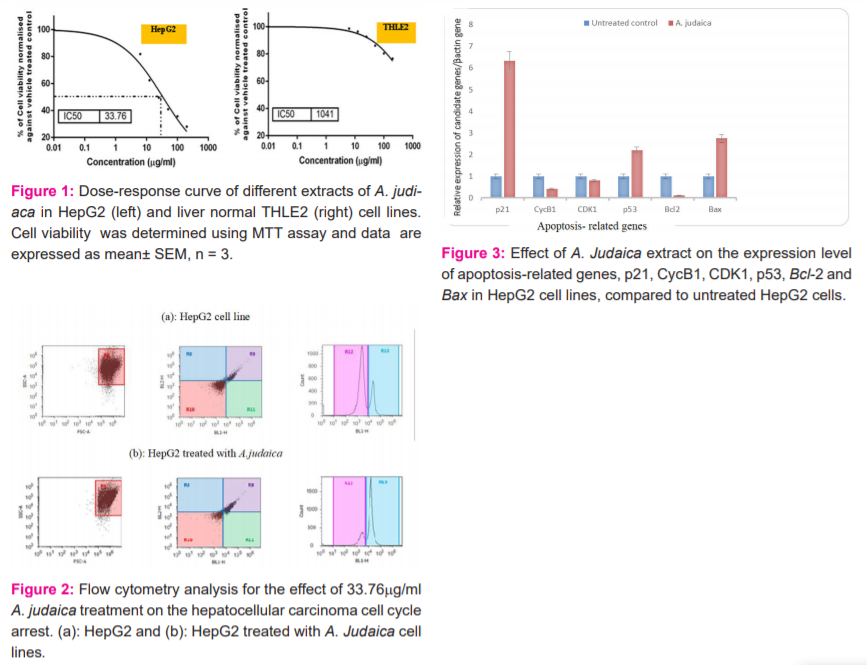
The DNA content in A.judaica treated HepG2 cells was assessed using the cell cycle phase distribution analysis. The cell cycle arrest was analyzed using flow cytometry (Fig. 2). According to the inhibition rate of liver cancer (HepG2) cell viability, the results revealed that the anti-proliferative effect of A. Judaica was associated with cell cycle arrest using flow cytometry-based cell cycle distribution (Table 1). Compared to the control group, the treatment with A.judaica crude extract at 33.76 μg/ml affected the cell cycle distribution on HepG2 cells, where, the G0/G1 phase showed decreased from 60% to 29% of cell cycle arrest, while the S-phase percentage increased slightly from 19% to 22% in the HepG2 and HepG2 treated with A.judaica crude extract, respectively. Interestingly, the percentage of HepG2 cells at the G2/M phase was increased (49%) compared to (21%) for the untreated cell line. These results suggested that A. Judaica inhibited the cellular proliferation of HepG2 cells via G2/M phase arrest of the cell cycle.
The expression level of apoptosis-related genes modulated by A. Judaica
To evaluate the molecular mechanism of A. Judaica-induced apoptosis in HepG2 cells, the expression level of apoptosis-related genes such as p21, cyclin B1 (CycB1), cyclin-dependent kinase (CDK1), p53, Bcl-2 and Bax in HepG2 cells (Fig. 3) was estimated by quantitative real-time PCR (qRT-PCR). Results revealed that compared to the untreated HepG2 group, the expression level of p21, p53 and Bax was increased (upregulated) in A.judaica treated HepG2 cell line, whereas, the expression level of Bcl-2, CycB1 and CDK1genes was down-regulated (decreased) in HepG2 treated with A. Judaica compared to the untreated HepG2 cell line.
DISCUSSION
Medicinal herbs have shown their pharmacological activities as a new drug reaching the market.33,34 Different forms of cancer are constantly gaining resistance to current drugs, creating a need for the discovery of new drugs. A. Judaica extract showed remarkable cytotoxic activity against the tested liver cancer cell lines in a dose-dependent manner. This could be due to the presence of artemisinin and other sesquiterpene lactones, which have been found in A. Judaica.35,36 Artemisinin which is a sesquiterpene lactone demonstrated antitumor agent and anticancer activity when tested in vitro and in vivo.37,38
These results agree with previous studies reporting biologically active compounds from the medicinal plant A. Judaica possess sesquiterpene lactones, essential oils and other derived phytochemicals as active constituents which have shown medicinal properties such as anticancer activities (cytotoxicity and antiproliferation) for cancer cell lines, with no cytotoxic effect for normal cell lines exposed to the plant extract.31
The cell cycle arrest was analyzed using flow cytometry-based cell cycle distribution. The results indicated that A. Judaica extract inhibited the liver cancer (HepG2) cell cycle through triggering G2/M phase arrest involved transcriptional suppression, where the percentage of HepG2 cells at the G2/M phase was increased and reached about 50 % (49% ± 2.08) compared to 21% for the untreated cell line. That coincided with Lang et al.,32 who found that Artemisia extracts induced accumulation of multinucleated cancer cells within 24 h of treatment, increased the number of cells in the S and G2/M phases of the cell cycle, followed by loss of mitochondrial membrane potential, caspase 3 activation, and formation of an apoptotic cell population. These data indicated that A. Judaica exhibited an antiproliferative effect by a cell cycle blocking at the G2/M phase and apoptosis mediated cytotoxicity in carcinoma cells. However, Honda et al.,39 found that equipotent concentrations of A.judiaca extract highly arrested the cell cycle in the G0/G1 phase of cancer cell lines.
