IJCRR - 13(6), March, 2021
Pages: 124-128
Date of Publication: 20-Mar-2021
Print Article
Download XML Download PDF
Diagnostic Implication of Phosphatase and Tensin Homolog Expression in Endometrial Lesions
Author: Abu Shahma Khan AS, Alam K, Akhtar K, Khan T, Alam F
Category: Healthcare
Abstract:Background: Expression of Phosphatase and Tensin homolog (PTEN) is lower in endometrial cancer in comparison with nor�mal or hyperplastic endometrium. Loss of PTEN function by mutational or other mechanisms is an early event in endometrial tumorigenesis. Objective: In this study, we evaluated PTEN gene expression in hyperplastic and malignant endometrium to assess its diag�nostic implications. Methods: This study was conducted on women presenting with abnormal uterine bleeding (AUB), attending the Outpatient and Inpatient Clinics of Obstetrics and Gynecology. Endometrial biopsy sections were routinely stained with HandE stain and im�munohistochemical staining for PTEN expression was performed in all the endometrial lesions. Results: The study comprised 278 cases of endometrial hyperplasias and carcinomas. In simple hyperplasia without atypia (n=143), 80.0% showed intense PTEN positivity and 20.0% showed moderate PTEN positivity. Simple hyperplasia with atypia (n=15), 93.3% cases showed moderate PTEN positivity and 6.7% case showed intense PTEN positivity. Complex hyperplasia without atypia (n=76), 14.4% cases showed mild PTEN positivity and moderate PTEN positivity was seen in 85.6% cases. Com�plex hyperplasia with atypia (n=34), 85.3% cases showed mild PTEN positivity and absent staining in14.7% cases. Adenocarci�noma cases showed mild PTEN positivity in 10.0% and absent staining in 90.0% cases. Conclusions: PTEN expression decreases as the lesion progress from benign to frank malignancy, and loss of PTEN function is an early event in the pathogenesis of endometrial carcinogenesis.
Keywords: PTEN, Endometrial hyperplasia, Carcinoma, Immunohistochemistry
Full Text:
INTRODUCTION
Endometrial diseases are the most common gynaecological disorders affecting women worldwide.1 They constitute around 70.0% of all gynecologic consultancies in the peri-menopausal and postmenopausal age group.2 They usually present with abnormal uterine bleeding (AUB), which is the most common and challenging problem presenting to the gynaecologist regardless of the age of the women.3 The International Federation of Gynaecology and Obstetrics ( FIGO) working group on menstrual disorders developed a FIGO classification system for the causes of AUB in non-gravid women of reproductive age group.4 Its main categories are: polyp, adenomyosis, leiomyoma, hyperplasia and malignancy, coagulopathy, ovulation, endometrial, iatrogenic and not yet classified.
Endometrial hyperplasia and malignancy are important neoplastic lesions causing AUB. Endometrial hyperplasia is characterized by an increased proliferation of endometrial glands relative to stroma resulting in increased gland to stromal ratio than normal endometrium. The hyperplastic glands vary in shape and size and may shows cytologic atypia, which may progress or co-exist with endometrial carcinoma. Hyperesterogenic state is most commonly associated with endometrial hyperplasia. Prolonged estrogenic stimulation of the endometrium may be due to anovulation, obesity, polycystic ovarian disease, functional granulosa cell tumors of the ovary and estrogen replacement therapy.5
The WHO 2014 classification divides endometrial hyperplasia into 2 categories: hyperplasia without atypia and atypical hyperplasia/endometrioid intraepithelial neoplasia. This reduction in to 2 categories reflects a new understanding of molecular genetic changes. Hyperplasia without atypia showed no relevant genetic changes. They are benign entities and will regress with time after the endocrine milieu (physiological gestagen levels) has normalized. Atypical endometrial hyperplasias exhibit many mutations typical for invasive endometrioid endometrial carcinoma. In approximately 60.0% of such cases, patients have coexisting invasive cancer or are at increased risk of developing invasive cancer.6,7
Phosphate and Tensin Homologue (PTEN) is a tumor suppressor gene. Expression of PTEN is lower in endometrial cancer than in normal endometrium. It is involved in the regulation of focal adhesion, cellular migration, and tumor cell proliferation and somatic mutations of this gene have been identified in a large number of other human malignancies.8 Although most pre-cancers and cancers had a mutation in only one PTEN allele, endometrioid endometrial adenocarcinomas showed complete loss of PTEN protein expression in 61% of cases, and 97.0% cases showed at least some diminution in expression. Cancers and most pre-cancers exhibited contiguous groups of PTEN negative glands, while endometria altered by unopposed estrogens showed isolated PTEN-negative glands. Loss of PTEN function by mutational or other mechanisms is an early event in endometrial tumorigenesis, and it may occur in response to known endocrine risk factors and offers an informative immune-histochemical biomarker for premalignant disease. Individual PTEN-negative glands in estrogen-exposed endometria are the earliest recognizable stage of endometrial carcinogenesis. Proliferation into dense clusters that form discrete premalignant lesion follows.9 PTEN mutations have been seen in about 55% of patients with endometrial hyperplasia. 10 Loss of PTEN function has, therefore, been proposed as an early event in the pathogenesis of endometrioid carcinogenesis.
Materials and methods
This study was performed on 278 women attending the Outpatient and Inpatient Departments of Obstetrics and Gynecology and Pathology with complaints of abnormal uterine bleeding (AUB), after approval from the hospital ethics committee. A thorough clinical history of patients was collected and complete physical examination findings and other relevant investigations were recorded. All the endometrial biopsies/curettage and hysterectomy specimen sent for histopathological evaluation with a history of abnormal uterine bleeding were included in the study, while all other malignancies of the female genital tract and patients not giving informed consent were excluded. Specimens were processed in 10% neutral buffered formalin (10 ml of 40% formaldehyde diluted in 90 ml of water). Tissues were fixed for 24 hours with two changes of formalin and embedded in paraffin which was further cut into 4-5 µm thick sections. The sections were routinely stained with haematoxylin and eosin (H&E) stain. Immunohistochemical staining using the PTEN monoclonal antibodies was performed on diagnosed cases of endometrial lesions. The various histomorphological and immunohistochemical patterns were identified and classified according to the percentage of cell stained and intensity of staining on immunostained slides. PTEN staining pattern was evaluated as: absent staining- 0, mild-1+, moderate-2+, Intense- 3+. 11
Statistical analysis was performed using Fisher’s exact test. Data was statistically analyzed using SPSS software version (20.0). Chi-square tests were used in the analysis of dichotomous or categorical variables. When expected cell frequencies were <5, the Fisher exact test was used. p-value of 0.05 or less was considered to be statistically significant.
RESULTS
The present study included 278 patients between ages of 11-70 who presented with abnormal uterine bleeding (AUB). The maximum number of patients were in the 5th decade, 112 (40.3%) followed by 83 (29.9%) patients in the 4th decade and 38 (13.7 %) patients in the 3rd decade of life.
Majority of the patients in our study were parous 261 cases (93.8%) and nulliparous constituted only 17 cases (6.1%). The incidence of abnormal uterine bleeding was high in grand multipara 109 (39.2%) and multipara 104 (37.4%) as compared with nulliparous 17(6.1%) and primiparous 48 (17.3%) females.