The current study inspected the ability of A.judiaca crude extracts to induce apoptosis where it is known to be the most promising pathway for a cancer therapy strategy. Consequently, results showed that the extract had the highest cytotoxic activity, have ingredients or molecules involved in the activation mechanisms of one or more antiproliferative pathways. A. Judaica medicinal plant induces apoptotic process which is modulated by different tumour suppressor genes including p53. The P53 induces apoptotic cell death by direct or indirect change expression of the Bcl-2 family of proteins, Bcl-2 and Bax.40 The Bcl-2 gene is an anti-apoptotic gene that suppresses initiation steps of apoptosis via inhibition of the pro-apoptotic proteins.41 P53 may modulate susceptibility of cells to apoptosis by downregulation of BCL-2 and causing up-regulation of BAX 27 and that coincides with our findings.
The cell cycle is mediated by the activation of a highly conserved protein kinase family, the cyclin-dependent kinases (CDKs).42 Cyclins can activate CDKs by forming complexes with Cdks, and these cyclin/CDK complexes are cell cycle regulators. Among them, the cyclin B1/CDK1 complex, in which B-type cyclins associate with CDK1, is considered as one of the primary regulators of transition from the G2 to M phase. This complex was originally discovered and defined as the maturation-promoting factor or M phase-promoting factor (MPF).43 In our study, the down-regulation of CycB1, Bcl-2 and CDK1 gene expression as responsible genes for Cyclins and cyclin-dependent kinase (the cell cycle regulators cyclin/CDK complexes) in HepG2 treated with A. Judaica compared to the untreated HepG2 cell line at G2/M phases of the cell cycle, confirm these findings.
Sesquiterpene lactone as active constituents of Artemisia was found to be a potent apoptotic inducer in cancer cells via multiple pathways especially in depletes intracellular GSH, disrupts cellular redox balance, depletes intracellular thiols, 27 triggers an intracellular reactive oxygen species that lead to mitochondrial dysfunction: loss of mitochondrial membrane potential, the onset of mitochondrial membrane transition, and release of mitochondrial pro-apoptotic proteins,28 decreases anti-apoptotic Bcl-2 protein expression and induce apoptosis.29
CONCLUSION
Based on the findings of the study, the crude extract of A. Judaica was found to had anticancer properties specific for human tumour cells, without any toxicity on normal cells. These results indicated that A. Judaica has an antiproliferative effect on HepG2 cell lines. Moreover, A. Judaica crude extract evaluated to identify the mechanisms behind the toxicity such as cell cycle blocking at the G2/M phase using flow cytometry. The real time-PCR to measure the mRNA levels of cyclin B1 and cyclin-dependent kinase (CDK1) genes (cell cycle regulator genes) and Bcl2, where those genes showed down-regulation expression levels in HepG2 treated with A. Judaica compared to the untreated cell line. Gene expression of ribonucleic acid (RNA) study confirms A. Judaica extract to induction of apoptosis via activation of p53, p21 and Bax and inactivated of Bcl2. Taken together, these data suggest that A. Judaica could be a promising candidate species as a source of anticancer molecules. Finally, natural products such as A.judaica are attractive sources for the development of new medicinal and therapeutic agents. Those with anticancer potential and apoptosis may be more selective and have weaker adverse effects on normal cells compared to chemotherapy used for cancer treatment. So, these research trends indicate that natural products will be among the most important sources of new drugs for cancer prevention and treatment in human being.
Conflicts of Interest: The authors declare that they have no conflicts of interest related to this study. This work was financially supported by us.
Acknowledgements: Authors acknowledge the immense help received from the scholars whose articles are cited and included in references to this manuscript. The authors are also grateful to authors/editors/publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
Source of funding: We expend from our account.