Out of the 278 cases, there were 268 (96.4%) cases of endometrial hyperplasia and 10 (0.36%) cases of endometrial cancer. Table 1 Endometrial hyperplasia was the commonest pathology seen in the perimenopausal age group (41-50 years). This included simple hyperplasia without atypia in 74 (51.7%) cases followed by complex hyperplasia without atypia in 46(60.5%) cases. The other common lesions encountered were endometrial carcinoma in 8 (80.0%) cases and atypical hyperplasia in 3 (6.2%) cases. Table 2
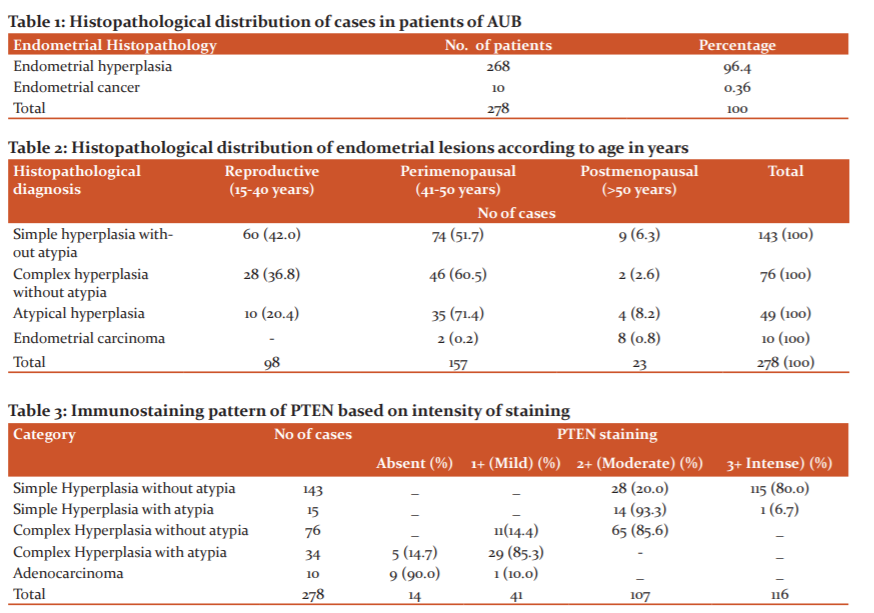
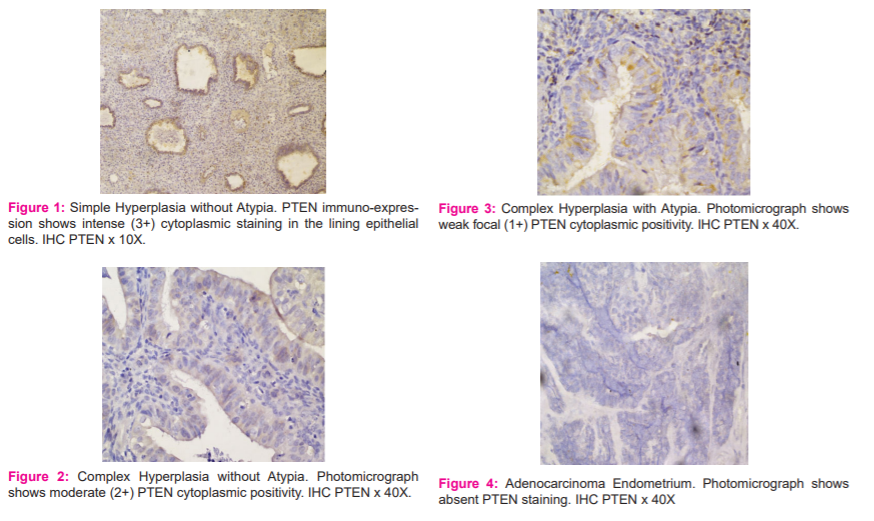
Of the 268 cases of endometrial hyperplasia, the most common subtype was simple hyperplasia without atypia in 143 (51.4%) cases (Figure 1) followed by complex hyperplasia without atypia in 7 (27.3%) cases. (Figure 2) Complex hyperplasia with atypia was seen in 34 (12.2%) cases (Figure 3) and the least common variant was simple hyperplasia with atypia seen in 15 (5.4 %) cases. Out of 10 cases of endometrial carcinoma 7(70.0%) cases presented with grade II, 2 (20.0%) cases presented with grade III, 1 (10.0%) case presented with grade I. 4 (40.0%) cases presented with <1/2 myometrial invasion, 3 (30.0%) cases presented with >1/2 myometrial invasion and 3 (30.0%) cases did not show myometrial invasion. 3 (30.0%) cases presented with lymphovascular invasion and 7 (70.0%) cases showed no lymphovascular involvement. Hence, accordingly 6 (60.0%) cases presented with stage II, 2 (20.0%) cases presented with stage III, 1 (10.0%) case each presented with stage I and stage IV.