References:
-
Fabiani R. Antitumoral Properties of Natural Products: Editorial. Molecules 2020;25:650.
-
Tang ZY. Hepatocellular Carcinoma-Cause, Treatment and Metastasis: Review. World J Gastroenterol 2002;7(4): 445–454.
-
Ekor M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front Neurol 2014;4:177.
-
Boulaaba M, Khaoula M, Soninkhishig T, Junkyu H, Abderrazak S, Kiyokazu K, et al. In Vitro Antiproliferative Effect of Arthrocnemum indicum extracts on Caco-2 Cancer Cells through Cell Cycle Control and Related Phenol LC-TOF-MS Identification. Evidence-Based Complement Altern Med 2013;2013: 529375.
-
Efferth T, Koch E: Complex interactions between phytochemicals. The multi-target therapeutic concept of phytotherapy. Curr Drug Targets 2011;12(1): 122-32.
-
Chen H, Jin ZL, Xu H. MEK/ERK signalling pathway in apoptosis of SW620 cell line and inhibition effect of resveratrol. Asian Pac J Trop Med 2016;9:49–53.
-
Millimouno FM, Dong J, Yang L, Li J, Li X. Targeting apoptosis pathways in cancer and perspectives with natural compounds from Mother Nature. Cancer Prev Res 2014;7:1086–1107.
-
Singh S, Sharma B, Kanwar SS, Kumar A. Lead phytochemicals for anticancer drug development. Front Plant Sci 2016;7:1667.
-
Aung TN, Qu Z, Kortschak RD, Adelson DL. Understanding the effectiveness of natural compound mixtures in cancer through their molecular mode of action. Int J Mol Sci 2017;18:656.
-
Giordano OS, Guerreiro E, Pestchanker MJ, Guzman J, Pastor D, Guardia T. The gastric cytoprotective effect of several sesquiterpene lactones. J Nat Prod 1990;53:803-809.
-
Rustaiyan A, Ezzatzadeh E. Sesquiterpene lactones and pentamethoxylated flavone from Artemisia kulbadica. Asian J Chem 2011;23:1774–1776.
-
Akrou A, Gonzalez LA, Hajer EJ. Antioxidant and antitumor activities of Artemisia campestris a tnd Thymelaeahirsuta from southern Tunisia. Food Chem Toxicol 2011;49:342-347.
-
Nasr FA, Noman OM, Mothana RA, Alqahtani AS, Al-Mishari AA. Cytotoxic, antimicrobial and antioxidant activities and phytochemical analysis of Artemisia judaica and A. sieberi in Saudi Arabia. Afr J Pharm Pharmacol 2020;14(8):278-284.
-
Elansary HO, Abdelgaleil SAM, Mahmoud EA, Yessoufou K, Elhindi K, El-Hendawy S. Effective antioxidant, antimicrobial and anticancer activities of essential oils of horticultural aromatic crops in northern Egypt. BMC Complemen Altern Med 2018;18:2.
-
Torres A, Vargas Y, Uribe D, Carrasco C, Torres C, Rocha R, et al. Pro-apoptotic and anti-angiogenic properties of the α/β-thujone fraction from Thuja occidentalis on glioblastoma cells. J Neuro Oncol 2016;128:9–19.
-
Alzweiri M, Alrawashdeh IM, Bardaweel SK. The development and application of novel IR and NMR-based model for the evaluation of carminative effect of Artemisia judaica L. essential oil. Int J Anal Chem 2014;627-636.
-
Abad MJ, Bedoya LM, Apaza L, Bermejo P. The Artemisia L. genus: a review of bioactive essential oils. Molecules 2012;17(3):2542-2566.
-
Chhetri BK, Al-Sokari SS, Setzer WN, Ali NAA. Essential oil composition of Artemisia abyssinica from three habitats in Yemen. Am J Essent Oils Nat Prod 2015;2(3):28-30.
-
Liu CZ, Murch SJ, El-Demerdash M, Saxena PK. Artemisia Judaica L.: micropropagation and antioxidant activity. J Biotechnol 2004;110:63-71.
-
Nezhadali M, Parsa M. Study of volatile compounds in Artemisia Sagebrush from Iran using HS/SPME/GC/MS. Int J Environ Sci Dev 2010;3:287-289.
-
Abdelgaleil SA, Abbassy MA, Belal AS, Abdel Rasoul MA. Bioactivity of two major constituents isolated from the essential oil of Artemisia Judaica L. Bioresour Technol 2008;99:5947-5950.