Out of 7 cases of stage I/II endometrial carcinoma 1/7 (14.3%) showed mild PTEN positivity and 6/7 (85.7%) showed absent PTEN staining. All of the 3 cases of stage III/IV endometrial carcinomas showed absent PTEN staining. 3 cases with absent lymphovascular invasion showed absent PTEN staining and 7 cases of lymphovascular invasion showed mild PTEN positivity. Histologically, 9 (90%) cases of endometrioid endometrial carcinomas showed absent PTEN staining (Figure 4) and 1 (10%) case of serous papillary carcinoma showed mild PTEN positivity.
Out of 143 cases of simple hyperplasia without atypia, 115 (80%) cases showed intense (3+) PTEN positivity and 28 (20%) cases showed moderate (2+) PTEN positivity. Out of 15 cases of simple hyperplasia with atypia, 14 (93.3%) cases showed moderate 2+ PTEN positivity, and 1 (6.7%) case showed intense (3+) PTEN positivity. Out of 76 cases of complex hyperplasia without atypia 11 (14.4) cases showed mild (1+) PTEN positivity and moderate (2+) PTEN positivity was seen in 65 (85.6%) cases. Complex hyperplasia with atypia was seen in 34 cases, 29 (85.3%) cases showed mild (1+) PTEN positivity and absent staining in 5 (14.7%) cases. Adenocarcinoma cases showed mild (1+) PTEN positivity in 1/10 (10%) case and absent staining in 9/10 (90%) cases. Table 3
Discussion
Abnormal uterine bleeding is one of the most commonly encountered complaints in gynaecological practice. It comprises more than two-thirds of all gynecological consultations in the peri and postmenopausal years.12 The causes may vary with respect to the age of patients. Histopathological diagnosis of endometrial biopsy and curettage play an important role in identifying the various disease spectrum causing uterine bleeding, especially in developing countries with limited resources. 40.3% of our patients were in the 5th decade of life, a finding in concordance with the study of Abdullah and Bondagji and Saraswathi et al., who have reported incidence of 32.1% and 33.5% respectively in the 5th decade of life.13,14 A slightly higher incidence of 48.1% was reported by Zhang et al and 59.1% by Rena et al.15,16
93.8% of our patients were parous, and the incidence of abnormal uterine bleeding was highest in grand multipara, who constituted 39.2% cases. These findings were consistent with reports of Sharma et al.17
Out of 278 patients, 39.6% of the patients presented with menorrhagia followed by 12.6% cases of infertility, findings concordant with Kaur et al.18 However Zhang et al have reported a slightly higher percentage of patients with menorrhagia at 51.9%.15 We reported 7.9% incidence of postmenopausal bleeding in our study, which is comparable with Agrawal et al.19 However, Rifat and Mahmoud had reported a slightly higher percentage (16.3%) of postmenopausal bleeding.20 This might be probably due to lesser number of patients in our study in this age group. Our study revealed that the occurrence of menstrual disorders increases with advancing age. Excessive bleeding was seen in 41–50 years age group in our study. A similar incidence was reported by Zhang et al and Mahapatra and Mishra et al. in their study.15,21
The maximum incidence of endometrial hyperplasia was found in the age group 40-49 years, which was similar to the study by Doroiswami et al.22 Hyperplasias occur usually in perimenopausal age group due to failure of ovulation. It is due to persistent unriped follicles which exposes the endometrium to an abnormally and excessive prolonged estrogenic secretion. Our study comprised of 51.4% cases of simple hyperplasia without atypia, with majority seen in the perimenopausal age group, a finding similar to Jairajpuri et al and Munro, who have reported 64.4% to 66.6% cases respectively.23,24 Complex hyperplasia without atypia (CH) was seen in 76/278 (27.3%) cases similar to Rifat and Mahmoud's reports.[20] There were 5.4% cases of simple atypical hyperplasia and 12.2% cases of complex atypical hyperplasia in our study, findings concordant with reports of Sajitha et al.25 Endometrial carcinoma was the least common pathology in our study, found in 10 (0.5%) cases. Similar results have been reported by Riaz et al and Abdullah et al.26,27