-
Bora KS, Sharma A. The genus Artemisia: a comprehensive review. Pharm Biol 2011;49:101-109.
-
Abood S, Eichelbaum S, Mustafi S, VeisagaML, López LA, Barbieri M. Biomedical Properties and Origins of Sesquiterpene Lactones, with a Focus on Dehydroleucodine. Nat Prod Commun 2017;12(6):995-1005.
-
Nigam M, Atanassova M, Mishra AP, Pezzani R, Devkota HR, Plygun S, et al. Bioactive Compounds and Health Benefits of Artemisia Species: Review. Nat Prod Commun 2019;15:1-17.
-
Charchari S. The essential oil of Artemisia Judaica L. from Algeria. J Essent Oil Res 2002;14:16-17.
-
El-Sharabasy HB. Acaricidal activities of Artemisia Judaica L. extracts against Tetranychus urticae Koch and its predator Phytoseiulus persimilis Athias Henriot (Tetranychidae: Phytoseiidae). J Biopestic 2010;3:514-519.
-
Choi JH, Ha J, Park JH, Lee JY, Lee YS, Park HJ, et al. Costunolide triggers apoptosis in human leukemia U937 cells by depleting intracellular thiols. Jpn J Canc Res 2002;93:1327-1333.
-
Lee M, Lee K, Chi S, Park J. Costunolide induces apoptosis by ROS-mediated mitochondrial permeability transition and cytochrome C release. Biol Pharm Bull 2001;24:303-306.
-
Park HJ, Kwon SH, Han YN, Choi JW, Miyamoto K, Lee SH, et al. Apoptosis-inducing costunolide and a novel acyclic monoterpene from the stem bark of Magnolia sieboldii. Arch Pharm Res 2001;24:342-348.
-
Taleghani A, Ahmad Emami S, Tayarani-Najaran Z. Artemisia a promising plant for the treatment of cancer, Bioorg Med Chem 2019;28(1):115180.
-
Mohammed HY, Al-Senosy NK, Ebeed NM, Fahmy Kh. Estimation of the anticancer effect of methanolic extract of artemisia herba alba on human cancer cell lines. AUJASCI, Arab Univ J Agric Sci 2019;27(4):2215-2222.
-
Lang SJ, Schmiech M, Hafner S, Paetz C, Steinborn C, Huber R, et al. Antitumor activity of an Artemisia annua herbal preparation and identification of active ingredients. Phytomedicine 2019;62.
-
Atanasov AG, Waltenberger B, Pferschy-Wenzig EM, Linder T, Wawrosch C. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol Adv 2015; 33(8):1582-1614.
-
Che CT, Zhang H. Plant natural products for human health. Int J Mol Sci 2019;20(4):830.
-
Arab HA, Rahbari S, Rassouli A, Moslemi M, Khosravirad F. Determination of artemisinin in Artemisia sieberi and anticoccidial effects of the plant extract in broiler chickens. Trop Anim Health Prod 2006;38(6):497-503.
-
Abbas ZK, Saggu S, Rehman H, Al Thbiani A, Ansari AA. Ecological variations and role of heat shock protein in Artemisia Judaica L. in response to temperature regimes of Tabuk, Saudi Arabia. Saudi J Biol Sci 2017;24(6): 1268-1273.
-
Appalasamy S, Lo KY, Ch'ng SJ, Nornadia K, Othman AS, Chan LK. Antimicrobial activity of artemisinin and precursor derived from in vitro plantlets of Artemisia annua L. BioMed Res Int 2014:215872.
-
Ferreira JF, Luthria DL, Sasaki T, Heyerick A. Flavonoids from Artemisia annua L. as antioxidants and their potential synergism with artemisinin against malaria and cancer. Molecules 2010;15(5):3135-3170.
-
Honda A, Yamashita K, Miyazaki H, Shirai M, Ikegami T, Xu G, et al. Highly sensitive analysis of sterol profiles in human serum by LC-ESI-MS/MS. J Lipid Res 2008;49:2063–2073.
-
Reed JC. Dysregulation of apoptosis in cancer. J Clin Oncol 1999;17:2941-2953.
-
Youle RJ, Strasser A. The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol 2008;9:47–59.
-
Stewart, ZA, Westfall MD, Pietenpol JA. Cell-cycle dysregulation and anticancer therapy. Trends Pharmacol Sci 2003;24(3):139-145.
-
Shangguan WJ, Li H, Zhang YH. Induction of G2/M phase cell cycle arrest and apoptosis by ginsenosideRf in human osteosarcoma MG?63 cells through the mitochondrial pathway. Oncol Rep 2014;31(1):305-313.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License