Diagnosis of endometrial hyperplasia is important because they are thought to be a precursor of endometrial carcinoma. The overall increased risk of progression of hyperplasia to cancer is 5-10.0%.28 Simple, complex, simple atypical (SAH) and complex atypical hyperplasia (CAH) have progression risks of 1.0%, 3.0%, 8.0% and 29.0% respectively to carcinoma.28
The mean scoring of PTEN staining decreased from simple hyperplasia without atypia to complex hyperplasia with atypia and was the least in adenocarcinoma. It can be seen that the intensity of PTEN decreased from benign to malignant lesions. Our finding was in concordance with the study by Scully et al, Westin et al and Ramalingam et al. 28-30
Conclusion
Loss of PTEN function by mutational or other mechanisms is an early event in endometrial tumorigenesis and offers an informative immune-histochemical biomarker for premalignant and malignant endometrial lesions. Hence, PTEN antibodies can be used to detect cancer at an early stage and can be used to target adjuvant therapy in endometrial cancers.
Conflict of interest: Nil
Source of funding: Nil
References:
-
Khan R, Sherwani RK, Rena S. Clinicopathologic pattern in women with DUB. Iran J Pathol 2016;11:12-16.
-
Mahajan N, Aggarwal M, Bagga A. Health issues of menopausal women in North India. J Midlife Health 2012;3:84-88.
-
Babacan A, Gun I, Kizilaslan C. Comparison of transvaginal ultrasonography and hysteroscopy in the diagnosis of uterine pathologies. Int J Clin Exp Med 2014;7:764-769.
-
Munro MG, Critchley HO, Broder MS, Fraser IS. FIGO classification system for causes of abnormal uterine bleeding in nongravid women of reproductive age. Int J Gynecol Obstet 2011;113:3?13.
-
Norris HJ, Connor MP, Kurman RJ. Preinvasive lesions of the endometrium. Clin Obstet Gynecol 1986;13:725-738.
-
Owings RA and Quick CM. Endometrial intraepithelial neoplasia. Arch Pathol Lab Med 2014;138: 484-491.
-
Trimble CL, Method M, Leitao M, Lu K, Ioffe O, Hampton M, et al. Society of Gynecologic Oncology Clinical Practice Committee. Management of endometrial precancers. Obstet Gynecol 2012;120: 1160-1175.
-
Aggelis S, Michail V, Thivi V, Viktoria K, Aikaterin T, Andreas C, et al. Expression of p53 and PTEN in human primary endometrial carcinomas: Clinicopathological and immunohistochemical analysis and study of their concomitant expression. Oncol Lett 2019;17:4575-89.
-
Sasnauskiene A, Jonusiene V, Krikstaponiene A, Butkyte S, Dabkeviciene D, Kanopiene D, et al. NOTCH1, NOTCH3, NOTCH4, and JAG2 protein levels in human endometrial cancer. Medicine 2014;50:14-18.
-
Daniilidou K, Frangou-Plemenou M, Grammatikakis J, Grigoriou O, Vitoratos N, Kondi-Pafiti A. Prognostic significance and diagnostic value of PTEN and p53 expression in endometrial carcinoma. A retrospective clinicopathological and immunohistochemical study. J BUON 2013;18:195-201.
-
Marques O, Santacana M, Valls J, Pallares J, Mirante's C, Gatius S, et al. Optimal protocol for PTEN immunostaining; Role of analytical and preanalytical variables in PTEN staining in normal and neoplastic endometrial, breast, and prostatic tissues. Hum Pathol 2014;45:522-532.
-
Mahajan N, Aggarwal M, Bagga A. Health issues of menopausal women in North India. J Midlife Health 2012; 3: 84-8.
-
Abdullah LS and Bondagji NS. Histopathological pattern of endometrial sampling performed for abnormal uterine bleeding. Bahrain Med Bull 2011;33:195?200.
-
Saraswathi D, Thanka J, Shalinee R, Aarthi R, Jaya V, Kumar PV, et al. Study of endometrial pathology in abnormal uterine bleeding. Obstet Gynecol Ind. 2011;61:424-430.
-
Zhang C, Sung CJ, Quddus MR. Association of ovarian hyperthecosis with endometrial polyp, endometrial hyperplasia, and endometrioid adenocarcinoma in postmenopausal women: a clinicopathological study of 238 cases. Hum Pathol 2017; 59:120-124.
-
Rena DK, Tanma S, Amitabh H, Basanta S. Histopathologic spectrum of Endometrial changes in Women presenting with abnormal uterine bleeding with reference to endometrial malignancies: A two Years Hospital-Based Study. Annals Applied Bio Sci 2016;3:152-156.
-
Sharma S, Makaju R, Shrestha S, Shrestha A. Histopathological Findings of Endometrial Samples and its Correlation Between the Premenopausal and Postmenopausal Women in Abnormal Uterine Bleeding. Kathmandu University Med J 2015;12:275-278.
-
Kaur P, Kaur A, Suri AK, Sidhu H. A two year histopathological study of endometrial biopsies in a teaching hospital in Northern India. Indian J Pathol Oncol 2016;3:508-511.
-
Agrawal S, Mathur S, Vaishnav K. Histopathological study of endometrium in abnormal uterine bleeding in all age groups in western Rajasthan. Int J Basic Applied Med Sci 2014; 4:15-18.
-
Rifat AG and Mahmoud MM. Endometrial Histopathological changes in women with Abnormal Uterine bleeding in Kirkuk City- A Clinicopathological Study. Med J Babylon. 2013;10:567-82.
-
Mahapatra M and Mishra P. Clinicopathological evaluation of abnormal uterine bleeding. J Health Res Rev 2015;2:45-46.
-
Doraiswami S, Johnson T, Rao S, Rajkumar A, Vijayaraghavan J, Panicker VK et al. Study of endometrial pathology in abnormal uterine bleeding. Indian J Obstet Gynecol 2011; 61:426-427.
-
Jairajpuri ZS, Rana S, Jetley S. Atypical uterine bleeding-Histopathological audit of endometrium. A study of 638 cases. Al Ameen J Med Sci 2013;6:21-28.
-
Munro MG. Practical aspects of the two FIGO systems for management of abnormal uterine bleeding in the reproductive years. Best Pract Res Clin Obstet Gynaecol 2017;40:3-22.
-
Sajitha K, Padma SK, Shetty KJ, Kishan HL, Permi HS, Hegde P et al. Study of histopathological patterns of endometrium in abnormal uterine bleeding. Chris Med J Health Res 2014;1:76-77.
-
Riaz S, Ibrar F, Dawood NS, Jabeen A. Endometrial pathology by endometrial curettage in menorrhagia in premenopausal age group. J Ayub Med Coll Abbottabad 2010; 22:161-164.
-
Abdullah LS, Rana K, Bondagji NS. Histopathological pattern of endometrial sampling performed for abnormal uterine bleeding. Bahrain Med Bull 2011;33:195?200.
-
Scully MM, Palacios-Helgeson LK, Wah LS, Jackson TA. Rapid estrogen signaling negatively regulates PTEN activity through phosphorylation in endometrial cancer cells. Horm Cancer 2014;5:218-231.
-
Westin SN, Ju Z, Broaddus RR, Krakstad C, Li J, Pal N et al. PTEN loss is a context-dependent outcome determinant in obese and non-obese endometrioid endometrial cancer patients. Mol Oncol 2015;9:1694-1703.
-
Ramalingam P, Masand RP, Eucher ED, Malpica A. Undifferentiated carcinoma of the endometrium: An expanded immunohistochemical analysis including PAX-8 and basal-like carcinoma surrogate markers. Int J Gynecol Pathol 2016;35:410-418.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License